Initial and Repeated Point Prevalence Surveys to Inform SARS-CoV-2 Infection Prevention in 26 Skilled Nursing Facilities — Detroit, Michigan, March–May 2020
Weekly / July 10, 2020 / 69(27);882-886
On July 1, 2020, this report was posted online as an MMWR Early Release.
Guillermo V. Sanchez, MSHS, MPH1; Caitlin Biedron, MD1; Lauren R. Fink, MPH2; Kelly M. Hatfield, MSPH1; Jordan Micah F. Polistico, MD3,4; Monica P. Meyer, MS, MPH3,4; Rebecca S. Noe, MN, MPH1; Casey E. Copen, PhD1; Amanda K. Lyons, MS1; Gonzalo Gonzalez, DNP2; Keith Kiama2; Mark Lebednick2; Bonnie K. Czander2; Amen Agbonze2; Aimee R. Surma, MS2; Avnish Sandhu, DO3,4; Valerie H. Mika, MS4; Tyler Prentiss, MA5; John Zervos, JD5; Donia A. Dalal2; Amber M. Vasquez, MD1; Sujan C. Reddy, MD1; John Jernigan, MD1; Paul E. Kilgore, MD4; Marcus J. Zervos, MD4,5; Teena Chopra, MD3,4; Carla P. Bezold, ScD2; Najibah K. Rehman, MD2 (View author affiliations)
View suggested citationSummary
What is already known about this topic?
Symptom-based screening in skilled nursing facilities (SNFs) is inadequate to detect SARS-CoV-2 transmission. Repeated point prevalence surveys can identify asymptomatic cases during outbreaks.
What is added by this report?
Repeated point prevalence surveys at 26 Detroit SNFs identified an attack rate of 44%; within 21 days of diagnosis, 37% of infected patients were hospitalized and 24% died. Among 12 facilities participating in a second survey and receiving on-site infection prevention and control (IPC) support, the percentage of newly identified cases decreased from 35% to 18%.
What are the implications for public health practice?
Repeated point prevalence surveys in SNFs can identify asymptomatic COVID-19 cases, inform cohorting and IPC practices, and guide prioritization of health department resources.
Discussion
Facility-wide testing conducted among residents living in 26 SNFs in an urban Detroit jurisdiction with high SARS-CoV-2 prevalence identified an overall attack rate of 44%, a 37% COVID-19 hospitalization rate, and a 24% fatality rate amid ongoing and widespread SARS-CoV-2 transmission. Repeated point prevalence surveys enabled early identification of COVID-19 cases (including asymptomatic patients), informed cohorting and IPC practices, and guided prioritization of health department resources.
Despite barriers to implementing rapid repeated point prevalence surveys, this assessment demonstrates benefits of conducting repeated surveys in SNFs. Among facilities participating in both surveys, the percentage of new laboratory-confirmed cases declined from 35% to 18%, suggesting that facility-wide testing and on-site IPC support might have contributed to reductions in SARS-CoV-2 transmission. Following testing and establishment of a COVID-19 care unit, IPC assessment and consultation were critical to assisting facilities in targeting interventions to mitigate suspected causes of ongoing transmission. These included incomplete resident and health care personnel cohorting, continued reintroduction of the virus (e.g., from admission of residents with unknown COVID-19 status or residents requiring routine outpatient medical treatment, such as hemodialysis), and space limitations prohibiting use of private rooms to isolate residents whose infection status was unknown. Repeated point prevalence surveys might also improve patient outcomes by enabling earlier identification and initiation of clinical patient monitoring (e.g., assessing vital signs more frequently) and, when warranted, rapid transfer to acute care facilities.
The findings in this report are subject to at least four limitations. First, although asymptomatic health care personnel with SARS-CoV-2 infection are a likely source of transmission, health care personnel were not tested on the same day as were residents, and results of health care personnel testing were not available for inclusion in this report. Second, the long testing interval might influence interpretation of results. The first point prevalence survey occurred approximately 1 month after SARS-CoV-2 introduction in most facilities; therefore, asymptomatic cases identified during the first survey might represent residents who recovered from illness but still had positive RT-PCR test results. Further, the 14-day interval between the two surveys might have resulted in less effective case identification than a shorter interval would have. Third, testing methods in the two surveys varied, as did test characteristics across different platforms and specimen sources (6). Finally, at the time of manuscript drafting, data for repeated point prevalence surveys were available for only 12 out of 26 facilities, which limited our ability to fully describe ongoing SARS-CoV-2 transmission among Detroit SNFs.
