Cholera Outbreak in Dadaab Refugee Camp, Kenya — November 2015–June 2016
Weekly / August 31, 2018 / 67(34);958–961
Qabale Golicha1; Sharmila Shetty2; Orkhan Nasiblov3; Abubakar Hussein1; Eliud Wainaina1; Mark Obonyo1; Daniel Macharia2,4; Raymond N. Musyoka4,5; Hussein Abdille3; Maurice Ope4,5; Rachael Joseph4,5; Willy Kabugi6; John Kiogora6; Munawwar Said6; Waqo Boru1; Tura Galgalo2,4; Sara A. Lowther2,4; Bonventure Juma2,4; Robert Mugoh7; Newton Wamola7; Clayton Onyango2,4; Zeinab Gura1; Marc-Alain Widdowson2,4; Kevin M. DeCock4; John W. Burton3 (View author affiliations)
View suggested citationSummary
What is already known about this topic?
Cholera, caused by infection with the bacterium Vibrio cholerae through ingestion of contaminated food or water, can spread rapidly in densely populated settings such as refugee camps.
What is added by this report?
During November 18, 2015–June 6, 2016, the largest cholera outbreak (1,797 cases; attack rate 5.1 per 1,000) in the history of Dadaab refugee camp in Kenya occurred. Significant risk factors included living in a compound where open defecation, visible human and solid waste, and eating from a shared plate were common. Chlorine levels in water were below standard, and handwashing facilities were insufficient.
What are the implications for public health practice?
Improvements to water and sanitation, expansion of capacity for community outreach, and enhanced camp security and disease surveillance systems in Dadaab camp and the surrounding area are urgently needed.
Dadaab Refugee camp in Garissa County, Kenya, hosts nearly 340,000 refugees in five subcamps (Dagahaley, Hagadera, Ifo, Ifo2, and Kambioos) (1). On November 18 and 19, 2015, during an ongoing national cholera outbreak (2), two camp residents were evaluated for acute watery diarrhea (three or more stools in ≤24 hours); Vibrio cholerae serogroup O1 serotype Ogawa was isolated from stool specimens collected from both patients. Within 1 week of the report of index cases, an additional 45 cases of acute watery diarrhea were reported. The United Nations High Commissioner for Refugees and their health-sector partners coordinated the cholera response, community outreach and water, sanitation, and hygiene (WASH) activities; Médecins Sans Frontiéres and the International Rescue Committee were involved in management of cholera treatment centers; CDC performed laboratory confirmation of cases and undertook GIS mapping and postoutbreak response assessment; and the Garissa County Government and the Kenya Ministry of Health conducted a case-control study. To prevent future cholera outbreaks, improvements to WASH and enhanced disease surveillance systems in Dadaab camp and the surrounding area are needed.
Investigation and Findings
Case ascertainment. A suspected cholera case was defined as the occurrence of acute watery diarrhea in any person aged ≥2 years seen at a camp health facility on or after November 18, 2015, or in a child aged <2 years who was epidemiologically linked to a confirmed cholera case. Stool specimens were collected from one of every 2–3 patients with suspected cholera and tested using standard microbiological methods*; cholera isolates were tested for antimicrobial resistance by disc-diffusion. Suspected cases with a stool culture positive for V. cholerae were considered to be laboratory-confirmed (3). Demographic and clinical data were recorded for all suspected and confirmed cases. Characteristics of cholera cases were described, and case fatality rates calculated. Geographic information system software was used to map the calculated cumulative attack rates by age, sex, and residential block in each of the five subcamps. Spatial clustering of cholera cases by block, adjusted to the block’s population density, was evaluated using the software’s Average Nearest Neighbor function (4), which indicated clustering in some blocks.
