Lower Levels of Antiretroviral Therapy Enrollment Among Men with HIV Compared with Women — 12 Countries, 2002–2013
, MBChB1; , PhD1; F, MD2; , MD2; , PhD3; , MPH3; , MD3; , MSc3; , MD4; , MD5; , MBChB6; , MD6; , MD6; , MSc6; , MD7; , MD8; , MD9; , MBChB9; , MD10; , MSAE11; , MD12; , MD13; , MD13; , MSc14; , MD15; , MD15; , MD15; , MD15; , MD16; , DrPH17; , MBChB17; , MD18; , PhD19; , MD20; , MPH19; , PhD21; , MD22; , MD23; , MSc20; , MD24; , MD24; , MPH25; , MSc26; , MD26; , MSc27; , MD27; , MD27; , MD28; , MD29; , PhD29; , MBChB29; , MD25; , MD30; , ScD30; , MPH31; , PhD31; , MD32; , MD32; , MD33; , MD33; , MD33; 33; , MD34; , MD34; , PhD34; , MD35; , MD36; , MPH1; , PhD1; , MD1; , MD1; Georgette Adjorlolo-Johnson, MD37; Richard Marlink, MD16; David Lowrance, MD38; Thomas Spira, MD1; Robert Colebunders, MD20; David Bangsberg, MD39; Aaron Zee, MPH1; Jonathan Kaplan, MD1; Tedd V. Ellerbrock, MD1
Equitable access to antiretroviral therapy (ART) for men and women with human immunodeficiency virus (HIV) infection is a principle endorsed by most countries and funding bodies, including the U.S. President's Emergency Plan for AIDS (acquired immunodeficiency syndrome) Relief (PEPFAR) (1). To evaluate gender equity in ART access among adults (defined for this report as persons aged ≥15 years), 765,087 adult ART patient medical records from 12 countries in five geographic regions* were analyzed to estimate the ratio of women to men among new ART enrollees for each calendar year during 2002–2013. This annual ratio was compared with estimates from the Joint United Nations Programme on HIV/AIDS (UNAIDS)† of the ratio of HIV-infected adult women to men in the general population. In all 10 African countries and Haiti, the most recent estimates of the ratio of adult women to men among new ART enrollees significantly exceeded the UNAIDS estimates for the female-to-male ratio among HIV-infected adults by 23%–83%. In six African countries and Haiti, the ratio of women to men among new adult ART enrollees increased more sharply over time than the estimated UNAIDS female-to-male ratio among adults with HIV in the general population. Increased ART coverage among men is needed to decrease their morbidity and mortality and to reduce HIV incidence among their sexual partners. Reaching more men with HIV testing and linkage-to-care services and adoption of test-and-treat ART eligibility guidelines (i.e., regular testing of adults, and offering treatment to all infected persons with ART, regardless of CD4 cell test results) could reduce gender inequity in ART coverage.
Three approaches to sampling and analysis were employed in the 12 studied countries (Table). In Botswana, Haiti, Mozambique, and Namibia, where large, centralized, electronic ART patient monitoring systems are employed, all available data from 2002–2013 were analyzed. In each of these countries, 67%–100% of all ART patients and 58%–100% of all ART facilities were captured in the electronic system. In Côte d'Ivoire, Nigeria, Swaziland, Vietnam, and Zimbabwe, nationally representative samples of ART facilities were selected, with probability of selection proportional to size. In Tanzania, Uganda, and Zambia, health facilities were purposively selected by investigators to represent the range of ART facilities in each country and ensure that the study remained feasible. Among the eight sample-based surveys, a sample frame of study-eligible ART patients was created at each selected facility, and simple random sampling was used to select the sample of records. Eligibility criteria included initiation of ART ≥6 months before data abstraction, during 2002–2013, and at age ≥15 years. Data were abstracted from ART records onto standardized abstraction forms by trained study personnel.
