Protracted Outbreak of Salmonella Newport Infections Linked to Ground Beef: Possible Role of Dairy Cows — 21 States, 2016–2017
Weekly / April 20, 2018 / 67(15);443–446
Katherine E. Heiman Marshall, MPH1; Mackenzie Tewell, MPH2; Selam Tecle, MPH3; Molly Leeper, MPH1; Jennifer Sinatra, DVM4; Bonnie Kissler, MPH4; Adrienne Fung, MPH5; Kerri Brown, MSPH6; Darlene Wagner, PhD1; Eija Trees, PhD1; Kelley B. Hise, MPH1; Vishnu Chaturvedi, PhD7; Linda K. Schlater, DVM8; Brenda R. Morningstar-Shaw, MS8; Laura Whitlock, MPH1; Kristin Holt, DVM4; Karen Becker, DVM4; Megin Nichols, DVM1; Ian T. Williams, PhD1; Michael Jhung, MD1; Matthew E. Wise, PhD1; Laura Gieraltowski, PhD1 (View author affiliations)
View suggested citationSummary
What is already known about this topic?
Previous outbreaks of salmonellosis were linked to contaminated ground beef produced from slaughtered dairy cows.
What is added by this report?
Contaminated ground beef was the likely source of a protracted outbreak of 106 Salmonella Newport infections, 42 hospitalizations, and one death in 21 states during October 2016–July 2017. While no direct link was found, whole genome sequencing suggests dairy cows were the ultimate outbreak source.
What are the implications for public health practice?
Foodborne outbreak investigations could be enhanced by improvements in the traceability of cows from their originating farms or sale barns, through slaughter and processing establishments, to ground beef sold to consumers.
Altmetric:
In January 2017, CDC identified a cluster of Salmonella enterica serotype Newport infections with isolates sharing an indistinguishable pulsed-field gel electrophoresis (PFGE) pattern, JJPX01.0010 (pattern 10), through PulseNet, the national molecular subtyping network for foodborne disease surveillance. This report summarizes the investigation by CDC, state and local health and agriculture departments, and the U.S. Department of Agriculture’s Food Safety and Inspection Service (USDA-FSIS) and discusses the possible role of dairy cows as a reservoir for strains of Salmonella that persistently cause human illness. This investigation combined epidemiologic and whole genome sequencing (WGS) data to link the outbreak to contaminated ground beef; dairy cows were hypothesized to be the ultimate source of Salmonella contamination.
Epidemiologic Investigation
A case was defined as infection with Salmonella Newport with PFGE pattern 10 closely related to the outbreak strain by WGS, with bacterial isolation during October 1, 2016, through July 31, 2017. A total of 106 cases were identified in 21 states (Figure 1). Most illnesses ([72%]) were reported from southwestern states, including Arizona (30), California (25), New Mexico (14), and Texas (seven). Illness onset dates ranged from October 4, 2016, through July 19, 2017 (Figure 2). Patients ranged in age from <1–88 years (median = 44 years), and 53 (50%) were female. Among 88 (83%) patients with known outcomes, 42 (48%) were hospitalized, and one died.
Initial interviews identified consumption of ground beef as a common exposure among patients. A focused questionnaire was developed to collect detailed information on ground beef exposure and to obtain shopper card information and receipts. Among 65 interviewed patients, 52 (80%) reported eating ground beef at home in the week before illness began. This percentage was significantly higher than the 2006–2007 FoodNet Population Survey, in which 40% of healthy persons reported eating ground beef at home in the week before they were interviewed (p<0.001) (1). Among the 52 patients who ate ground beef at home, 31 (60%) reported that they bought it or maybe bought it from multiple locations of two national grocery chains, and 21 (40%) reported that they bought ground beef from locations of 15 other grocery chains. Specific ground beef information was available for 35 patients. Among these, 15 (43%) purchased ground beef as chubs (rolls) of varying sizes (range = 2–10 lbs), 18 purchased it on a tray wrapped in plastic, and two purchased preformed hamburger patties. Twenty-nine patients reported that they bought fresh ground beef, four bought frozen ground beef, and four did not recall whether it was fresh or frozen when purchased. When asked about ground beef preparation, 12 (36%) of 33 patients reported that they definitely or possibly undercooked it.
Traceback Investigation
USDA-FSIS conducted traceback on ground beef purchased within 3 months of illness onset for 11 patients who provided shopper card records or receipts. Approximately 20 ground beef suppliers belonging to at least 10 corporations were identified; 10 of the 11 records traced back to five company A slaughter/processing establishments, seven of 11 traced back to five company B slaughter/processing establishments, and four of 11 traced back to two company C slaughter/processing establishments.
