Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Update: Influenza Activity — United States and Worldwide, May 20–September 22, 2012
During May 20–September 22, 2012, the United States experienced low levels of seasonal influenza activity overall; however, more seasonal influenza viruses were detected than in the summer months of previous years. Influenza A (H1N1)pdm09 (pH1N1), influenza A (H3N2), and influenza B viruses were detected worldwide and were identified sporadically in the United States. In July, influenza A (H3N2) variant viruses (H3N2v) were first detected in Indiana, and since July 12, a total of 306 cases have been reported from 10 states. This report summarizes influenza activity in the United States and worldwide since May 20, 2012.
United States
The U.S. influenza surveillance system is a collaborative effort between CDC and its federal, state, local, and territorial partners. CDC uses eight systems* to collect influenza information (1), six of which operate year-round: 1) U.S. World Health Organization (WHO) collaborating laboratories; 2) the National Respiratory and Enteric Virus Surveillance System (NREVSS); 3) reports of novel influenza A virus cases from the National Notifiable Disease Surveillance System (NNDSS); 4) the U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet); 5) the 122 Cities Mortality Reporting System; and 6) the Influenza-Associated Pediatric Mortality Reporting System.
During May 20–September 22, 2012,† U.S. WHO and NREVSS collaborating laboratories tested 42,562 respiratory specimens for influenza viruses; 2,986 (7.0%) tested positive for influenza (Figure), indicating higher levels of activity than typically seen in summer months, but lower levels than during winter months and the height of influenza virus circulation. During the summer months of the previous 6 years (excluding the summer during the 2009 pandemic) the average number of respiratory specimens tested for influenza was 29,728 (range: 20,652–39,523), with an average of 375 (1.3%) specimens testing positive (range: 245–541). Of the 2,986 specimens positive for influenza in the summer months of 2012, a total of 1,497 (50%) were influenza A viruses, and 1,489 (50%) were influenza B viruses. Influenza B viruses predominated and were reported more frequently than influenza A viruses from May until mid-July; influenza A (H3N2) viruses were more commonly reported from mid-July to September. Of the influenza A viruses, 1,117 (75%) were subtyped: 759 (68%) were influenza A (H3N2) viruses, 263 (24%) were H3N2v viruses,§ and 95 (9%) were pH1N1 viruses. Influenza viruses were reported from 44 states and Puerto Rico in all 10 U.S. Department of Health and Human Services (HHS) Regions. The largest proportion of positive samples came from the southeastern United States (HHS Region 4: Alabama, Florida, Georgia, Kentucky, Mississippi, North Carolina, South Carolina, and Tennessee) with 1,133 (38%), followed by western states (HHS Region 9: Arizona, California, Hawaii, and Nevada) with 726 (24%).
During May 20–September 22, 2012, data from ILINet indicated that the weekly percentage of outpatient visits to ILINet providers for influenza-like illness (ILI)¶ remained below the national baseline** of 2.4% and ranged from 0.9% to 1.3%. The percentage of deaths attributed to pneumonia and influenza (P&I), as reported by the 122 Cities Mortality Reporting System, remained below the epidemic threshold†† and ranged from 5.5% to 6.6%. Two influenza-associated pediatric deaths during the period May 20–September 22 were reported; one was associated with an influenza B virus, and one was associated with a pH1N1 virus.
Novel Influenza A Virus Infection
During July 12–September 28, 2012, 306 cases of H3N2v virus were reported from 10 states (Hawaii [one], Illinois [four], Indiana [138], Maryland [12], Michigan [six], Minnesota [four], Ohio [107], Pennsylvania [11], West Virginia [three], and Wisconsin [20]), with 16 H3N2v-associated hospitalizations and one H3N2v-associated death. Although cases have been identified from 10 states, two states (Indiana and Ohio) have reported 245 (80%) of the 306 cases. Direct contact with swine has been reported by the vast majority of cases, and influenza A (H3N2) viruses have been identified from swine that are genetically similar to H3N2v viruses from humans (2). Suspected human-to-human transmission has been identified in a small number of cases, but ongoing community transmission of this virus has not been detected. The median age of patients was 6 years, with 284 (93%) aged <18 years; 52% were female (CDC, unpublished data, 2012).