When repeated point prevalence surveys are implemented as part of COVID-19 response strategies in SNFs, testing results should inform prompt and specific actions, such as 1) using transmission-based precautions for resident care and excluding health care personnel with positive test results from work; 2) strict cohorting of residents and health care personnel; 3) active clinical monitoring of confirmed COVID-19 cases; 4) managing safe transitions of care to and from outside facilities; and 5) discontinuing transmission-based precautions if a test-based strategy is used (7,8). In response to a confirmed case, CDC now recommends repeat testing (e.g., every 3–7 days) of all residents and health care personnel who previously had negative test results until testing identifies no new cases of COVID-19 among residents or health care personnel (9). Widescale testing activities should be integrated with intensified IPC support from local and state health departments.
Repeated point prevalence surveys coupled with IPC support might have reduced SARS-CoV-2 transmission in SNFs in Detroit and have the potential to improve outcomes among SNF residents. New cases continued to be identified during the second survey; however, reductions in 21-day hospitalization and mortality rates were observed throughout the implementation period. Future studies of COVID-19 in SNFs should further explore the impact of repeated point prevalence surveys on morbidity and mortality, the role of asymptomatic health care personnel in SARS-CoV-2 transmission, and the role of serologic testing in reopening SNFs following outbreaks. As the availability of SARS-CoV-2 testing increases, repeated point prevalence surveys and intensified IPC support from public health practitioners are essential components of COVID-19 IPC strategies in SNFs experiencing COVID-19 outbreaks.
Acknowledgments
Arrow Strategies; City of Detroit Emergency Medical Services; City of Detroit Fire Department; City of Detroit Health Department; City of Detroit Mayor’s Office; Detroit Medical Center; Henry Ford Global Health Initiative; Wayne State University College of Nursing; Wayne State University School of Medicine; volunteers who assisted in collecting and testing specimens from residents of skilled nursing facilities.
Corresponding author: Guillermo V. Sanchez, xkv4@cdc.gov.
1CDC COVID-19 Response Team; 2Detroit Health Department, Detroit, Michigan; 3Detroit Medical Center, Detroit, Michigan; 4Wayne State University, Detroit, Michigan; 5Henry Ford Health System, Detroit, Michigan.
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. John Zervos and Tyler Prentiss report grants from the United Way of Southeastern Michigan, Vattikuti Foundation, and Abbott Laboratories during the conduct of the study. Marcus J. Zervos reports grants from Pfizer, Merck, and Serono, outside the submitted work. No other potential conflicts of interest were disclosed.
References
- Arons MM, Hatfield KM, Reddy SC, et al.; Public Health–Seattle and King County and CDC COVID-19 Investigation Team. Presymptomatic SARS-CoV-2 infections and transmission in a skilled nursing facility. N Engl J Med 2020;382:2081–90. CrossRef PubMed
- Kimball A, Hatfield KM, Arons M, et al.; Public Health – Seattle & King County; CDC COVID-19 Investigation Team. Asymptomatic and presymptomatic SARS-CoV-2 infections in residents of a long-term care skilled nursing facility—King County, Washington, March 2020. MMWR Morb Mortal Wkly Rep 2020;69:377–81. CrossRef PubMed
- Dora AV, Winnett A, Jatt LP, et al. Universal and serial laboratory testing for SARS-CoV-2 at a long-term care skilled nursing facility for veterans—Los Angeles, California, 2020. MMWR Morb Mortal Wkly Rep 2020;69:641–4. CrossRef PubMed
- Food and Drug Administration. Emergency use authorizations. Silver Spring, MD: US Department of Health and Human Services, Food and Drug Administration; 2020. https://www.fda.gov/medical-devices/emergency-situations-medical-devices/emergency-use-authorizations-medical-devices
- CDC. Interim clinical guidance for management of patients with confirmed coronavirus disease (COVID-19). Atlanta, GA: US Department of Health and Human Services, CDC; 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-guidance-management-patients.html
- Rhoads DD, Cherian SS, Roman K, Stempak LM, Schmotzer CL, Sadri N. Comparison of Abbott ID Now, Diasorin Simplexa, and CDC FDA EUA methods for the detection of SARS-CoV-2 from nasopharyngeal and nasal swabs from individuals diagnosed with COVID-19. J Clin Microbiol 2020;JCM.00760–20. CrossRef PubMed
- CDC. Discontinuation of transmission-based precautions and disposition of patients with COVID-19 in healthcare settings (interim guidance). Atlanta, GA: US Department of Health and Human Services, CDC; 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/disposition-hospitalized-patients.html
- CDC. Responding to coronavirus (COVID-19) in nursing. Atlanta, GA: US Department of Health and Human Services, CDC; 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/nursing-homes-responding.html
- CDC. Testing guidance for nursing homes. Atlanta, GA: US Department of Health and Human Services, CDC; 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/nursing-homes-testing.html
* Hospitalizations with admission dates documented as 2 days before, through 21 days after, the specimen collection date for a positive SARS-CoV-2 test result were counted; deaths within 21 days of positive specimen collection date were counted. Missing dates were considered to be within 21 days of specimen collection.
