Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Outbreak of Meningococcal Disease Associated with an Elementary School — Oklahoma, March 2010
During March 10–31, 2010, the Oklahoma State Department of Health (OSDH) investigated an outbreak of meningococcal (Neisseria meningitidis) disease involving a consolidated school district of 1,850 students in rural northeastern Oklahoma. An OSDH field investigation team and the Rogers County Health Department (RCHD) established operations at the affected elementary school as soon as the outbreak was recognized. Five cases of meningococcal disease (including one probable case) were identified among four elementary school students and one high school student. Two students died; two recovered fully, and one survivor required amputation of all four limbs and facial reconstruction. All N. meningitidis isolates were serogroup C with the same multilocus sequence type and an indistinguishable pulsed-field gel electrophoresis pattern. To interrupt the outbreak, mass vaccination and chemoprophylaxis clinics were conducted in the population at risk; 1,459 vaccinations and 1,063 courses of antibiotics were administered. Children eligible for the Vaccines for Children (VFC) program received 1,092 of the vaccine doses, demonstrating that VFC is a feasible funding source for vaccine during an outbreak response.
Outbreak and Response
On the morning of March 10, 2010, OSDH was notified that a boy aged 7 years (student A) had been hospitalized with suspected meningococcal meningitis on the basis of a preliminary cerebrospinal fluid (CSF) culture result (Figure). After N. meningitidis confirmation, RCHD conducted a routine contact investigation. Four household members received chemoprophylaxis, and one close contact of the patient was advised to seek chemoprophylaxis.
The next morning, during a 2-hour period, three additional cases of suspected meningococcal disease (in students B, C, and D), including one fatality (student B), were reported to OSDH (Figure). All four patients attended a prekindergarten through 2nd grade lower elementary school in a consolidated school district with a total enrollment of approximately 1,850 students. Four noncontiguous buildings (lower elementary, upper elementary, middle school, and high school) on a single campus provided classrooms and other facilities for children in prekindergarten through 12th grade.
The occurrence of four cases within 48 hours prompted OSDH and RCHD to begin outbreak control measures consisting of providing chemoprophylaxis to children in selected grades and to other patient contacts to provide short-term protection of the population at risk. RCHD mobilized personnel to operate an interim mass chemoprophylaxis clinic onsite at the school. The clinic began operation at noon on March 11 (Figure), after the OSDH field epidemiology team arrived with antibiotics.
Chemoprophylaxis was targeted initially to the 443 students and 50 faculty members in the lower elementary school and to close contacts of the patients. American Academy of Pediatrics guidelines recommend oral rifampin in 4 doses over 2 consecutive days or a single-dose intramuscular ceftriaxone injection for use as chemoprophylaxis against meningococcal disease among children (1). Intramuscular ceftriaxone was selected as the agent for children in the lower elementary school to ensure rapid initiation of chemoprophylaxis and to alleviate concerns regarding noncompliance with a 4-dose regimen of an unpalatable medication.
At approximately 4:00 p.m., OSDH was notified that student D had died and that two additional lower elementary students had been hospitalized with fever and rash (students E and F). Although these two illnesses were eventually found not to be cases of N. meningitidis infection on the basis of laboratory and clinical findings, that evidence did not become available until the following week. With the reports of an additional death and additional patients, the OSDH field team expanded chemoprophylaxis eligibility to select older students who had participated in reading instruction that placed them in direct contact with younger children in classrooms where cases were identified and to select persons who rode on buses with the patients, a total of approximately 400 additional contacts. During March 11–12, the first two chemoprophylaxis clinics operated for approximately 16 hours and administered 846 chemoprophylaxis doses.
On March 12, suspected meningococcal disease in a high school student in the district (student G) was reported to OSDH on the basis of clinical suspicion (Figure). As an additional precaution and to facilitate the chemoprophylaxis clinics, school district officials, in consultation with OSDH, dismissed all classes and canceled all extracurricular activities 1 day earlier than the scheduled, week-long spring break. Also on March 12, OSDH recommended mass meningococcal vaccination for all students, faculty, and staff members in the affected school district as a definitive outbreak control measure. Culture confirmation of N. meningitidis infection in student G was obtained on March 15.
