Kansas Uses Syndromic Data to Improve Case Reporting for EVALI
Injury, Collaboration

The U.S. outbreak of e-cigarette, or vaping, product use-associated lung injury (EVALI) received major media attention when CDC deployed epidemiologists to investigate a cluster of teenagers and young adults in Wisconsin and Illinois presenting with unexplained, sudden onset lung injuries in August 2019. As a regional investigation unfolded, states searched for potential cases in their emergency department data.
Epidemiologists at the Kansas Department of Health and Environment (KDHE) received a request from their counterparts in Wisconsin asking that they conduct syndromic surveillance to look for potential cases of lung injury. After identifying the first case in Kansas, KDHE issued a statewide health alert to clinicians summarizing the multistate outbreak. Case reports started to come in, but KDHE epidemiologists questioned whether some cases were being missed. They began comparing clinician-reported cases with syndromic surveillance results and refined syndromic queries to target the EVALI-risk population.
The use of syndromic data to identify potential cases and subsequent comparison with clinician-reported cases helped in two ways. First, by identifying potential cases, the emerging picture of EVALI was more accurate. Second, by asking facility clinicians and infection prevention staff for case records and explaining why the records were needed, Kansas raised facility awareness of the condition and improved case reporting for EVALI. Funding from the National Syndromic Surveillance Program supports the use of syndromic surveillance in improving the nation’s public health.
Public Health Problem
The U.S. outbreak of e-cigarette, or vaping, product use-associated lung injury (EVALI) has received major media attention ever since the Centers for Disease Control and Prevention (CDC) deployed a team of epidemiologists to investigate a cluster of teenagers and young adults in Wisconsin and Illinois presenting with unexplained, sudden onset lung injuries in August 2019. Given the popularity of e-cigarette, or vaping, product use among youth, combined with the severity of this lung injury, public health officials pushed to identify and prevent additional cases quickly. As the regional investigation unfolded, other states searched for potential cases in their emergency department data. With the need for timely data to understand what was happening in states, ESSENCE1 and syndromic inquiries took center stage.
On August 1, 2019, epidemiologists at the Kansas Department of Health and Environment (KDHE) received a request from their counterparts in Wisconsin asking that they query ESSENCE for potential cases of lung injury. After the first case in Kansas was identified on August 30, 2019, KDHE issued a statewide health alert to clinicians summarizing the multistate outbreak. This alert used initial syndromic surveillance numbers for respiratory syndrome with mention of e-cigarette, or vaping, product use to highlight the possibility of the outbreak reaching Kansas. After issuing a health alert, reports of cases started to come in, but KDHE epidemiologists questioned whether some cases were being missed.
After the second EVALI-reported death in Kansas, KHDE Secretary Dr. Lee Norman testified to Congress about what he recognized as “a public health emergency of considerable importance.” As heightened surveillance became more critical, there was an increased need to identify cases, investigate potential cases, and encourage clinician reporting.
Actions Taken
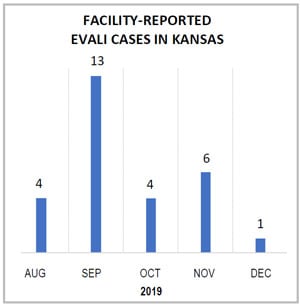
Shown here are EVALI cases reported to the Kansas Epidemiological Hotline via the electronic disease surveillance system, EpiTrax. Kansas epidemiologists noticed a sharp increase in provider reporting after the initial health alert was issued in August. Reported cases declined in October before increasing slightly in November after efforts were made to actively identify unreported cases.
