Vaccine and Other Medical Countermeasures
Vaccine and Other Medical Countermeasures
Medical countermeasures (MCMs) are central to the public health response to mitigate the impact of influenza pandemics. Safe, effective, and readily available MCMs, including vaccines, antiviral drugs, therapeutics, diagnostics, ventilators, and respiratory protection devices, are critical to HHS’s goal in preventing influenza and reducing its effects on health and society during a future pandemic. An effective influenza pandemic response includes developing, manufacturing, distributing, dispensing, and administering MCMs in the shortest time possible, and monitoring their impact when used during a public health emergency.
For a summary of planning priorities for pandemic influenza MCMs, please see the HHS 2017 pandemic influenza plan update: Domain 3- Medical Countermeasures: Diagnostics, Devices, Vaccines and Therapeutics
Vaccine
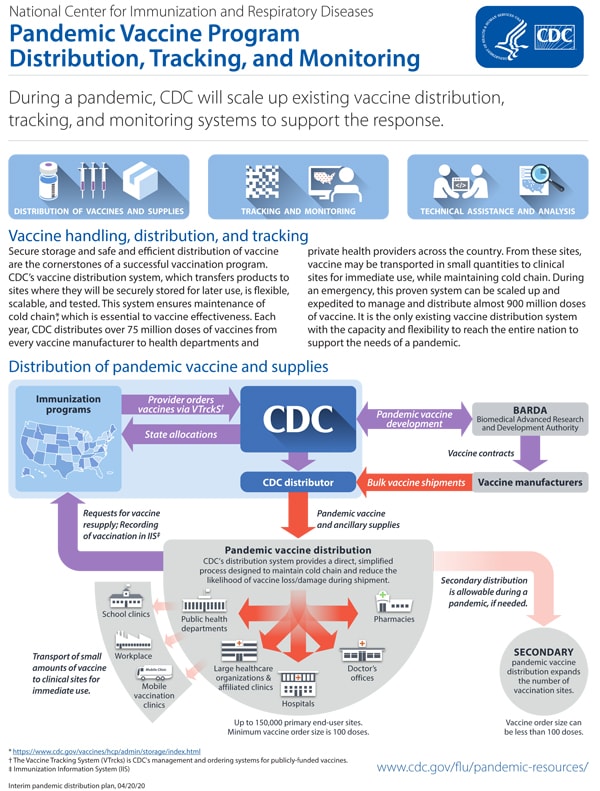
Vaccination is the most effective medical countermeasure for mitigating the potentially devastating impact of an evolving influenza pandemic. This requires developing a well-matched, safe, and effective vaccine in the shortest time possible; determining the appropriate dose; and administering it promptly. If a severe influenza pandemic emerges, a well-coordinated vaccination campaign would need to be implemented immediately to reduce morbidity and mortality.
The following are a collection of federal resources designed to guide influenza pandemic planning related to influenza vaccines during a pandemic:
Other Medical Countermeasures
Other MCMs are critically important to mitigate emerging pandemics before effective vaccines become available. These include pharmaceuticals (e.g. antiviral drugs, antibiotics), and critical materiel (personal protective equipment including respiratory protective equipment, and ventilators). Additional resources designed to guide influenza pandemic planning related to non vaccine medical countermeasures is forthcoming.
- HHS Biomedical Advanced Research and Development Authority (BARDA) Influenza Division is charged with the advanced development of medical countermeasures for pandemic influenza preparedness and response.
- HHS’ Strategic National Stockpile
- Making a Candidate Vaccine Virus (CVV) for a Highly Pathogenic Avian Influenza (Bird Flu) Virus
- National Institute of Allergy and Infectious Disease Influenza Vaccines
- FDA Medical Countermeasure (MCM) Legal, Regulatory and Policy Framework
- FDA Medical Countermeasure (MCM) Legal, Regulatory and Policy Framework
- FDA Emergency Use Authorities
- FDA Emergency Use Authority of Medical Products and Related Authorities (April 2016)
- FDA Emergency use of an Investigational Drug or Biologic
- WHO’s summary report on the antigenic and genetic characteristics of zoonotic influenza viruses and candidate vaccine viruses developed for potential use in human vaccines