Product and Animal Testing
Opened, leftover samples of ground beef from three patients’ homes were collected for testing. All were purchased from one of two national grocery chains that had been identified by a majority of patients. One sample, collected from ground beef removed from its original packaging, yielded the outbreak strain. The other two samples did not yield Salmonella.
The outbreak strain was also isolated from four New Mexico dairy cattle (Table). One was collected from a spontaneously aborted fetus in July 2016, and one was isolated from feces from a young calf in November 2016. The third isolate was identified by searching the USDA Animal and Plant Health Inspection Service National Veterinary Services Laboratory (USDA-APHIS NVSL) database for Salmonella Newport isolates collected from cattle in Arizona, California, Texas, New Mexico, and Wisconsin during January 2016–March 2017. Eighteen Salmonella Newport isolates were identified, including 13 from Texas, three from New Mexico, and two from Wisconsin. The only Salmonella Newport pattern 10 isolate identified was from a fecal sample from a New Mexico dairy cow collected during November 2016. The fourth isolate was from a USDA-FSIS routine cattle fecal sample collected at a Texas slaughter establishment in December 2016; USDA-FSIS determined the sample was from a dairy cow and identified the New Mexico farm of origin. Because of confidentiality practices, officials were not able to identify the farm or farms of origin for the dairy cows associated with the other three samples or whether the four dairy cows were associated with a single farm. None of the 11 patients with information for traceback ate ground beef produced at the Texas slaughter establishment.
Laboratory Investigation
Whole genome high-quality single nucleotide polymorphism (SNP) analysis* showed that 106 clinical isolates were closely related to each other genetically, to the four dairy cattle isolates, and to the leftover ground beef isolate (range = 0–12 SNP differences), suggesting that the Salmonella bacteria found in patients, ground beef, and dairy cattle all shared a common source. Thirty-nine additional clinical isolates with PFGE pattern 10 were determined to not be closely related and were excluded from the outbreak. No antibiotic resistance was detected among three clinical isolates tested by CDC’s National Antimicrobial Resistance Monitoring Laboratory.†
Public Health Response
Because the USDA-FSIS traceback investigation did not converge on a common production lot of ground beef or a single slaughter/processing establishment, and no ground beef in the original packaging yielded the outbreak strain, a recall of specific product was not requested. A public warning was not issued to consumers because specific, actionable information was not available (e.g., a specific brand or type of ground beef). Officials in New Mexico visited the dairy farm that was the source of the cow at the Texas establishment and noted no concerns about conditions or practices. However, this visit occurred late in the investigation, and conditions at the time of the visit might not have represented those present immediately before and during the outbreak. No samples from the environment or cows were collected during this visit.
Discussion
Epidemiologic and laboratory evidence indicated that contaminated ground beef was the likely source of this protracted outbreak of Salmonella Newport infections. A significantly higher percentage of patients than expected ate ground beef at home, and a patient’s leftover ground beef yielded the outbreak strain. Dairy cows colonized or infected with the outbreak strain before slaughter are hypothesized to be the ultimate outbreak source. Most U.S. ground beef is produced from beef cattle; however, 18% is produced from dairy cows (2). Dairy cows are sold for beef production through sale barns or directly to slaughter establishments as they age or if their milk production is insufficient (2). Previous studies have demonstrated long-term persistence of Salmonella Newport in dairy herds (3,4), and a 1987 Salmonella Newport outbreak was linked to contaminated ground beef from slaughtered dairy cows (5). In the current outbreak, as has been observed in previous outbreaks, ground beef purchases traced back to numerous lots and slaughter/processing establishments (6). One possible explanation is that dairy cows carrying a high Salmonella load that overwhelmed antimicrobial interventions could have gone to multiple slaughter/processing establishments (7), resulting in contamination of multiple brands and lots of ground beef. This might explain the reason for failure to identify a single, specific source of contaminated ground beef.
This investigation identified the outbreak strain only in samples from dairy cattle from New Mexico. All four isolates from dairy cattle samples were closely related genetically by WGS to isolates from patients, providing further evidence of a connection between dairy cattle in New Mexico and the outbreak. The disproportionate geographic distribution of cases in the U.S. Southwest, including New Mexico, also suggests a possible regional outbreak source. Although limited in scope, the query of the USDA-APHIS NVSL data identified the outbreak strain only from one New Mexico dairy cow (isolate 3), and the sample collection date was consistent with the timing of illnesses in this outbreak. The overall prevalence and geographic distribution of the outbreak strain in cattle is not known, and it is possible that cattle in states other than New Mexico might have been infected or colonized with the outbreak strain.
