Vaccine Adverse Events
This information is about preventing or responding to smallpox. For the latest information about the current mpox outbreak, including information on symptoms, prevention, and vaccines, please visit CDC’s Mpox site.
The smallpox vaccine is generally safe and effective, but some people do experience side effects and adverse reactions. Severe adverse reactions are more common in people who are being vaccinated for the first time and among young children (<5 years of age).
CDC has published criteria to use for surveillance case definition and classification for smallpox vaccine (vaccinia) adverse reactions. CDC also provides consultation for clinicians to help diagnose and manage patients with suspected vaccinia virus vaccine adverse reactions. Clinicians may reach CDC medical staff by calling the Emergency Operations Center at 770-488-7100.
Localized Reactions
Superinfection of the vaccination site or regional lymph nodes
Vaccination progression and normal local reactions are difficult to distinguish from a superinfection of the vaccination site or regional lymph nodes. Secondary infections of the vaccination site are uncommon and are typically mild to moderate in clinical severity. Children and individuals who frequently manipulate and contaminate the vaccination site are at greatest risk.
Robust take
A robust take is a vaccinial cellulitis and is defined as >3 inches of redness with swelling, pain, and warmth at the vaccination site. The symptoms peak 6 to 12 days after vaccination and regress within the following 24 to 72 hours.
Unintentional Transfer of Vaccinia Virus
Inadvertent autoinoculation
Inadvertent autoinoculation is the unintentional transfer of vaccinia virus from the vaccination site to another place on the vaccinee’s body. Vaccinees can transfer vaccinia to their hands or fomites. The most common sites are the eye and surrounding orbit (ocular vaccinia), followed by the face, nose, mouth, lips, genitalia, and anus.
Contact transmission
A vaccinee can also spread vaccinia virus from the vaccination site (or other lesions distant from the vaccination site) to close contacts through direct contact or through other vectors such as clothing, bedding, or bandages contaminated by vaccinia virus. Viral shedding can occur until the scab detaches from the vaccination site (or any distant lesions), revealing healthy skin underneath. Infection through contact transmission can result in the same adverse events observed after smallpox vaccination.
Ocular vaccinia
There are different forms of ocular vaccinia: blepharitis (inflammation of the eyelid), conjunctivitis, keratitis (inflammation of the cornea, including epithelial and stromal forms), iritis, or combinations thereof. Infections can be clinically mild to severe and can lead to vision loss.
Diffuse Dermatologic Complications
There are two groups of diffuse dermatological complications: those thought to be free of vaccinia virus (e.g., erythema multiforme minor, Stevens-Johnson syndrome, and other nonspecific post-vaccination rashes), and those thought to be caused by replicating vaccinia virus at the site of the skin lesions. This section covers the second group.
Generalized vaccinia
Generalized vaccinia is a disseminated vesicular or pustular rash and is usually benign and self-limited among immunocompetent hosts. Modern case definitions call for the presence of lesions removed from the initial vaccination site and laboratory evidence supporting the diagnosis of vaccinia virus infection and/or excluding other causes of papular or vesicular eruptions. First-time vaccinees are at higher risk for generalized vaccinia than revaccinees. Generalized vaccinia is often more severe among persons with underlying immunodeficiency who might have been inadvertently vaccinated.
Eczema vaccinatum
Eczema vaccinatum is a localized or systemic spread of vaccinia virus. It occurs most often in vaccine recipients who have a history of atopic dermatitis. The rash is often accompanied by fever and lymphadenopathy, and affected persons are frequently systemically ill. Eczema vaccinatum tends to be most severe among first-time vaccinees, unvaccinated close contacts of vaccinees, and young children. It can be fatal.
Progressive Vaccinia
Progressive vaccinia is rare, severe, and often fatal. It occurs when a vaccination site fails to heal and vaccinia virus replication persists. The skin surrounding the vaccination site becomes infected with vaccinia virus, and secondary metastatic vaccinia lesions can occur. Lesions can appear necrotic, fungated, piled-up, or well demarcated. Concomitant bacterial superinfection also can occur. Progressive vaccinia typically occurs in persons with an underlying humoral or cellular immune deficit.
Rare Adverse Reactions
Fetal vaccinia
Rarely, smallpox vaccination of a pregnant woman can result in fetal vaccinia. Transmission to the fetus can occur any time during pregnancy. Miscarriage, stillbirth, or live birth (usually premature, followed by death) or birth of a surviving but pox-scarred infant can occur after the mother’s exposure to vaccinia.
Postvaccinial central nervous system disease
Postvaccinial central nervous system disease such as postvaccinial encephalitis or encephalomyelitis may occur. It is most common among infants aged less than 12 months. Clinical symptoms reflect cerebral or cerebellar dysfunction with headache, fever, vomiting, altered mental status, lethargy, seizures, and coma. No clinical criteria, radiologic findings, or laboratory tests that are diagnostic for these adverse reactions exist.
Cardiac Adverse Events
Myo/pericarditis
Reports of inflammatory disease of the myocardium, pericardium, or both after smallpox vaccination suggest an association between smallpox vaccination with the New York City Board of Health vaccinia strain and myo/pericarditis. Clinical presentation may include chest pain, dyspnea, and palpitations that range from subtle to severe. The case definition found in the Surveillance Guidelines for Smallpox Vaccine (vaccinia) Adverse Reactions provides more information about the spectrum of abnormalities found in inflammatory heart disease.
Dilated cardiomyopathy and cardiac ischemia
Dilated cardiomyopathy and cardiac ischemia have been noted in temporal association to smallpox vaccination, but have not been demonstrated to be linked etiologically. Dilated cardiomyopathy is a known sequelae of viral myocarditis and can present weeks to months after acute infection. The causal relation between smallpox vaccination and dilated cardiomyopathy is unclear, but is biologically plausible.
Smallpox Vaccination and Adverse Reactions: Guidance for Clinicians, 2003 Source: MMWR 2003, 52(RR04);1-28. (Note: This guidance has references to Dryvax®, which is no longer available. However, guidance is relevant for all U.S. vaccine formulations that contain the New York City Board of Health vaccinia strain which includes ACAM2000® and APSV.)
Surveillance Guidelines for Smallpox Vaccine (vaccinia) Adverse Reactions, 2006 Source: MMWR 2006, 55(RR01);1-16.
See Examples of Autoinoculation Sites and Adverse Reactions to the Smallpox Vaccine
Report all serious or unusual adverse events, including inadvertent inoculation, to the Vaccine Adverse Events Reporting System (VAERS). Record cases of inadvertent inoculation of a contact under the contact’s name. Include information on the vaccinee as the person who infected the contact in the notes in section 7.

A patient’s arm showing a robust take. The patient’s vaccination site is surrounded by a large area of erythema and swelling, and shows lymphangitic streaking. Source: CDC
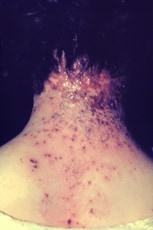
This photograph displays the nape of a neck of a woman with eczema vaccinatum. This patient had a history of eczema. Source: CDC.
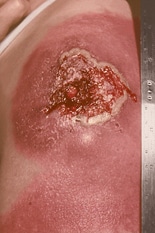
The initial vaccination site of a patient with progressive vaccinia. Source: CDC/Dr. Clement R. Boughton.