Lipids Standardization Program
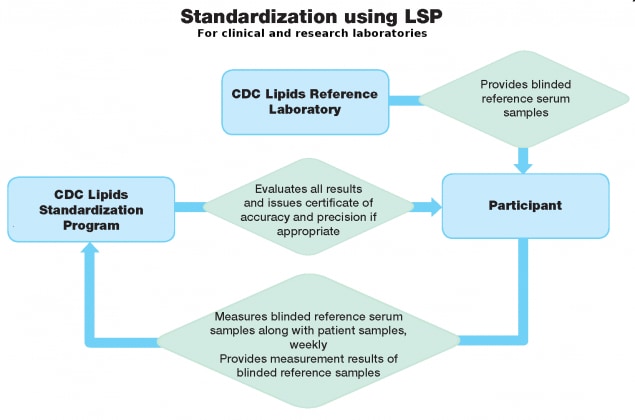
The Lipids Standardization Program (LSP) monitors the accuracy of research and clinical laboratories over time. LSP ensures the analytical accuracy and precision of measurements performed in research studies and routine clinical laboratories by providing blinded standards (LSP samples) traceable to the CDC Reference Laboratory for measuring total cholesterol (TC), glycerides (TG), high-density lipoprotein cholesterol (HDL-C), apolipoprotein A-I (apo A-I), and apolipoprotein B (apo B). The LSP is unique among external quality-control systems (EQAS) because it provides a way to establish, assess, and improve the analytical accuracy and precision measurements over time.
LSP participants (clinical and research laboratories) report their measurement results from the LSP samples to CDC where results are evaluated. Those participants meeting defined performance criteria for accuracy and precision receive a certificate of performance and are considered CDC-certified.
Disclaimer
Use of trade names is for identification only and does not constitute endorsement by the CDC or the U.S. Department of Health and Human Services.