For each of the 12 countries, the ratio of women to men who were newly enrolled in ART during 2002–2013 was compared with the current ratio of women to men among cumulative ART patients who were alive on ART by the end of each calendar year and with UNAIDS estimates of the ratio of women to men among adults living with HIV for each calendar year. To assess a country's ART program accessibility to women with HIV compared with men with HIV, the percent difference between the most recently available female-to-male new ART enrollee ratio and the UNAIDS estimate of the ratio of women to men among persons with HIV in the general population for the same calendar year was calculated. Data were analyzed using statistical software, and study design was controlled for during analyses.
Across the 12 countries, 765,087 adult ART patient records were analyzed. (Graphs of data for all countries are available online at http://stacks.cdc.gov/view/cdc/35684.) In all countries except Vietnam, the most recent estimates of the female-to-male ratio among new ART enrollees, and the ratio of women to men currently enrolled in ART exceeded the UNAIDS female-to-male ratios among persons with HIV. In addition, in seven countries (Botswana, Côte d'Ivoire, Haiti, Nigeria, Mozambique, Swaziland, and Zambia), point estimates of the ratio of female-to-male new ART enrollees increased more sharply over time than did the UNAIDS female-to-male ratios among persons with HIV. The trends in female-to-male ratios of current ART enrollees closely paralleled the new ART enrollee ratio trends.
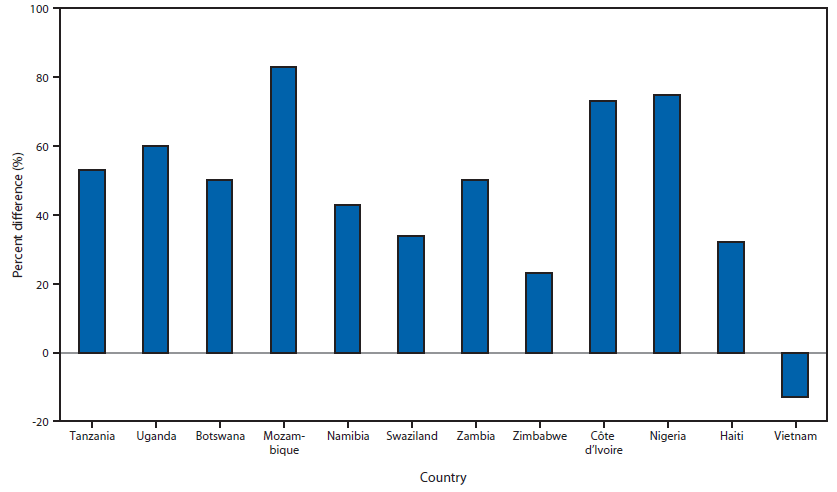
In east Africa, the most recent female-to-male new ART enrollee ratios were 2.10 in both Tanzania and Uganda for 2009; in contrast, the 2009 UNAIDS female-to-male ratios among adults with HIV were 1.38 and 1.31, respectively. Compared with males, adult females with HIV were approximately 53% and 60% more likely to access ART in Tanzania and Uganda, respectively (Figure).
In southern Africa, the most recent female-to-male new ART enrollee ratios were 1.95 in Botswana (2013); 2.73 in Mozambique (2013); 1.61 in Namibia (2012); 1.91 (95% confidence interval [CI] = 1.70–2.13) in Swaziland (2010); 1.57 in Zambia (2009); and 1.76 (95% CI = 1.53–1.99) in Zimbabwe (2009); whereas the corresponding calendar year UNAIDS female-to-male ratios among adults with HIV for these countries were 1.30, 1.49, 1.13, 1.43, 1.05, and 1.43, respectively. Compared with males living with HIV in southern Africa, females living with HIV were 23%–83% more likely to access ART (Figure).
In west Africa, the most recent female-to-male new ART enrollee ratios were 2.21 (95% CI = 1.77–2.64) in Côte d'Ivoire (2007) and 2.34 (95% CI = 1.86–2.83) in Nigeria (2011); the corresponding calendar year UNAIDS female-to-male ratios among adults with HIV were 1.28 and 1.34, respectively. Compared with men, adult women with HIV were about 73% and 75% more likely to access ART in Côte d'Ivoire and Nigeria, respectively.
