Agar Dilution Antimicrobial Susceptibility Testing
Antimicrobial Agents and Range of dilutions (mg/L):
Standard panel:
Penicillin G: 0.008 to 64.0
Tetracycline: 0.06 to 64.0
Spectinomycin: 128.0
Cefixime: 0.002 to 2.0
Ceftriaxone: 0.001 to 2.0
Ciprofloxacin: 0.001 to 16.0
Azithromycin: 0.008 to 16.0
Optional agent: Cefpodoxime: 0.008 to 8.0
Retest criteria:
If the MICs of strains are not determined at the highest concentration of agent tested, the MIC should be retested for susceptibility to a higher range of two-fold dilutions. An endpoint must be
determined. See below for specific repeat testing criteria.
Preparation of Antibiotic-Containing Media
Difco GC medium base (Becton Dickinson, Cockeysville, MD) supplemented with 1% IsoVitaleX (or an equivalent supplement) is used.
- Prepare the required volume of GC base medium in single strength according to the manufacturer’s directions.
- Autoclave the medium at 121 C for 15 min. Cool to 50 C in a waterbath.
- Reconstitute the dehydrated IsoVitaleX with the provided diluent according to the manufacturer’s directions.
- Add 10 ml of supplement per liter of base medium i.e., 1% (v/v); mix thoroughly. This medium is GCS medium.
- Dispense the required volume of medium into individual containers for the addition of antimicrobial solutions. Maintain media at 50 C in a waterbath.
- Prepare the working solutions and dilutions of antimicrobial agents from the stock solutions or standard powder.
Note: It is important that no longer than 1 hour elapse between the time that the stock solution is thawed, the dilutions are prepared and added to the base medium and the plates are poured. - Add the required volumes of the prepared working solutions and dilutions of the antimicrobial agents to the respective bottles of GCS medium, mix thoroughly and dispense into clearly labeled plates. Thorough mixing of the antibiotics in the medium can be accomplished by swirling the contents three times in a clockwise and counterclockwise motion followed by inverting the bottle three times, minimizing bubble formation.
- Allow the plates to cool. Invert the plates and store them in sealed plastic bags at 4 C to 8 C for no longer than two weeks prior to use.
Agar-Dilution Susceptibility Test Procedure
- Grow pure cultures of isolates to be tested on chocolate agar at 35 C to 36.5 C for 16 to 18 h in a carbon dioxide-enriched (5%) atmosphere. Use pure cultures on chocolate agar to prepare the inoculum. Do not use the first subculture from a frozen culture; subculture these isolates again before using to prepare the inoculum. Do not use isolates grown on antibiotic-containing media to prepare the inoculum. The complete set of control strains provided by the CDC should be included in each run. If two or more sets of plates (prepared in the same batch) are being inoculated on the same day, it is not necessary to inoculate the complete set of control strains on each set of plates. Two or three control strains may be inoculated on each set of plates, but all control strains should be included in each day’s test runs.
- Use a cotton applicator or a bacteriologic loop to suspend isolated colonies (or cells from less dense areas of growth on the plate) in approximately 2 ml of Mueller-Hinton (MH) broth. (The exact volume of broth required will depend on the method for measuring the turbidity of the suspension. If a spectrophotometer is used, the volume of broth must be sufficient to completely cover the light path and will vary according to the type of spectrophotometer).
- Adjust the density of the suspension to contain 108 colony forming units (CFU)/ml by comparison with a 0.5 McFarland BaSO4 turbidity standard. (If a spectrophotometer is used to measure the optical density of the suspensions, set the wavelength at 600 nm or 625 nm. Adjust the turbidity or optical density to approximately 0.08-0.13. It may be necessary to dilute this suspension further than indicated in step #4. Determine the viable count for the suspension and either adjust the initial optical density to which the suspension is prepared or the dilution to give a final viable count of 107 CFU/ml as indicated in step #4.).)
- Dilute this suspension 1:10 in MH (or equivalent) broth to give 107 CFU/ml.
- Dispense an equal volume of each suspension into wells of a replicating device, e.g., Steer’s or Cathra replicators. These replicating devices deliver 0.001 – 0.005 ml of the bacterial suspension to the surface of the medium, i.e., 104 CFU.
- Inoculate each plate of the set of antibiotic containing media plus a plate of GCS (as a control to determine that all isolates grew). You may also wish to inoculated a GCS plate between each set of antibiotic-containing medium to ensure against carry-over of antimicrobial agents from one medium to another; these plates also allow for monitoring for contamination of the inocula during the inoculation process.Note. The time elapsing between the preparation of the strain suspensions and inoculation of the plates should not exceed 1 h.
- Allow the inoculated plates to air-dry at room temperature for approximately 15 min. Invert the plates and incubate at 35 C to 36.5 C in a CO2-enriched (5%) atmosphere for 24 h.
- Examine the plates for growth. Use a separate sheet to record the results for each antibiotic tested and record the growth for each isolate on each antibiotic concentration tested. Record the growth as good (+), poor (±), or no growth (-). By using this scheme, the results can be reviewed at a later date for transcription errors and, when isolates grow on the highest concentration tested, the degree of growth will indicate whether the isolate is growing strongly or is partially inhibited.