On March 19, RCHD operated a mass meningococcal vaccination clinic at the school gymnasium, followed by vaccination outreach at the community physician's office during March 22–26 (Figure). A total of 1,459 doses of meningococcal vaccine (i.e., 1,426 doses of quadrivalent meningococcal conjugate vaccine, plus 33 doses of quadrivalent meningococcal polysaccharide vaccine [MPSV4] for those aged >55 years) were administered, resulting in vaccination of approximately 68% of students aged 4–18 years. During March 11–31, OSDH conducted active surveillance in six surrounding counties but identified no additional outbreak-linked cases.
Case Characteristics
A confirmed case of invasive meningococcal disease was defined as isolation of N. meningitidis from a normally sterile body site. A probable case was defined as N. meningitidis DNA detected by polymerase chain reaction without organism isolation in a suspected patient. A suspected case was defined as physician-reported fever and any rash in a person linked epidemiologically to a patient with a confirmed case. Two suspected cases (in students E and F) ultimately were excluded. Four cases (in students A, C, D, and G) were confirmed, and one case (in student B) was classified as probable (Table). The five patients ranged in age from five to 18 years.
All five patients required hospitalization. Meningococcemia was present in four patients, two of whom also had isolation of N. meningitidis from cerebrospinal fluid (Table). Formalin-fixed, paraffin-embedded central nervous system tissues were obtained at autopsy from the two decedents, and immunohistochemical and molecular evidence of infection with N. meningitidis was observed in the tissues. The four patients with confirmed cases had isolation of N. meningitidis serogroup C, further characterized as multilocus sequence type ST-11 with an indistinguishable pulsed-field gel electrophoresis pattern (H46N06.0037).
The five patients were in three different grades. Three of the patients, including the index patient (student A) were in the same 2nd grade classroom. One patient with confirmed meningococcal disease (student C) was in a kindergarten classroom with a younger sibling of the index patient, and the fifth patient (student G) sang in close proximity to an older sibling of the index patient in two school choirs. None of the five patients had received a meningococcal vaccination previously.
Reported by
Kristy Bradley, DVM, Lauri Smithee, PhD, Oklahoma State Dept of Health. Thomas Clark, MD, Henry Wu, MD, Raydel Mair, MS, Brian Harcourt, PhD, Leonard Mayer, PhD, Susanna Schmink, Div of Bacterial Diseases, National Center for Immunization and Respiratory Diseases; Christopher Paddock, MD, Sherif Zaki, MD, Div of High-Consequence Pathogens and Pathology, National Center for Emerging and Zoonotic Infectious Diseases; Steven Grube, MD, EIS Officer, CDC. Corresponding contributor: Steven Grube, sgrube@cdc.gov, 404-791-4663.
Editorial Note
School-related meningococcal disease outbreaks generate considerable community anxiety and require rapid, intensive public health response. In the United States, annual incidence of invasive meningococcal infection is approximately 0.5 cases per 100,000 population (2) with <1,200 cases of invasive disease reported to CDC in 2008 (3). In Oklahoma, during 2005–2009, incidence was 0.5 cases per 100,000 population, the lowest rate since 1978. In both Oklahoma and nationwide, children aged <1 year experience the highest incidence of disease (3). Approximately 5% of all cases occur during outbreaks, and elementary schools account for only 25% of school-based outbreaks (4). Case-fatality ratios are higher for outbreak-associated cases than for sporadic cases (5).
In 2009, estimated meningococcal vaccination coverage among Oklahoma teens was 29.5%, ranking 46th among states; coverage for the United States overall was 53.6% (6). During the meningococcal disease outbreak described in this report, the majority of those affected were aged 5–7 years, typically considered an age group at low risk and not recommended for routine immunization against meningococcal disease (7). In this outbreak, the rapid succession of reported cases suggested that additional cases were likely, necessitating immediate public health intervention.