In Haiti in 2013, the female-to-male new ART enrollee ratio was 1.89, and the UNAIDS female-to-male ratio among persons with HIV was 1.43. Compared with men, adult women with HIV were 32% more likely to access ART in 2013. Finally, in Vietnam in 2009, the female-to-male new ART enrollee ratio was 0.34 (95% CI = 0.27–0.41), which was similar to the UNAIDS female-to-male ratio among persons with HIV (0.39).
Discussion
This analysis of 765,087 adult ART patient records from 12 countries is the most up-to-date and comprehensive assessment of disproportionate ART enrollment among adult women with HIV compared with men, in resource-limited settings (2). In 10 African countries and Haiti (countries with generalized HIV epidemics) women with HIV were more likely to access ART than men with HIV. In addition, in six African countries and Haiti, gender-related disparities in ART coverage appear to be increasing over time. The adult ART program sex distribution was largely reflective of the UNAIDS female-to-male ratio among persons with HIV in only one country, Vietnam.
Higher ART coverage among adult women with HIV in the African countries and Haiti could occur for a number of potential reasons. First, HIV testing and counseling is a part of routine antenatal care, which provides an early entry point to ART for women with HIV. Second, ART eligibility criteria are currently more inclusive for adult women with HIV than men because, to prevent mother-to-child transmission (PMTCT) for pregnant women with HIV, all 12 countries except Nigeria§ have adopted guidelines recommending universal, lifelong ART, regardless of the results of the CD4 cell count test (referred to as PMTCT Option B+). Third, differences between men and women in health-seeking behavior might also play a role, with men considered more likely to delay access to health care for reasons that include stigma, male norms that discourage admitting ill health, and employment responsibilities, which might involve within-country and cross-border migration (3).
In many of the countries studied, gender inequity in ART coverage appears to be increasing. At the patient level, the recent initiation of PMTCT B+ might explain recent disproportionate accelerations in ART coverage among women in some countries (e.g., Mozambique initiated PMTCT B+ in 2013). However, at governance- and funder-levels, lack of initiatives to address gender inequities in ART coverage might result from tacitly holding men responsible for failing to access ART services, rather than assigning responsibility for improving male ART coverage to global health programs (4). Recent data show that men's health is often considered a lower priority than women's health in global health programs (5). However, this prioritization is not based on disease burden as estimated using disability-adjusted life years: HIV and the other nine top contributors to global disability-adjusted life years are more burdensome in men than in women (5).
Of the 12 countries studied, only Vietnam had female-to-male new ART enrollee ratios similar to UNAIDS female-to-male ratios among persons with HIV. A possible explanation is that Vietnam has a concentrated epidemic, affecting predominantly male persons who inject drugs, and therefore, from the beginning, the ART program in Vietnam has been focused on addressing the disease within this population (6). In Vietnam, men with HIV commonly access ART through routine HIV testing and counseling at needle and syringe exchange programs and methadone maintenance therapy clinics (6). In contrast, women with HIV primarily access HIV testing and linkage to ART via outreach activities to female sex workers, and through routine HIV testing at antenatal care clinics; this coverage was low in 2005, but is increasing (6,7). Continued monitoring of Vietnam's ART program gender ratios is warranted, as women account for increasing proportions of new HIV infections (6).
The findings in this report are subject to at least four limitations. First, UNAIDS estimates of female-to-male ratios among all persons with HIV are derived from epidemic models with inherent uncertainty, limiting the ability to make statistical comparisons between UNAIDS-derived and cohort-derived ratios. Second, cohort data varied in size and generalizability. Third, this study analyzed average female-to-male ratios for adults; future analyses to examine effect modification across adult age groups are needed. Finally, this analysis did not evaluate gender ratios among persons being tested for HIV or linking to care, which would help explain observed ratios among ART enrollees.