This was a complex and challenging investigation for several reasons. First, the PFGE pattern in the outbreak was not uncommon in PulseNet, making it difficult to distinguish outbreak cases from sporadic illnesses associated with the same Salmonella Newport pattern. WGS analysis provided more discriminatory power to refine the outbreak case definition and excluded 39 cases of illness from the outbreak. However, sequencing is not currently performed in real time for Salmonella, thereby slowing the process of determining which cases were likely outbreak-associated. In addition, a direct pathway linking outbreak cases to dairy cows infected with the outbreak strain of Salmonella Newport could not be established. This is because product traceback did not converge on a single contaminated lot of ground beef, and investigators were unable to ascertain a link between the beef slaughter/processing establishments identified during traceback and the farms with dairy cows that yielded the outbreak strain. Tracing back ground beef purchased by patients to slaughter/processing establishments requires documentation such as receipts or shopper card records, and only 10% of patients had this information available. For this outbreak, tracing back cows at slaughter/processing establishments to the farm from which they originated was problematic because cows were not systematically tracked from farm to slaughter/processing establishments.
Four points along the “farm to fork” continuum provide opportunities to prevent consumers from becoming ill from contaminated ground beef. First, farms can implement good management practices for cattle health, including vaccination, biosecurity (e.g., controlling movement of persons and animals on farms, keeping a closed herd [so that no animals on the farm are purchased, loaned to other farms, or have contact with other animals], planning introduction of new animals and quarantining them, and performing microbiologic testing of animals), and cleaning and disinfection measures to decrease Salmonella burden in animals and the environments in which they reside, reducing the likelihood that Salmonella will enter beef slaughter/processing establishments (8). Second, slaughter/processing establishments are required to maintain Hazard Analysis and Critical Control Points systems to reduce Salmonella contamination as well as slaughter and sanitary dressing procedures to prevent carcass contamination (9). Third, although Salmonella is not considered an adulterant in not-ready-to eat (NRTE) meat products, USDA-FSIS likely will consider the product to be adulterated when NRTE meat products are associated with an outbreak (9). Finally, consumers are advised to cook ground beef to 160°F (71°C) as measured by a food thermometer to destroy any bacteria that might be present. Consumers are also advised to wash hands, utensils, and surfaces often; separate and not cross-contaminate foods; and refrigerate foods promptly and properly.
This investigation emphasizes the utility of WGS during outbreak investigations and identifies the need for improvements in traceability from the consumer to the farm. It also highlights the importance of continued evaluation of farm practices to help reduce persistent Salmonella contamination on farms, contamination of ground beef, and ultimately human illness.
Acknowledgments
Kis Robertson Hale, Food Safety and Inspection Service, U.S. Department of Agriculture; territorial, state, city, and county health departments and laboratories; Danya Alvarez, John Crandall, Hillary Berman-Watson, California Department of Public Health Microbial Diseases Laboratory.
Conflict of Interest
No conflicts of interest were reported.
Corresponding author: Katherine E. Heiman Marshall, kmarshall2@cdc.gov, 404-639-4405.
1Division of Foodborne, Waterborne, and Environmental Diseases, National Center for Emerging and Zoonotic Infectious Diseases, CDC; 2Arizona Department of Health Services; 3California Department of Public Health; 4Food Safety and Inspection Service, U.S. Department of Agriculture; 5Texas Department of State Health Services; 6Colorado Department of Public Health; 7California Department of Public Health Microbial Diseases Laboratory; 8National Veterinary Services Laboratory, Animal and Plant Health Inspection Service, U.S. Department of Agriculture.