Meningococcal vaccination was implemented as the definitive outbreak control measure based on a primary attack rate of 162 per 100,000 population (three primary cases in a school complex population of 1,850) (8). Vaccination is the preferred method for establishing long-term protection, and in contrast to polysaccharide vaccine, use of conjugate vaccine can achieve greater impact at lower coverage levels because of herd immunity (9,10). However, the immune response to vaccination takes 7–10 days to develop, whereas the majority of cases occur soon after the index case in school-based outbreaks (4). One third of cases occur within 2 days and three fourths within 14 days. Close household contacts of patients are at 500- to 1,000-fold increased risk and are recommended to receive chemoprophylaxis. In contrast, the estimated incidence of secondary meningococcal disease among school children is 2.5 per 100,000, or a relative risk of 2.3 (4).
If a public health decision is made to implement vaccination as an outbreak control measure after two or more cases are reported in a school, administration of chemoprophylaxis to the population at risk also should be considered, both to offer short-term protection to at-risk persons and potentially to limit transmission. Mass chemoprophylaxis is most likely to be effective when administered quickly and completely to a well-defined or closed cohort. The rapid occurrence of cases in school clusters suggests that transmission occurs rapidly among susceptible children. Each school-based outbreak has unique characteristics, including various case numbers and frequency, serogroup, and physical setting; public health and school officials should tailor their responses accordingly.
When a case in a high school student was identified during this outbreak, the population considered "at risk" was expanded, and the vaccination campaign was extended to all unvaccinated students, faculty members, and administrative personnel at the four district schools. VFC-eligible children aged 11–18 years were able to receive meningococcal vaccine at no charge. Because an outbreak was declared, thereby classifying the children involved in the outbreak as at increased risk for disease, children aged 4–10 years who met federal VFC program eligibility criteria also were able to receive free meningococcal vaccine.
State funds were available to purchase only 25% of the total 1,459 doses of vaccine administered among all age groups, including all vaccine administered to adult school employees. Of the 1,426 quadrivalent meningococcal conjugate vaccine doses administered, 1,250 doses were administered to children aged 4–18 years, of whom 1,092 (87%) were deemed VFC-eligible, illustrating that use of VFC funds can bolster vaccination coverage among eligible children in an outbreak setting. In addition, federal Section 317 funding* was used to purchase vaccine for children ineligible for VFC vaccine. Subsequently, a joint resolution by the Advisory Committee on Immunization Practices and VFC clarified that children aged 9 months–10 years who are associated with an outbreak of disease caused by a vaccine-preventable meningococcal serogroup are among those eligible for VFC (7).
Early in the outbreak investigation, school officials expeditiously agreed to host the chemoprophylaxis and vaccination clinics. Local emergency medical services were present and assisted in monitoring for adverse events associated with the chemoprophylaxis and vaccination clinics held at the school. The school used its automated parent-calling system to provide details regarding who was being advised to receive preventive chemoprophylaxis and vaccine. In addition to county and state public health agencies, local medical, fire, and law enforcement personnel mobilized quickly to help support the chemoprophylaxis clinics. The rapid, coordinated response was associated with high compliance with vaccination, which likely contributed to outbreak cessation.
References
- American Academy of Pediatrics. Meningococcal infections. In: Pickering LK, Baker CJ, Kimberlin DW, Long SS, eds. Red book: 2009 report of the Committee on Infectious Diseases. 28th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2009.
- Cohn AC, MacNeil JR, Harrison LH, et al. Changes in Neisseria meningitidis disease epidemiology in the United States, 1998–2007: implications for prevention of meningococcal disease. Clin Infect Dis 2010;50:184–91.
- CDC. Summary of notifiable diseases—United States, 2008. MMWR 2010;57(54).
- Zangwill KM, Schuchat A, Riedo FX, et al. School-based clusters of meningococcal disease in the United States: descriptive epidemiology and a case-control analysis. JAMA 1997;277:389–95.