Increasing ART coverage among men with HIV would reduce morbidity and mortality in this group and contribute to reducing HIV incidence among their sex partners (8), including adolescent girls and young women, a priority population for PEPFAR.¶ Strategic program changes needed to reach more HIV-infected men with ART include identification of routine HIV testing systems, similar to HIV testing and counseling for women in antenatal care settings, and adoption of test-and-treat guidelines, which was recommended by the World Health Organization for the first time this year (9). Although more data on how to increase HIV testing and linkage to ART among HIV-infected men in resource-limited settings are needed, available evidence suggests a strategic combination of facility- and community-based approaches is required (10). From a program management perspective, ensuring that men are not overlooked in gender-related strategic documents prepared by funders (5), special initiatives to reach men with HIV, performance-based financing that provides incentives to reach both men and women, and tailored program evaluation strategies, including gender disaggregation of HIV treatment cohort data (5), are needed.
1Division of Global HIV/AIDS, Center for Global Health, CDC; 2National Institute of Health, Mozambique; 3Ministry of Health, Botswana; 4Ministry of Health and Social Services, Namibia; 5Ministry of Health, Swaziland; 6Ministry of Health, Zimbabwe; 7Muhimbili University of Health and Allied Sciences, Tanzania; 8Infectious Diseases Institute, Makerere University College of Health Sciences, Uganda; 9Tropical Diseases Research Center, Zambia; 10Ministry of Health, Côte d'Ivoire; 11Directorate General of Budget and Finance, Côte d'Ivoire; 12Ministry of Health, Nigeria; 13Vietnam Authority of HIV/AIDS Control, Vietnam; 14Hanoi School of Public Health, Vietnam; 15Programme National Lutte Sida/MSPP, Haiti; 16Harvard T.H. Chan School of Public Health, Boston, Massachusetts; 17ICAP, New York, New York; 18Gates Foundation, Seattle, Washington; 19Social and Behavioral Health Sciences, FHI 360, Washington, DC; 20Institute of Tropical Medicine, Department of Clinical Sciences, Belgium; 21FHI 360, Zambia; 22FHI 360, Haiti; 23FHI 360, Tanzania; 24Global Health, Population and Nutrition, FHI 360, Durham, North Carolina; 25University of Texas, School of Biomedical Informatics, Houston, Texas; 26National Alliance of State & Territorial AIDS Directors, Washington, DC; 27Division of Global HIV/AIDS, Center for Global Health, CDC, Mozambique; 28Division of Global HIV/AIDS, Center for Global Health, CDC, Botswana; 29Division of Global HIV/AIDS, Center for Global Health, CDC, Namibia; 30Division of Global HIV/AIDS, Center for Global Health, CDC, Swaziland; 31Division of Global HIV/AIDS, Center for Global Health, CDC, Zimbabwe; 32Division of Global HIV/AIDS, Center for Global Health, CDC, Côte d'Ivoire; 33Division of Global HIV/AIDS, Center for Global Health, CDC, Nigeria; 34Division of Global HIV/AIDS, Center for Global Health, CDC, Vietnam; 35Division of Global HIV/AIDS, Center for Global Health, CDC, Haiti; 36Division of Global Health Protection, Center for Global Health, CDC, South Africa; 37Elizabeth Glaser Pediatric AIDS Foundation, Los Angeles, California; 38Division of Global Health Protection, Center for Global Health, CDC, Haiti; 39Massachusetts General Hospital, Boston, Massachusetts.
Corresponding author: Andrew Auld, aauld@cdc.gov, 404-639-8997.
References
- Office of the President of the United States. President's Emergency Plan for AIDS Relief. Updated gender strategy: December 2013. Washington, DC: Office of the President of the United States; 2015. Available at http://www.pepfar.gov/documents/organization/219117.pdf.
- Muula AS, Ngulube TJ, Siziya S, et al. Gender distribution of adult patients on highly active antiretroviral therapy (HAART) in Southern Africa: a systematic review. BMC Public Health 2007;7:63.
- Cornell M, Schomaker M, Garone DB, et al.; International Epidemiologic Databases to Evaluate AIDS Southern Africa Collaboration. Gender differences in survival among adult patients starting antiretroviral therapy in South Africa: a multicentre cohort study. PLoS Med 2012;9:e1001304.