In addition, three cases of influenza A (H1N2) variant (H1N2v) virus infection and one case of influenza A (H1N1) variant (H1N1v) virus were detected during this period as a result of enhanced surveillance activities for H3N2v. All four patients reported direct exposure to swine in the week before illness onset; one was hospitalized, and all four have recovered.
Worldwide
During May 20–September 22, 2012, typical seasonal patterns of influenza activity occurred in the temperate climate Southern Hemisphere countries. In Australia, influenza activity began increasing in late May, and peaked in mid-July; influenza A (H3N2) virus predominated with smaller numbers of cases of influenza B virus infection reported. In New Zealand, influenza activity began increasing in late June, peaked in early August, and has since been decreasing. Influenza A (H3N2) virus was overwhelmingly predominant, with lower levels of influenza B virus detected. In South Africa, influenza activity began to increase in early June with increased levels of activity being reported through August. Influenza A (H3N2) viruses have been reported most commonly, but a larger proportion of influenza-positive specimens in South Africa are influenza B viruses than in Australia or New Zealand. In South America, influenza activity peaked earlier in the season and is now decreasing. Influenza A viruses were reported more frequently than influenza B viruses, but the predominant subtype varied by country. Argentina reported a larger proportion of positive specimens as pH1N1 viruses than other countries in the region, but the overall number of influenza positive specimens there was lower than in previous seasons.
Influenza activity also has been reported from countries with tropical influenza seasonality. The overall level of activity compared with previous seasons and the predominant subtype have varied by country. In South America, influenza A viruses have predominated in Brazil, but in Ecuador and Peru influenza B viruses have been reported most commonly. Southern and Southeast Asia also have seen a mix of predominant influenza types and subtypes with influenza B and pH1N1 viruses cocirculating in several countries, including Bangladesh, India, Sri Lanka, and Thailand. In temperate climate Northern Hemisphere countries, influenza activity remains low, compared with levels of activity during the usual influenza season, with small numbers of influenza A (H3N2), pH1N1, and influenza B viruses identified.
Antigenic Characterization of Influenza Virus Isolates
The WHO Collaborating Center for Surveillance, Epidemiology, and Control of Influenza, located at CDC, receives and analyzes virus isolates from laboratories worldwide. Among the 111 pH1N1 viruses collected from May 20 to September 22, 2012 and analyzed (28 from the United States, 57 from South America, one from Oceania, 17 from Asia, and eight from Africa), 102 (92%) were antigenically similar to A/California/7/2009, the influenza A (H1N1) component of the 2012-2013 influenza vaccine for the Northern Hemisphere. Of the 241 influenza A (H3N2) viruses characterized (157 from the United States, 55 from South America, four from North America, four from Oceania, 20 from Asia, and one from Africa), all were antigenically similar to A/Victoria/361/2011, the recommended influenza A (H3) vaccine component for the 2012–2013 Northern Hemisphere influenza season. Finally, of 271 influenza B isolates from specimens collected during this period and analyzed by CDC, 113 (42%) belong to the B/Yamagata lineage (76 from the United States, 17 from Asia, two from Oceania, and 18 from South America) and were antigenically similar to B/Wisconsin/1/2010, the recommended influenza B component for the 2012–2013 Northern Hemisphere influenza vaccine. The remaining 158 (58%) belonged to the B/Victoria lineage (93 from the United States, 47 from South America, 15 from Asia, and three from Africa), nearly all of which (95%) were antigenically similar to B/Brisbane/60/2008, the recommended influenza B component in the 2011–2012 Northern Hemisphere influenza vaccine.
