Purpose
NSSP’s BioSense Platform is the core component of an integrated, nationwide system for public health syndromic surveillance. Onboarding is a collaborate process that involves setting up a live data feed. This guide describes each phase in the process.

The process
This content is part of a series of online job aids related to onboarding. These resources are meant to be used together along with New Facility Onboarding —The Complete Guide.
Download this job aid: New Facility Onboarding Overview
Onboarding is a collaborative process. NSSP and site teams will work with a facility, health department, electronic health records vendor, or health information exchange to transmit syndromic data from medical record systems to the NSSP BioSense Platform, comply with messaging guidance, and begin a live data feed.
NSSP's BioSense Platform is the core component of this integrated, nationwide system for public health syndromic surveillance. The BioSense Platform is a secure, cloud-based computing environment that enables public health agencies to conduct syndromic surveillance and analyze and share syndromic data on a common platform. Everyone has access to the same analytic and surveillance tools, follows the same standards for data exchange, and uses the same process to transmit syndromic data.
This guide describes each phase in the process. At the conclusion of onboarding, site and facility staff will have worked with the NSSP onboarding team to successfully transmit compliant data.
New facility onboarding overview
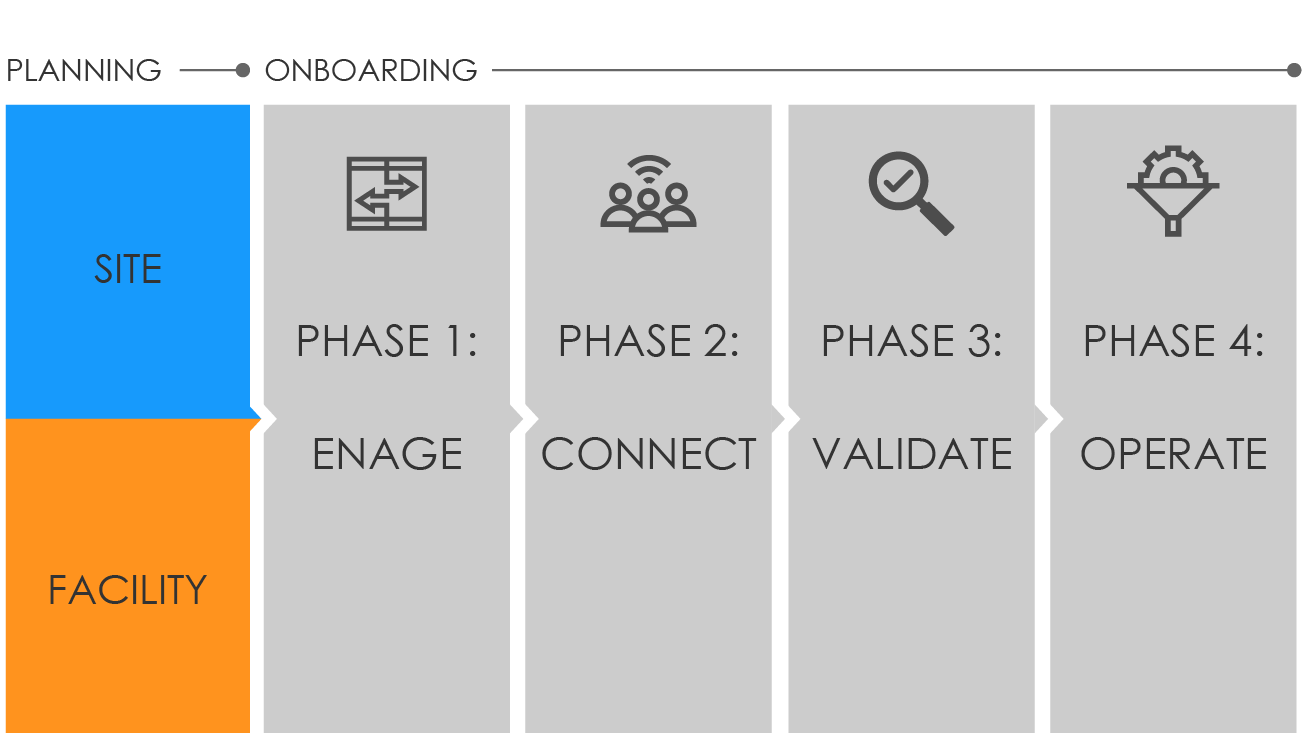
NSSP uses a four-phase approach to bring facilities onboard the BioSense Platform:
Phase 1: Engage begins when a site administrator creates an NSSP Service Desk ticket and requests onboarding for a facility. A site administrator can also initiate the process by adding a new facility to the Master Facility Table (MFT) and setting the facility status to Onboarding. During Engage, the site will:
- Engage with facility and NSSP for onboarding
- Prepare the facility for onboarding by adding the facility details to the MFT
- Prepare HL7®1 messages to be sent to the onboarding environment for testing and validation
- Prepare facility for connectivity
Phase 2: Connect begins once a new facility starts the process of connecting to a feed. As part of the Connect phase, the site will either connect the facility to an existing feed or request setup for a new feed via the NSSP Service Desk. In this scenario, the site will:
- Choose between SFTP and PHINMS secure data exchange technologies (SFTP is preferred)
- Successfully connect with BioSense Platform servers
- Upload a "hello world" test message
- Submit live facility data to the onboarding feed
Phase 3: Validate measures whether the received messages comply with PHIN Messaging Guide for Syndromic Surveillance and BioSense Platform requirements. During the Validate phase, the site will:
- Ensure timeliness of message delivery is consistently maintained, preferably with messages received within 24 to 48 hours of the visit
- Identify and resolve data issues to achieve requirements for validity, completeness
- Strive to improve data quality in all areas beyond minimum requirements
- Update facility status in MFT to Active and receive approval from NSSP
- Submit live facility data to the production feed
Phase 4: Operate begins once a feed or facility has been approved to send live data into production. As part of the Operate phase, sites will:
- Maintain data feeds
- As needed, collaborate to improve data quality
- Monitor data feeds for timeliness and consistency
- Monitor feed, facility connections, and data quality by using NSSP tools such as the Data Quality Dashboard, Data Quality on Demand programs, ESSENCE, Posit Workbench/RStudio, and SAS Studio
Resources
Key BioSense Platform tools and resources are listed below.
- HL7® and FHIR® are the registered trademarks of Health Level Seven International, and their use of these trademarks does not constitute an endorsement by HL7.
