Frequently Asked Questions and Answers About NoroSTAT
Q: What is NoroSTAT?
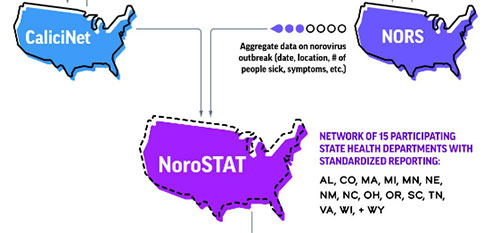
A: NoroSTAT is a collaborative network of fifteen state health departments and CDC, created to improve timeliness and completeness of norovirus outbreak reporting. The NoroSTAT network establishes and maintains standardized reporting practices that allow CDC to quickly link epidemiologic and laboratory data submitted to CDC’s National Outbreak Reporting System (NORS) and CaliciNet by participating state health departments.
Q: Why is NoroSTAT important?
A: NoroSTAT focuses on norovirus, the leading cause of acute gastroenteritis and foodborne illness in the United States.
NoroSTAT improves the timeliness, completeness, and consistency of norovirus outbreak reporting to CDC’s National Outbreak Reporting System (NORS) and CaliciNet. This allows CDC and state health departments to quickly evaluate current outbreak activity, make comparisons to previous years, and assess strain-specific norovirus outbreak characteristics, including the impact of new strains on outbreak frequency and severity.
NoroSTAT also enhances communication among epidemiologists and laboratorians in state health departments and CDC, enabling timely exchange of information regarding norovirus outbreak surveillance.
Q: How are data reported through NoroSTAT?
A: State health departments that participate in NoroSTAT:
- Report preliminary epidemiologic data to CDC through NORS within seven business days of receiving notification of suspected or confirmed norovirus outbreaks.
- Report laboratory data to CDC through CaliciNet within seven business days of receiving specimens.
- Provide data elements, such as the outbreak setting and transmission mode.
- Use consistent identification codes for each outbreak in their reports to help CDC and others link and integrate data from NORS and CaliciNet.
This graphic shows the process for reporting norovirus outbreak through NoroSTAT.
Reporting through NoroSTATQ: What types of data are reported?
A: State health departments report the following preliminary information:
- Epidemiologic data, including the outbreak onset date, primary transmission mode, total number of people sick, outbreak setting, and whether norovirus is the suspected or confirmed cause. They report these data to NORS within seven business days of receiving notification of suspected or confirmed norovirus outbreaks.
- Genetic sequences of norovirus strains. They report this to CaliciNet within seven business days of receiving the specimens.
After their initial report, state health departments often submit additional epidemiologic data, such as patient demographics, symptoms, and clinical outcomes, , through NORS as these data become available.
For more information, see NoroSTAT data.
Q: How can the data be used?
A: NoroSTAT provides information that can be used to quickly evaluate:
- Current norovirus outbreak activity.
- Intensity of current outbreak activity compared to prior years.
- Outbreak characteristics of new norovirus strains, including the impact of the new strains on outbreak frequency and severity.
Q: How is NoroSTAT supported?
A: CDC’s Division of Viral Diseases supports NoroSTAT through the Epidemiology and Laboratory Capacity (ELC) Cooperative Agreement. This cooperative agreement aims to enhance the capacity of state, local, and territorial health departments to effectively detect, respond, prevent, and control known and emerging (or re-emerging) infectious diseases.
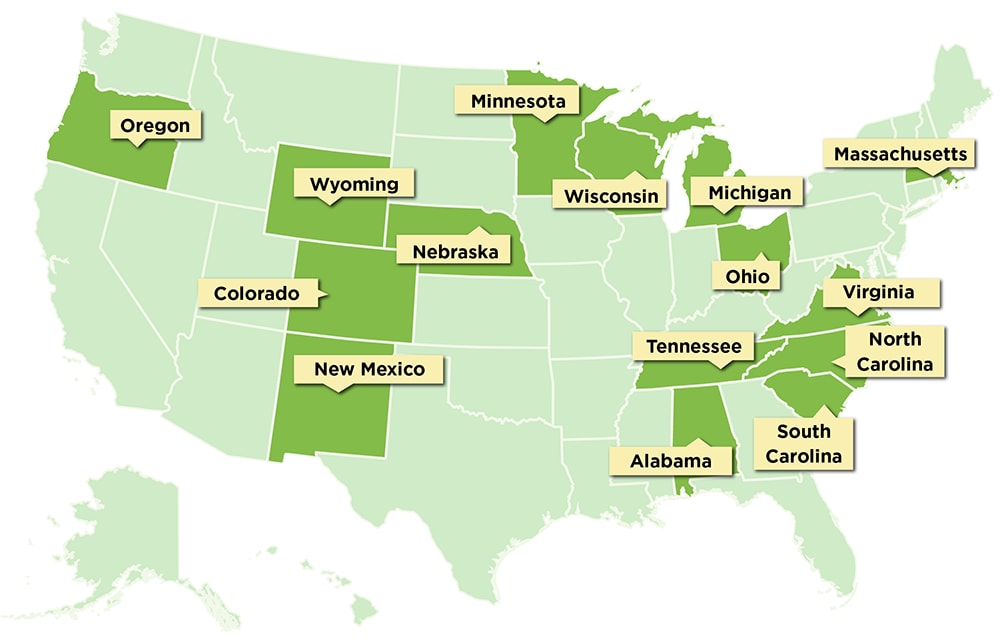
Q: Which state health departments currently participate in NoroSTAT?
A: The map below shows the fifteen states that participate in NoroSTAT. The health departments in these states demonstrate consistent surveillance practices and high norovirus outbreak reporting rates through the National Outbreak Reporting System (NORS) and CaliciNet
Q: Who can access NORS and CaliciNet data?
A: A limited set of data on all outbreaks submitted to CDC through NORS, including those caused by norovirus, can be viewed and downloaded using the NORS Dashboard. Those interested in additional NORS data can submit a NORS data request; however, CDC generally does not distribute any information about specific people or facilities involved in outbreaks.
Only CDC and state and local health departments participating in the CaliciNet network can directly access CaliciNet data. Participating laboratories must get permission from CDC to use the data in research studies.