What’s Improved?
Why It Matters
Healthcare professionals order more than 14 billion laboratory tests annually in the United States. Many medical decisions are highly dependent on the test results. To ensure quality of care, laboratory measurements must be accurate and reliable. Inaccurate measurements and incorrect decisions can have far-reaching consequences for individuals and the population at large.

Consider the following:
- The decision as to whether a patient has diabetes and needs treatment is based on a blood test, for example, when the A1c test shows a value higher than 6.5%.
- The decision as to whether a person is vitamin D-deficient, and therefore at high risk for rickets, is based on a blood test, for example, vitamin D levels lower than 30 nmol/L.
- The decision as to whether a treatment for breast cancer is working is based on a blood test, for example, when estradiol levels are lower than 10 pg/mL because of aromatase inhibitor therapy.
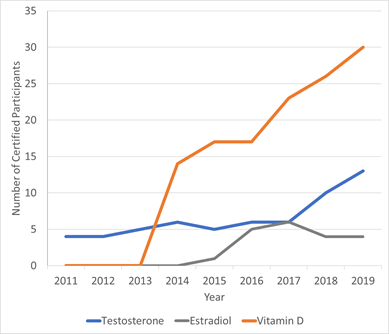
CDC Clinical Standardization Programs (CDC CSPs) are voluntary; however, participation has continued to increase over time, with over 220 program participants in 36 countries.
CDC CSPs Hormone and Vitamin D Programs Participation Over Time