References
- CDC. Foodborne Diseases Active Surveillance Network (FoodNet) population survey atlas of exposure, 2006–2007. Atlanta, GA: US Department of Health and Human Services, CDC; 2008. https://www.cdc.gov/foodnet/surveys/foodnetexposureatlas0607_508.pdf
- US Department of Agriculture. Economic opportunities for dairy cow culling management options, 1996. Fort Collins, CO: US Department of Agriculture, Animal and Plant Health Inspection Service; 1996. http://www.aphis.usda.gov/animal_health/nahms/dairy/downloads/dairy96/Dairy96_is_BiosecurityPrac.pdf
- Cobbold RN, Rice DH, Davis MA, Besser TE, Hancock DD. Long-term persistence of multi–drug-resistant Salmonella enterica serovar Newport in two dairy herds. J Am Vet Med Assoc 2006;228:585–91. CrossRef PubMed
- Clegg FG, Chiejina SN, Duncan AL, Kay RN, Wray C. Outbreaks of Salmonella Newport infection in dairy herds and their relationship to management and contamination of the environment. Vet Rec 1983;112:580–4. CrossRef PubMed
- Spika JS, Waterman SH, Hoo GWS, et al. Chloramphenicol-resistant Salmonella Newport traced through hamburger to dairy farms. N Engl J Med 1987;316:565–70. CrossRef PubMed
- CDC. Outbreak of Salmonella Heidelberg infections linked to a single poultry producer—13 states, 2012–2013. MMWR Morb Mortal Wkly Rep 2013;62:553–6. PubMed
- Brichta-Harhay DM, Guerini MN, Arthur TM, et al. Salmonella and Escherichia coli O157:H7 contamination on hides and carcasses of cull cattle presented for slaughter in the United States: an evaluation of prevalence and bacterial loads by immunomagnetic separation and direct plating methods. Appl Environ Microbiol 2008;74:6289–97. CrossRef PubMed
- Stuttgen S, Baumann L, Konkle D, Mauer C, Skjolaas C, Patton E. Factsheet: Salmonella biosecurity: protecting yourself and your cattle. Medford, WI: University of Wisconsin-Extension; 2016. https://datcp.wi.gov/Documents/BiosecuritySalmonellaCattle.pdf
- US Department of Agriculture. HACCP plan reassessment for not-ready-to-eat comminuted poultry products and related agency verification procedures. Fed Regist 2012 Dec 6;77(235):72686–91. https://www.federalregister.gov/documents/2012/12/06/2012-29510/haccp-plan-reassessment-for-not-ready-to-eat-comminuted-poultry-products-and-related-agency
 FIGURE 1. Infections with the outbreak strain of Salmonella Newport (n = 106), by state of residence — 21 states, October 2016–July 2017
FIGURE 1. Infections with the outbreak strain of Salmonella Newport (n = 106), by state of residence — 21 states, October 2016–July 2017
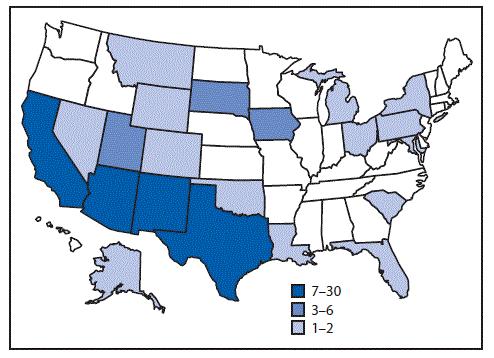
 FIGURE 2. Isolates of the outbreak strain of Salmonella Newport from patients (n = 106), dairy cattle* (n = 3), and leftover ground beef (n = 1) — 21 states, October 2016–July 2017
FIGURE 2. Isolates of the outbreak strain of Salmonella Newport from patients (n = 106), dairy cattle* (n = 3), and leftover ground beef (n = 1) — 21 states, October 2016–July 2017
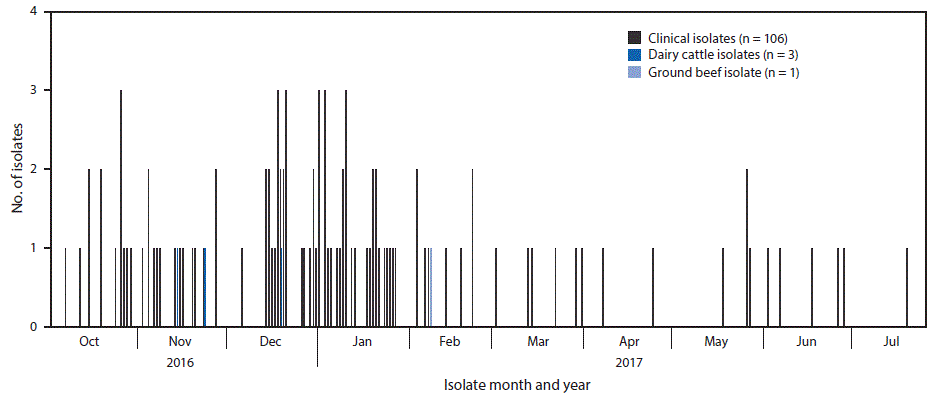
*The isolate collected from a dairy cow fetus in July 2016 is not displayed because cases were reported during July–October 2016 but were not investigated as part of this outbreak.
Abbreviation: NVSL = USDA Animal and Plant Health Inspection Service National Veterinary Services Laboratory.
*Because of the anonymity of samples from cows routinely tested by NVSL, it is possible that this isolate is from the same sample as isolate 2.
Suggested citation for this article: Marshall KE, Tewell M, Tecle S, et al. Protracted Outbreak of Salmonella Newport Infections Linked to Ground Beef: Possible Role of Dairy Cows — 21 States, 2016–2017. MMWR Morb Mortal Wkly Rep 2018;67:443–446. DOI: http://dx.doi.org/10.15585/mmwr.mm6715a2.
MMWR and Morbidity and Mortality Weekly Report are service marks of the U.S. Department of Health and Human Services.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All HTML versions of MMWR articles are generated from final proofs through an automated process. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (https://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.