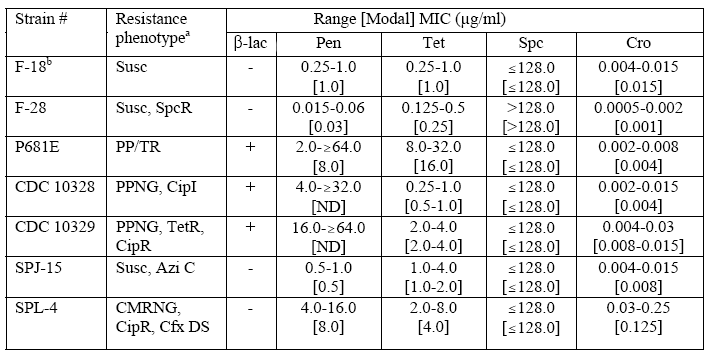
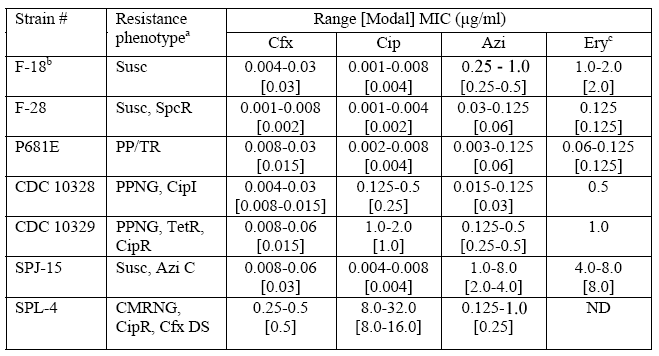
Agar Dilution Susceptibility Testing–CDC Reference Strains of Neisseria gonorrhoeae
Abbreviations: ß-lac, ß-lactamase; Pen, penicillin; Tet, tetracycline; Spc, spectinomycin; Cro, ceftriaxone; Cfx, cefixime; Cip, ciprofloxacin; Azi, azithromycin; Susc, susceptible to penicillin and tetracycline (MICs <2.0 µg/ml); SpcR, spectinomycin-resistant (MIC >128.0 µg/ml); PP/TR, ß-lactamase-positive strains with MICs ≥16.0 µg/ml of tetracycline; PPNG, penicillinase (ß-lactamase)-producing N. gonorrhoeae; TetR, MIC <2.0 µg/ml of penicillin and MIC ≥2.0 µg/ml of tetracycline; CipI, strain with MIC of 0.125-0.5 µg/ml of ciprofloxacin; CipR, strain with MIC of ≥1.0 µg/ml of ciprofloxacin; Azi C, isolates exhibiting critical MICs (MIC ≥1.0 µg/ml) of azithromycin; CMRNG, ß-lactamase-negative and MIC >2.0 µg/ml of penicillin and MIC ≥2.0 to 8.0 µg/ml of tetracycline; Cfx DS, isolates exhibiting decreased susceptibility (MIC ≥0.5 µg/ml) to cefixime; ND, Not Determined.
aResistance phenotypes are composed of resistance phenotypes for penicillin and tetracycline according to GISP definitions supplemented with specific designations for spectinomycin, ciprofloxacin, azithromycin, and cefixime.
bATCC49226, NCCLS-recommended quality control strain for N. gonorrhoeae; MIC ranges are those published by NCCLS.
cErythromycin range and modal values were calculated from fewer results than range and modal values for the other antimicrobial agents.
Isolates with Alert Value MICs
Isolates with the following MICs require confirmation by retesting:
Ceftriaxone: MIC ≥ 0.125 µg/ml
Cefixime: MIC ≥ 0.25 µg/ml
For ceftriaxone and cefixime, isolates should be tested for growth on medium containing the antibiotics at concentrations ranging from two dilutions below the initial MIC to sufficient concentrations above the initial MIC to obtain an endpoint MIC.
Ciprofloxacin: MIC ≥ 1.0 µg/ml
For ciprofloxacin, isolates should be tested for growth on medium containing two-fold dilutions of ciprofloxacin up to, and including, a concentration of ciprofloxacin that will provide an endpoint MIC.
Azithromycin: MIC ≥ 2.0 µg/ml
For azithromycin, isolates should be tested for growth on medium containing azithromycin at concentrations ranging from two dilutions below the initial MIC to sufficient concentrations above the initial MIC to obtain an endpoint MIC.
Spectinomycin: MIC ≥ 128 µg/ml
For spectinomycin, isolates should be retested for growth on 128.0 µg/ml of spectinomycin.
The GISP Coordinator and Manager in ESB, DSTDP, NCHSTP, CDC should be notified within one working day by telephone or by e-mail of any isolate(s) identified to have a high MIC; the GISP Coordinator or Manager will then immediately communicate the results to the Sentinel Site and the pertinent local and/or state STD programs. Such isolates also should be shipped rapidly in triplicate to CASPIR, CDC; laboratory personnel should not wait until the request lists arrive from CDC to ship these isolates. A sheet marked “Alert Value Isolate Packing Form” that contains the original and the retest MIC values for each isolate should be included with the isolate shipment; a copy of this same sheet should also be sent separately to the GISP Coordinator.
Isolate Preservation: All isolates should be suspended in trypticase soy broth containing 20% (v/v) glycerol and frozen at -70 C in duplicate at the Regional Laboratories. When isolates are requested by CDC for further characterization, triplicate copies of each requested isolate should be shipped to CASPIR, CDC. A copy of each shipped isolate must be maintained at the Regional Laboratory until notified by the GISP Coordinator or GISP Manager that the isolates have been received. This will generally require maintenance of isolates from the current year and the previous year.