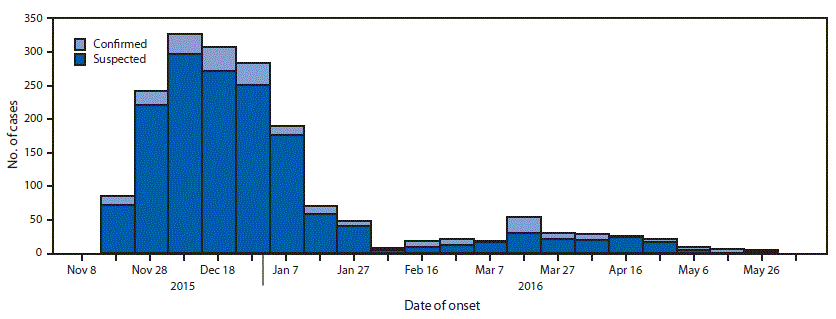
During November 18, 2015–June 6, 2016, a total of 1,797 cases of cholera, including 1,548 suspected and 249 confirmed, were reported among the camp’s 348,781 residents (Figure); 20 cases that occurred in persons from the host community and were treated in the camp health facilities are included. Males accounted for 904 (51%) cases. The overall attack rate was 5.1 per 1,000 residents, with the highest attack rate occurring in children aged 2–4 years (16.9); attack rates varied by subcamp (Table 1). Fourteen deaths were reported (case fatality rate = 0.79%).
After identification of the two index cases on November 18 and 19, the outbreak quickly spread in the subcamps of Hagadera (attack rate = 8.0 per 1,000; peak = December 18–27), Dagahaley (7.7; November 28–December 7), and Kambioos (5.3; December 28–January 6) (Table 1); among 252 residential blocks in these three subcamps, 195 (77%) reported at least one case. Fewer cases were reported in Ifo (attack rate = 0.6 per 1,000) and Ifo2 (1.7), where only 10%–30% of residential blocks reported at least one case. Incidence among affected residential blocks ranged from 0.1% to 20%; spatial clustering of cases occurred in all the subcamps within the residential blocks (p<0.01).
Identification of risk factors. In December 2015, the Kenya Ministry of Health conducted a case-control study in the subcamps most affected (Dagahaley and Hagadera) to identify risk factors for cholera. Persons with suspected or confirmed cholera (one per household) clinically evaluated before December 31, 2015, were eligible for inclusion. Eligible controls were Dagahaley or Hagadera residents aged ≥2 years with no history of acute watery diarrhea during the same period. Each case-patient was frequency matched to two controls by subcamp and age group (2–4 years, 5–14 years, 15–24 years, and ≥25 years). A standardized questionnaire was developed that adopted some questions from previous efforts, and it was administered to case-patients and controls (or their caregivers) to collect demographic and exposure information. Partially adjusted odds ratios (ORs) and 95% confidence intervals (CIs) were calculated. Unconditional logistic regression using stepwise forward selection was used for building a multivariate model. Independent variables with p–values ≤0.2 in univariate analysis were considered for inclusion. Adjusted ORs (aORs) and 95% CIs were calculated from the final multivariate model.
From a calculated sample size of 38 cases and 76 controls, 32 case-patients and 64 controls were enrolled in the case-control study (Table 2). Identified risk factors for suspected or confirmed cholera included observation by interviewer of 1) human fecal and solid waste in a compound, 2) soiled communal latrines or self-reported open defecation, 3) swimming in rainwater pools, 4) sharing of food from a common plate, and 5) reported sharing of a latrine with someone with diarrhea. Always washing hands with soap and water after using a latrine and household latrine ownership were protective. Living in a compound with visible human and solid waste (aOR = 7.7; 95% CI = 2.0–30.0), self-reported open defecation (13.0; 3.0–61.0), and sharing food on a common plate (5.9; 1.5–23.0) remained significant in the final multivariate model. No evidence of disease clustering by ethnic background or geographic origin was found.
Assessment of outbreak control measures. In late January 2016, as the outbreak waned, CDC and the United Nations High Commissioner for Refugees conducted site visits in four subcamps to assess outbreak control measures. Residual chlorine levels were below outbreak standards in various water sources, including tap stands (outbreak standard = 1.0 mg/L) and households (outbreak standard = 0.5 mg/L), and handwashing facilities in schools, markets, and eateries were insufficient. Pools of stagnant water where children played were observed near affected residential blocks in Dagahaley and Ifo2 subcamps. Although the average number of persons per latrine in Dadaab met the international standard for refugee camps (one latrine per 20 persons) (5,6), in some subcamps, up to 60 persons were observed to be sharing one latrine. In addition, at the outbreak onset in November 2015, only 168 community health workers were in the camp (approximately one per 2,000 residents), one quarter of the internationally recommended standard of one per 500 residents (5). Some households anecdotally reported cases of cholera in multiple household members, although this information was not systematically collected.