† Total tested refers to residents tested at any time through May 8, 2020. Tested refers to residents tested in each period who were not previously known to have SARS-CoV-2 infection.
§ Dashes indicate that facilities did not participate in the follow-up survey.
 FIGURE. Skilled nursing facility residents with confirmed COVID-19 diagnosed by May 8, 2020, (A) by date of first positive SARS-CoV-2 test result (n = 1,190)*; (B) date of hospital admission (n = 331)†,§; and (C) date of death (n = 282)§,¶ — 26 facilities,** Detroit, March 7–May 29, 2020
FIGURE. Skilled nursing facility residents with confirmed COVID-19 diagnosed by May 8, 2020, (A) by date of first positive SARS-CoV-2 test result (n = 1,190)*; (B) date of hospital admission (n = 331)†,§; and (C) date of death (n = 282)§,¶ — 26 facilities,** Detroit, March 7–May 29, 2020
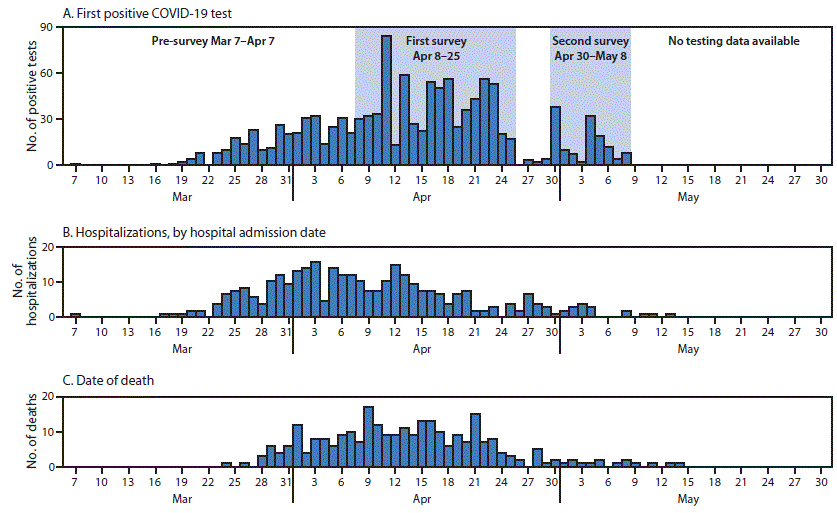
Abbreviation: COVID-19 = coronavirus disease 2019.
* Seventeen dates of first positive test results are not known.
† Five residents had multiple admissions; 120 had unknown hospitalization dates.
§ Hospitalization and mortality data were current as of May 29, 2020. Hospitalizations with admission dates documented as 2 days before, through 21 days after the specimen collection date for a positive SARS-CoV-2 test were counted; deaths within 21 days of positive specimen collection date were counted.
¶ Five dates of death are not known.
** Data from all 26 facilities are displayed; only 12 facilities were tested during the second survey. COVID-19 testing data are not shown after May 8.
Suggested citation for this article: Sanchez GV, Biedron C, Fink LR, et al. Initial and Repeated Point Prevalence Surveys to Inform SARS-CoV-2 Infection Prevention in 26 Skilled Nursing Facilities — Detroit, Michigan, March–May 2020. MMWR Morb Mortal Wkly Rep 2020;69:882-886. DOI: http://dx.doi.org/10.15585/mmwr.mm6927e1.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.