Antiviral Resistance Profiles of Influenza Virus Isolates
The WHO Collaborating Center for Surveillance, Epidemiology, and Control of Influenza at CDC tested 594 isolates from specimens collected during May 20–September 22, 2012, for resistance to influenza antiviral medications. Of the 594 isolates tested for resistance to the neuraminidase inhibitor medications oseltamivir and zanamivir, 238 were international isolates (84 were pH1N1, 76 were influenza A (H3N2), and 78 were influenza B viruses), and 356 were U.S. isolates (30 were pH1N1, 158 were influenza A (H3N2), and 168 were influenza B viruses). Only one virus (a pH1N1 virus from the United States) was found to be resistant to oseltamivir but sensitive to zanamivir, and it contained the H275Y mutation in the neuraminidase. High levels of resistance to the adamantanes (amantadine and rimantadine) persist among pH1N1 viruses and influenza A (H3N2) viruses currently circulating globally (3). All 117 H3N2v viruses available for testing were sensitive to oseltamivir and resistant to the adamantanes.
Reported by
World Health Organization Collaborating Center for Surveillance, Epidemiology, and Control of Influenza. Scott Epperson, MPH, Lynnette Brammer, MPH, Lenee Blanton, MPH, Desiree Mustaquim, MPH, Margaret Okomo-Adhiambo, PhD, Larisa Gubareva, MD, Teresa Wallis, MS, Alexander Klimov, PhD, Joseph Bresee, MD, Nancy Cox, PhD, Lyn Finelli, DrPH, Influenza Div, National Center for Immunization and Respiratory Diseases, CDC. Corresponding contributor: Scott Epperson, sepperson@cdc.gov, 404-639-3747.
Editorial Note
During May 20–September 22, 2012, pH1N1, influenza A (H3N2), and influenza B viruses cocirculated worldwide. In the United States, more seasonal influenza viruses were detected than in the summer months of previous years (excluding the 2009 pandemic), and influenza A (H3N2) viruses were predominant. Although neither the influenza viruses that will predominate nor the severity of influenza-related disease during the 2012–13 season in the United States can be predicted, antigenic characterization of viral isolates from specimens submitted during the summer demonstrated that the majority of influenza A viruses are antigenically similar to the influenza vaccine strains contained in the Northern Hemisphere 2012–13 vaccine.
H3N2v viruses with the matrix (M) gene from the pH1N1 virus were first detected in the United States in July 2011 (4). Since the first identification of this virus in humans, direct contact with swine has been documented in almost all cases, but limited human-to-human spread also is suspected. Consistent with the age distribution of cases, serologic studies suggest there is little or no cross-reactive antibody to H3N2v in young children, but some cross-reactive antibodies are evident in older children and adults (5). Although community transmission of this virus has not been identified, the potential for this virus to develop the ability to transmit efficiently from person-to-person is of concern. Rapid and intensive investigation of each variant case is necessary to evaluate the spread of disease and the possibility of person-to-person transmission. State and local health departments should consider increased specimen collection among patients with ILI who 1) seek care at an ILINet provider; 2) are part of an ILI outbreak among children in child-care and school settings, because these settings were associated with person-to-person H3N2v virus transmission in 2011; 3) have an unusual or severe presentation of ILI, including a need for hospitalization; or 4) have medically attended ILI or acute respiratory infection, especially children in counties or states where H3N2v cases have occurred (6).
Annual influenza vaccination remains the best method for preventing influenza and its associated complications (7), but the 2012–13 seasonal influenza vaccine does not provide protection against the H3N2v virus. Treatment with influenza antiviral medications is recommended as early as possible for patients with confirmed or suspected influenza (either seasonal influenza or variant influenza infection) who have severe, complicated, or progressive illness; who require hospitalization; or who are at higher risk for influenza-related complications (8).§§
Influenza surveillance reports for the United States are posted online weekly and are available at http://www.cdc.gov/flu/weekly. Additional information regarding influenza viruses, influenza surveillance, influenza vaccine, influenza antiviral medications, and novel influenza A infections in humans is available at http://www.cdc.gov/flu.