- Brooks R, Woods CW, Benjamin, DK Jr, Rosenstein NE. Increased case-fatality rate associated with outbreaks of Neisseria meningitidis infection, compared with sporadic meningococcal disease, in the United States, 1994–2002. Clin Infect Dis 2006;43:49–54.
- CDC. National, state, and local area vaccination coverage among adolescents aged 13–17 years—United States, 2009. MMWR 2010;59:1018–23.
- CDC. Advisory Committee on Immunization Practices, Vaccines for Children Program. Vaccines to prevent meningococcal disease. Resolution no. 6/11-1. Atlanta, GA: Advisory Committee on Immunization Practices; 2011. Available at http://www.cdc.gov/vaccines/programs/vfc/downloads/resolutions/06-11mening-mcv.pdf. Accessed April 3, 2012.
- CDC. Control and prevention of serogroup C meningococcal disease; evaluation and management of suspected outbreaks: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 1997;46(No. RR-05):13–21.
- Krause G, Blackmore C, Wiersma S, Lesneski C, Gauch L, Hopkins RS. Mass vaccination campaign following community outbreak of meningococcal disease. Emerg Infect Dis 2002;8:1398–403.
- Weiss D, Stern EJ, Zimmerman C, et al. Epidemiologic investigation and targeted vaccination initiative in response to an outbreak of meningococcal disease among illicit drug users in Brooklyn, New York. Clin Infect Dis 2009;48:894–901.
* Additional information available at http://www.hhs.gov/recovery/programs/cdc/immunizationgrant.html.
What is already known on this topic?
Meningitis caused by Neisseria meningitidis is an uncommon, often fatal, infectious disease. Outbreak response can include vaccination, and if vaccination is implemented, postexposure chemoprophylaxis may be administered to protect persons until vaccine-induced immunity develops.
What is added by this report?
In 2010, an outbreak of five cases of meningococcal disease, two fatal, occurred in an Oklahoma prekindergarten through 12th grade school complex. Four patients attended the same lower elementary school, and one was a high school student. All cases were caused by indistinguishable isolates of serogroup C N. meningitidis. To stem the outbreak, public health authorities provided chemoprophylaxis to 1,063 persons and vaccination to 1,459. Of 1,250 children aged 4–18 years who received quadrivalent meningococcal conjugate vaccine, 87% were eligible for the federal Vaccines for Children (VFC) program.
What are the implications for public health practice?
During a rapidly progressive meningococcal disease outbreak in a school, a prompt and coordinated public health response following established outbreak guidelines is needed to protect those at risk. VFC is a potential funding source for purchasing vaccine for eligible children aged 9 months–18 years in an outbreak setting.
FIGURE. Timeline of major events involving invasive meningococcal disease outbreak based in an elementary school and public health response — Oklahoma, March 2010
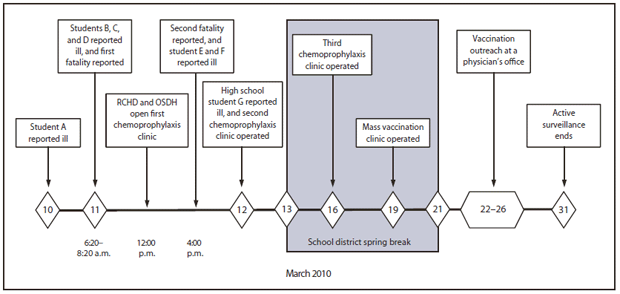
Abbreviations: RCHD = Rogers County Health Department, OSDH = Oklahoma State Department of Health.
Alternate Text: The figure above shows the timeline of major events in an invasive meningococcal disease outbreak based in an elementary school in Oklahoma during March 2010. The occurrence of four cases within 48 hours prompted the Oklahoma State Department of Health and the Rogers County Health Department to begin outbreak control measures consist¬ing of providing chemoprophylaxis to children in selected grades and to other patient contacts to provide short-term protection of the population at risk. With the report of a case in a high school student, eligibility for chemoprophylaxis was expanded. A week after the first case was reported, a mass meningococcal vaccination clinic was conducted.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