- Cornell M, Myer L. Moving beyond gender stereotypes. Lancet 2013;382:506.
- Hawkes S, Buse K. Gender and global health: evidence, policy, and inconvenient truths. Lancet 2013;381:1783–7.
- Kato M, Long NH, Duong BD, et al. Enhancing the benefits of antiretroviral therapy in Vietnam: towards ending AIDS. Curr HIV/AIDS Rep 2014;11:487–95.
- Dinh TH, Detels R, Nguyen MA. Factors associated with declining HIV testing and failure to return for results among pregnant women in Vietnam. AIDS 2005;19:1234–6.
- Cohen MS, Chen YQ, McCauley M, et al. Prevention of HIV-1 infection with early antiretroviral therapy. N Engl J Med 2011;365:493–505.
- Lundgren JD, Babiker AG, Gordin F, et al. Initiation of antiretroviral therapy in early asymptomatic HIV Infection. N Engl J Med 2015;373:795–807.
- Hensen B, Taoka S, Lewis JJ, Weiss HA, Hargreaves J. Systematic review of strategies to increase men's HIV-testing in sub-Saharan Africa. AIDS 2014;28:2133–45.
* East Africa: Tanzania, Uganda; Southern Africa: Botswana, Mozambique, Namibia, Swaziland, Zambia, Zimbabwe; West Africa: Côte d'Ivoire, Nigeria; Caribbean: Haiti; Southeast Asia: Vietnam.
† Additional information available at http://aidsinfo.unaids.org/.
§ Additional information available at http://www.hivpolicywatch.org/.
¶ Additional information available at http://www.pepfar.gov/partnerships/ppp/dreams/index.htm.
Summary
What is already known on this topic?
Equitable access to antiretroviral therapy (ART) for human immunodeficiency virus (HIV)-infected men and women is a principle endorsed by most countries and funding bodies, including the U.S. President's Emergency Plan for AIDS Relief (PEPFAR).
What is added by this report?
To evaluate gender equity in ART access, 765,087 adult ART patient medical records from 12 countries were analyzed to estimate the female-to-male new ART enrollee ratio for each calendar year during 2002–2013. This annual ratio was compared with corresponding Joint United Nations Programme on HIV/AIDS (UNAIDS) estimates of adult female-to-male ratios among all persons with HIV. In all 10 African countries and Haiti, the most recent estimate of the ratio of women to men newly enrolled in ART significantly exceeded the UNAIDS estimate of the ratio of women to men among persons with HIV by 23%–83%.
What are the implications for public health practice?
Reaching more men with HIV testing and linkage-to-care services and adoption of test-and-treat ART-eligibility guidelines could reduce gender inequity in ART coverage. Government- and donor-level policy and management shifts, including endorsement of male-health–focused strategies, performance-based financing that provides incentives to reach both men and women, and gender disaggregation of HIV treatment cohort data are also needed. Prioritizing increased ART coverage among men with HIV could decrease male morbidity and mortality and reduce HIV incidence among sexual partners.
FIGURE. Percent difference between female-to-male new antiretroviral therapy enrollee ratios and corresponding UNAIDS ratios of females to males among all persons with HIV, by country* — 12 countries, 2002–2013
Abbreviations: AIDS = acquired immunodeficiency syndrome; HIV = human immunodeficiency virus; UNAIDS = Joint United Nations Programme on HIV/AIDS.
* East Africa: Tanzania, Uganda; Southern Africa: Botswana, Mozambique, Namibia, Swaziland, Zambia, Zimbabwe; West Africa: Côte d'Ivoire, Nigeria; Caribbean: Haiti; Southeast Asia: Vietnam.
Alternate Text: The figure above is a bar chart showing the percent difference between female-to-male new antiretroviral therapy enrollee ratios and corresponding UNAIDS ratios of females to males among all persons with HIV, by country, in 12 countries during 2002-2013.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