During the last week of September 2019, the Kansas Syndromic Surveillance Program2 began using syndromic data to identify potential cases of EVALI. KDHE epidemiologists compared clinician-reported cases with ESSENCE query results, revealing that both queries—one based on vaping-related terms and one on severe lung injury—were insufficient for case finding. Consequently, they refined these syndromic queries to improve performance, balancing sensitivity with specificity. Sometimes, though, diagnoses were masked in emergency department visits by problems associated with co-occurring drug or alcohol use. Initially, KDHE epidemiologists excluded terms for drug use to decrease noise. But with the limited size of the Kansas population, epidemiologists later opted to include the terms, thereby increasing query sensitivity. They also refined the queries to exclude very young children and the elderly, making the queries more specific to the EVALI-risk population, limiting the follow-up workload, and improving timeliness.
After an interdepartmental discussion, KDHE initiated a two-step process to help epidemiologists identify potential cases:
- After pulling line-level data, use the following criteria to narrow the lists:
- Eliminate visits for patients with non-informative chief complaints or another defined cause of respiratory distress (heart failure, pulmonary embolism, etc.).
- Eliminate visits if patients are older than 25 years and report no related exposure, such as nicotine dependence, or use of THC-containing products.
- Eliminate visits for patients 25 years and younger only if an alternative cause exists for pneumonia or respiratory distress.
- Based on the refined lists, request and examine health records to identify any that meet CDC’s case definition for EVALI before contacting patients to complete the investigation.
1ESSENCE stands for Electronic Surveillance System for the Early Notification of Community-based Epidemics. This disease surveillance system was developed by the Johns Hopkins University Applied Physics Laboratory.
2The Kansas Syndromic Surveillance Program is partially supported by CDC’s Epidemiology and Laboratory Capacity Cooperative Agreement.
Outcome
In October 2019, nine records were identified using the method described above. After further review of the patients’ full medical records, three patients met the EVALI case definition, which prompted a full investigation. The use of syndromic surveillance to identify potential cases and subsequent comparison with records from the caregiving facility helped in two ways. First, by identifying potential cases, the emerging picture of EVALI was more accurate. Second, by asking facility clinicians and infection prevention staff for case records and explaining why the records were needed, KDHE helped raise facility awareness of the condition and improve provider reporting.
Discussion
Periodic syndromic surveillance within the public health investigation provided important information to clinicians at the forefront of EVALI response efforts. Further investigation of any increase or decrease in provider reporting can ensure continued situational awareness during an extended outbreak. Kansas epidemiologists for the tobacco-use prevention program will continue using the established query to monitor for additional cases of EVALI. Should the need arise, Kansas is now prepared to conduct timely surveillance and active case finding for future public health problems using similar strategies.
Lessons Learned
- Share data with health providers. Share surveillance findings to give healthcare providers context about what is happening in their communities. This can encourage provider vigilance regarding a new condition. Emphasize the importance of accurate and timely case reporting.
- Meet regularly with your surveillance team to assess how well queries are detecting potential cases. Compare syndromic surveillance data with traditional surveillance results for reportable conditions.
- Leverage ESSENCE’s inherent flexibility. If the national queries and vaping-related terms are not detecting all cases, refine the query so that it is more specific to your public health jurisdiction and needs. A query for active case finding will look different than one intended to monitor trends and will vary from jurisdiction to jurisdiction.
- Continue to run surveillance periodically. Syndromic surveillance is useful for monitoring the effectiveness of communications and health advisories. By running surveillance at regular intervals, epidemiologists are more apt to detect whether a decrease in cases is genuine.
Contacts
Vital and Health Statistics Data Analysis
Gabriel Ann Haas, Epidemiologist,
Vital Bureau of Epidemiology and Public Health Informatics
KDHE.Syndromic@ks.gov
Office of Public Health Data, Surveillance, and Technology
Division of Health Informatics and Surveillance
www.cdc.gov/nssp
This success story shows how NSSP
- Improves Data Representativeness
- Improves Data Quality, Timeliness, and Use
- Strengthens Syndromic Surveillance Practice
- Informs Public Health Action or Response
The findings and outcomes described in this syndromic success story are those of the authors and do not necessarily represent the official position of the National Syndromic Surveillance Program or the Centers for Disease Control and Prevention.
