Histoplasma in the Environment
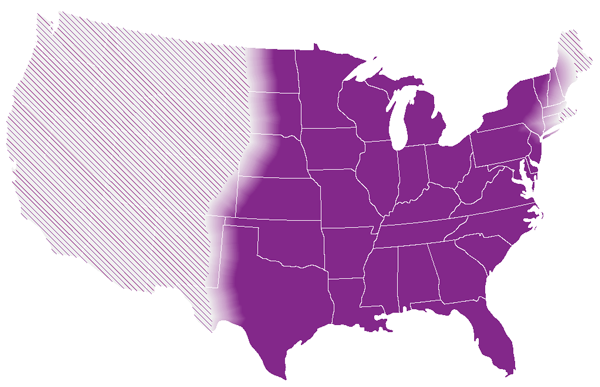
This map shows CDC’s current estimate of where the fungi that cause histoplasmosis live in the environment in the United States. These fungi are not distributed evenly in the shaded areas, might not be present everywhere in the shaded areas, and can also be outside the shaded areas. Darker shading shows areas where Histoplasma is more likely to live. Diagonal shading shows the potential range of Histoplasma.
Histoplasma, the fungus that causes histoplasmosis, lives throughout the world, but it’s most common in North America and Central America.1 In the United States, Histoplasma mainly lives in soil in the central and eastern states, particularly areas around the Ohio and Mississippi River Valleys, but it can likely live in other parts of the country as well, especially if the environmental conditions are highly suitable. In the environment, Histoplasma is undetectable to the naked eye. Learn more about the approximate areas with Histoplasma.
Histoplasma grows especially well in soil or other environmental material containing large amounts of bird or bat droppings. Bird and bat droppings act as a source for the growth of Histoplasma already present in soil.2 Fresh bird droppings on surfaces such as sidewalks and windowsills likely does not pose a risk for histoplasmosis because birds themselves are rarely infected with Histoplasma. Unlike birds, bats can become infected with Histoplasma and may be able to excrete it in their droppings.3
A large number of birds or bats, even without obvious accumulations of droppings, can also pose a risk for Histoplasma exposure. The types of birds typically associated with histoplasmosis outbreaks include chickens, blackbirds (starlings and grackles), pigeons, and gulls.4,5 Presence of birds, bats, or large accumulations of their droppings is not required for Histoplasma to grow, and many patients with histoplasmosis do not have such exposures.6
Histoplasma has been detected in some organic fertilizers in Latin America, but more studies are needed to understand whether the fungus can survive commercial fertilizer manufacturing processes.7,8
For any accumulations of bird or bat droppings, soil, or other environmental material suspected to be contaminated with Histoplasma, the safest approach is to assume the material is contaminated, take exposure precautions, and remediate it accordingly.
Detecting Histoplasma in environmental samples can be challenging. Routine environmental sampling for Histoplasma is not recommended, although research is needed to evaluate potential targeted testing strategies. Focused polymerase chain reaction (PCR) or culture testing can help confirm a suspected environmental source during a histoplasmosis outbreak. In outbreak settings without a clear exposure, large-scale environmental testing is unlikely to be useful. In outbreak settings with a very clear exposure, environmental testing may not be necessary, particularly if the results would not change public health recommendations to prevent future cases. Also, an environmental sample that tests negative for Histoplasma does not necessarily mean that the fungus is not present or was not present at the time the exposure occurred.
- Ashraf N, Kubat RC, Poplin V, Adenis AA, Denning DW, Wright L, McCotter O, Schwartz IS, Jackson BR, Chiller T, Bahr NC [2020]. Re-drawing the maps for endemic mycoses. Mycopathologia 185(5):843–865.
- Ajello L [1964]. Relationship of Histoplasma capsulatum to avian habitats. Public health reports 79:266–270.
- Hoff GL, Bigler WJ [1981]. The role of bats in the propagation and spread of histoplasmosis: a review. J Journal of Wildlife Diseases 17(2):191–196.
- Benedict K, Mody RK [2016]. Epidemiology of histoplasmosis outbreaks, United States, 1938-2013. Emerg Infect Dis 22(3):370–378.
- Waldman RJ, England AC, Tauxe R, Kline T, Weeks RJ, Ajello L, Kaufman L, Wentworth B, Fraser DW [1983].A winter outbreak of acute histoplasmosis in northern Michigan. Am J Epidemiol 117(1):68–75.
- Benedict K, McCracken S, Signs K, Ireland M, Amburgey V, Serrano JA, Christophe N, Gibbons-Burgener S, Hallyburton S, Warren KA, Keyser Metobo A, Odom R, Groenewold MR, Jackson BR [2020]. Enhanced surveillance for histoplasmosis—nine states, 2018–2019. Open Forum Infect Dis 7(9):ofaa343.
- Gómez LF, Torres IP, Jiménez-A MDP, McEwen JG, de Bedout C, Peláez CA, Acevedo JM, Taylor ML, Arango M [2018]. Detection of Histoplasma capsulatum in organic fertilizers by Hc100 nested polymerase chain reaction and its correlation with the physicochemical and microbiological characteristics of the samples. Am J Trop Med Hyg 98(5):1303–1312.
- Londono LFG, Leon LCP, Ochoa JGM, Rodriguez AZ, Jaramillo CAP, Ruiz JMA, Taylor ML, Arteaga MA, Alzate MDJ [2019]. Capacity of Histoplasma capsulatum to survive the composting process. Applied and Environmental Soil Science 2019.