Assessment of antibiotic susceptibility of cholera isolates. V. cholerae serogroup O1, serotype Ogawa was isolated from 312 (39%) of 791 stool specimens. All isolates were sensitive to tetracycline, ceftriaxone, cotrimoxazole, gentamycin, and chloramphenicol; 97% were sensitive to ciprofloxacin. All isolates had intermediate sensitivity to erythromycin and were resistant to furazolidone and nalidixic acid.
Public Health Response
Cholera treatment centers were established by Médecins Sans Frontiéres and the International Rescue Committee, and active surveillance for cases of acute watery diarrhea was enhanced. A health promotion and hygiene campaign was conducted, primarily through mobilization of community health workers (from 168 during the first few weeks of the outbreak to 286) and hygiene promoters and use of media networks, especially radio. Frequent coordination meetings were held among stakeholders to provide updates and revise recommendations. Water from boreholes was chlorinated, soap was distributed, and bedding and latrine disinfection in affected households was carried out. WASH partners were advised to maintain adequate chlorination levels, install additional tap stands and latrines (especially in unofficial settlement areas), and install additional handwashing facilities in schools, eateries, and marketplaces.
Discussion
Cholera is an acute diarrheal illness caused by the toxin secreted during infection with V. cholerae bacterium after ingestion of contaminated food or water. The infection is frequently mild or asymptomatic; however, approximately 5%–10% of infected persons develop severe disease and profuse watery diarrhea. Without prompt treatment, persons with severe disease can die within hours (7). Cholera outbreaks can spread rapidly in densely populated settings such as refugee camps (8). Rapid detection and control of cholera outbreaks is a goal for the implementation of the World Health Organization’s International Health Regulations and for global health security.
This cholera outbreak is the largest reported in Dadaab refugee camp since its establishment in 1992. Existing laboratory capacity and effective disease surveillance in the camp facilitated early detection and eventual control of the outbreak. Antimicrobial susceptibility testing confirmed that V. cholerae circulating in the camp was susceptible to antibiotics used in treating patients with severe cholera (9).
All subcamps were affected, but the highest rates occurred in the two largest subcamps. These two areas had poorer water drainage and were in proximity to food markets where persons from areas with active cholera transmission interact. These factors might have contributed to the large number of cases and high attack rates in these areas. Furthermore, in these subcamps, large numbers of housing structures were outside the official boundaries of the camp, in areas that had poorer water and sanitation infrastructure. Inadequate residual chlorine levels in drinking water and the presence of standing bodies of water used for swimming and bathing suggest possible waterborne transmission routes. Reported infection of multiple household members and spatial case clustering among residential blocks suggest household transmission or common exposures to a contaminated food or water source.
Limited promotion of hygiene messaging during the early weeks of the outbreak could also have increased vulnerability to cholera transmission. Sustained and intensive hygiene promotion and WASH interventions in the most affected blocks were also recommended.
The findings in this report are subject to at least two limitations. First, camp insecurity affected case finding and study enrollment, precluding achieving the calculated sample size for the case-control study. Second, other cases of acute watery diarrhea might have been included as cases of suspected cholera, reducing the power of the study.
The last reported cholera case in the camp occurred on June 6, 2016, and the outbreak was declared over on June 21, 2016. Thereafter, two small clusters of cases not associated with traveling outside the camp or contact with visitors outside the camp were reported in July (seven cases) and August (five cases) 2016. During April–August 2017, a cholera outbreak involving 511 cases occurred after flooding that destroyed approximately 9,000 latrines. In September 2017, four cases were reported, and during October–December 2017, 109 cases were reported. Both outbreaks were controlled immediately with no deaths. Improvements to WASH and disease surveillance are needed to prevent future outbreaks.
Acknowledgments
Garissa County, Dadaab County, Fafi Sub-county health teams; Dadaab field offices for the United Nations High Commissioner for Refugees; International Rescue Committee; Médecins Sans Frontiéres; Islamic Relief Organization; the Refugee Community in Dadaab.