Acknowledgments
State, local, and territorial health departments and public health laboratories; U.S. WHO collaborating laboratories; National Respiratory and Enteric Virus Surveillance System collaborating laboratories; U.S. Outpatient Influenza-like Illness Surveillance Network; Influenza-Associated Pediatric Mortality Surveillance System; 122 Cities Mortality Reporting System; WHO FluNet.
References
- Brammer L, Blanton, L, Epperson S, et al. Surveillance for influenza during the 2009 influenza A (H1N1) pandemic—United States, April 2009–March 2010. Clin Infect Dis 2011;52 (Suppl 1):S27–35.
- CDC. Notes from the field: outbreak of influenza A (H3N2) virus among persons and swine at a county fair—Indiana, July 2012. MMWR 2012:61;561.
- World Health Organization. Summary of influenza antiviral susceptibility surveillance findings, September 2010–March 2011. Geneva, Switzerland: World Health Organization; 2011. Available at http://www.who.int/csr/disease/influenza/influenzanetwork/flunet/antiviral_susceptibility/en/index.html. Accessed October 1, 2012.
- CDC. Swine-origin influenza A (H3N2) virus infection in two children—Indiana and Pennsylvania, July–August 2011. MMWR 2011;60:1213–5.
- CDC. Antibodies cross-reactive to influenza A (H3N2) variant virus and impact of 2010–11 seasonal influenza vaccination on cross-reactive antibodies—United States. MMWR 2012;61:237–41.
- CDC. Interim guidance for enhanced influenza surveillance: additional specimen collection for detection of influenza A (H3N2) variant virus infection. Atlanta, GA: US Department of Health and Human Services, CDC; 2012. Available at http://www.cdc.gov/flu/swineflu/h3n2v-surveillance.htm. Accessed October 1, 2012.
- CDC. Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP)—United States, 2012–13 influenza season. MMWR 2012;61:613–8.
- CDC. Antiviral agents for the treatment and chemoprophylaxis of influenza—recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 2011;60(No. RR-1).
* The CDC influenza surveillance system collects five categories of information from eight data sources: 1) viral surveillance (World Health Organization collaborating laboratories, the National Respiratory and Enteric Virus Surveillance System, and novel influenza A virus case reporting); 2) outpatient illness surveillance (U.S. Outpatient Influenza-like Illness Surveillance Network); 3) mortality (122 Cities Mortality Reporting System and influenza-associated pediatric mortality reports); 4) hospitalizations (FluSurv-NET, which includes the Emerging Infections Program and surveillance in four additional states); and 5) summary of the geographic spread of influenza (state and territorial epidemiologist reports).
† Data as of September 28, 2012.
§ Influenza viruses that circulate in swine are called swine influenza viruses when isolated from swine, but are called variant viruses when isolated from humans. A variant virus (human isolate) might or might not have the M gene from the influenza A (H1N1)pdm09 virus, along with other genetic changes. Seasonal influenza A (H3N2) viruses that circulate worldwide in the human population have significant antigenic and genetic differences from influenza A (H3N2) viruses circulating in swine. Additional information is available at http://www.who.int/influenza/gisrs_laboratory/terminology_ah3n2v/en/index.html.
¶ Defined as a temperature of ≥100°F (≥37.8°C), oral or equivalent, and cough and/or sore throat, without a known cause other than influenza.
** The national and regional baselines are the mean percentage of visits for ILI during noninfluenza weeks for the previous three seasons plus two standard deviations. A noninfluenza week is a week during which <10% of specimens tested positive for influenza. National and regional percentages of patient visits for ILI are weighted on the basis of state population. Use of the national baseline for regional data is not appropriate.
†† The seasonal baseline proportion of P&I deaths is projected using a robust regression procedure in which a periodic regression model is applied to the observed percentage of deaths from P&I that were reported by the 122 Cities Mortality Reporting System during the preceding 5 years. The epidemic threshold is set at 1.645 standard deviations above the seasonal baseline.
§§ Persons at higher risk include children aged <5 years (especially those aged <2 years); adults aged ≥65 years; persons with chronic pulmonary (including asthma), cardiovascular (except hypertension alone), renal, hepatic, hematologic (including sickle cell disease), metabolic disorders (including diabetes mellitus), or neurologic or neurodevelopmental conditions (including disorders of the brain, spinal cord, peripheral nerve, and muscle, such as cerebral palsy, epilepsy [seizure disorders], stroke, intellectual disability [mental retardation], moderate to severe developmental delay, muscular dystrophy, or spinal cord injury); persons with immunosuppression, including that caused by medications or by human immunodeficiency virus infection; women who are pregnant or postpartum (within 2 weeks after delivery); persons aged ≤18 years who are receiving long-term aspirin therapy; American Indians/Alaska Natives; persons who are morbidly obese (i.e., body mass index ≥40); and residents of nursing homes and other chronic-care facilities.
What is already known on this topic?
CDC collects, compiles, and analyzes data year-round on influenza activity in the United States. The influenza season generally begins in the fall and continues through the winter and spring months; however, the timing and severity of circulating influenza viruses can vary by geographic location and season.
What is added by this report?
Worldwide, influenza activity from May 20 to September 22, 2012, was elevated in the temperate Southern Hemisphere and tropical regions, compared with their levels outside the usual influenza season. In the United States, low levels of seasonal influenza activity were detected, and influenza A (H3N2) viruses were most commonly identified. More than 300 cases of influenza A (H3N2) variant virus were detected in 10 states; the majority of these cases were associated with direct contact with swine. The majority of recent influenza A viruses are well-matched to the influenza vaccine for this season
What are the implications for public health practice?
To prevent influenza and its associated complications, influenza vaccination is recommended for all persons aged ≥6 months. While vaccination is the best way to prevent influenza, treatment with influenza antiviral medications can reduce severe outcomes of influenza, especially when initiated as early as possible, in patients with confirmed or suspected influenza.
FIGURE. Number* and percentage of respiratory specimens testing positive for influenza reported to CDC, by type, subtype, and surveillance week — World Health Organization and National Respiratory and Enteric Virus Surveillance System collaborating laboratories, United States, May 20–September 22, 2012.†
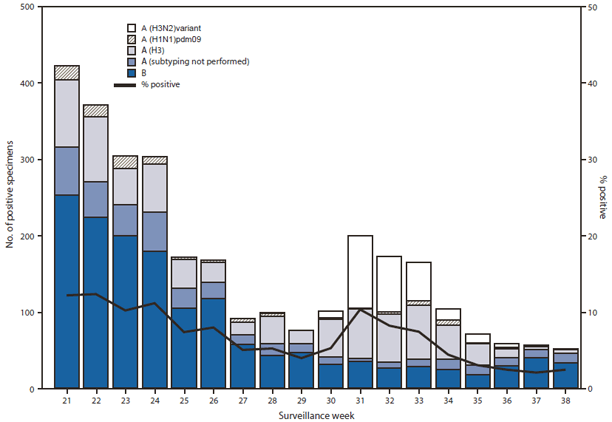
* N = 2,986.
† As of September 28, 2012.
Alternate Text: The figure above shows the number and percentage of respiratory specimens testing positive for influenza reported to CDC, by type, subtype, and surveillance week, in the United States during May 20-September 22, 2012. During May 20-September 22, 2012, U.S. World Health Organization and National Respiratory and Enteric Virus Surveillance System collaborating laboratories tested 42,562 respira¬tory specimens for influenza viruses; 2,986 (7.0%) tested positive for influenza.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