Corresponding author: Qabale Golicha, qgolicha@gmail.com, +254 716624326.
1Kenya Field Epidemiology and Laboratory Training Program, Ministry of Health, Nairobi, Kenya; 2Division of Global Health Protection, Center for Global Health, CDC; 3United Nations High Commissioner for Refugees, Nairobi, Kenya; 4CDC-Kenya, Nairobi; 5Division of Global Migration and Quarantine, National Center for Emerging and Zoonotic Infectious Diseases, CDC; 6International Rescue Committee, Nairobi, Kenya; 7Kenya Medical Research Institute, Nairobi, Kenya.
All authors have completed and submitted the ICMJE form for disclosure of potential conflicts of interest. No potential conflicts of interest were disclosed.
* Rapid diagnostic tests (RDT) for cholera were also used for cholera diagnosis in the camp; however, cholera RDT testing and results were not collected systematically, and are not presented here.
References
- United Nations High Commissioner for Refugees. Camp population statistics by country of origin, sex and age group. New York, NY: United Nations High Commissioner for Refugees; 2018. https://data2.unhcr.org/en/situations/horn/location/178
- George G, Rotich J, Kigen H, et al. Notes from the field: ongoing cholera outbreak—Kenya, 2014–2016. MMWR Morb Mortal Wkly Rep 2016;65:68–9. CrossRef PubMed
- Ministry of Public Health and Sanitation. Technical guidelines of integrated disease surveillance and response in Kenya. 2nd ed. Nairobi, Kenya: Ministry of Public Health and Sanitation; 2012.
- Mitchel A. The ESRI guide to GIS analysis, vol. 2: spatial measurements and statistics. Redlands, CA: ESRI Press; 2005.
- The Sphere Project. Humanitarian charter and minimum standards in humanitarian response. 3rd ed. Rugby, United Kingdom: Practical Action Publishing; 2011.
- United Nations High Commissioner for Refugees. A guidance for UNHCR field operations on water and sanitation services. New York, NY: United Nations High Commissioner for Refugees; 2008. https://reliefweb.int/report/world/guidance-unhcr-field-operations-water-and-sanitation-services
- CDC. Cholera–Vibrio cholerae infection. Atlanta, GA: US Department of Health and Human Services, CDC; 2014. https://www.cdc.gov/cholera/general/index.html
- United Nations Children’s Fund. Cholera toolkit 2013. New York, NY: United Nations Children’s Fund; 2016. https://www.unicef.org/cholera/Cholera-Toolkit-2013.pdf
- Nelson EJ, Nelson DS, Salam MA, Sack DA. Antibiotics for both moderate and severe cholera. N Engl J Med 2011;364:5–7. CrossRef PubMed
 FIGURE. Suspected and confirmed cholera cases (N = 1,797), by week of illness onset — Dadaab refugee camp, Kenya, November 18, 2015–June 6, 2016
FIGURE. Suspected and confirmed cholera cases (N = 1,797), by week of illness onset — Dadaab refugee camp, Kenya, November 18, 2015–June 6, 2016
Abbreviation: CFR = case fatality rate.
* Twenty cholera cases that occurred in the host community are not included.
† Estimated based on the assumption that among 54,804 children aged 0–4 years, 60% were aged 2–4 years.
Abbreviations: CI = confidence interval; OR = odds ratio.
* All participants used communal piped water from tap stands; levels of free residual chlorine and fecal coliforms unknown.
† Unconditional large sample logistic regression. All partially adjusted models included age and residence. Final ORs are adjusted for age, residence, visible solid and human waste in compound, practicing open defecation, and sharing food from a common plate.
§ Adjusted OR not calculated because p–value >0.05 for partially adjusted bivariate association.
Suggested citation for this article: Golicha Q, Shetty S, Nasiblov O, et al. Cholera Outbreak in Dadaab Refugee Camp, Kenya — November 2015–June 2016. MMWR Morb Mortal Wkly Rep 2018;67:958–961. DOI: http://dx.doi.org/10.15585/mmwr.mm6734a4.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.