For Healthcare Professionals
Varicella (chickenpox) is an acute infectious disease. It is caused by varicella-zoster virus (VZV), which is a DNA virus that is a member of the herpesvirus group. After the primary infection, VZV stays in the body (in the sensory nerve ganglia) as a latent infection. Primary infection with VZV causes varicella. Reactivation of latent infection causes herpes zoster (shingles).
- Clinical Features
- Transmission
- Complications from Varicella
- People at High Risk for Severe Varicella
- Managing People at High Risk for Severe Varicella
- Assessing Immunity to Varicella
- Preventing Varicella in Healthcare Settings
- Management of Patients with Varicella
- Impact of the U.S. Varicella Vaccination Program
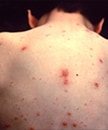
Characteristic pancorporeal varicella lesions in an unvaccinated person.
Incubation Period and Prodrome
The average incubation period for varicella is 14 to 16 days after exposure to a varicella or a herpes zoster rash, with a range of 10 to 21 days. A mild prodrome of fever and malaise may occur 1 to 2 days before rash onset, particularly in adults. In children, the rash is often the first sign of disease.
Varicella in Unvaccinated Persons
The rash is generalized and pruritic. It progresses rapidly from macular to papular to vesicular lesions before crusting. Lesions are typically present in all stages of development at the same time. The rash usually appears first on the chest, back, and face, then spreads over the entire body. The lesions are usually most concentrated on the chest and back. Symptoms typically last 4 to 7 days.
In healthy children, varicella is generally mild, with an itchy rash, malaise, and temperature up to 102°F for 2 to 3 days. Infants, adolescents, adults, pregnant women, and immunocompromised people are at risk for more severe disease and have a higher incidence of complications. Recovery from primary varicella infection usually provides immunity for life. In otherwise healthy people, a second occurrence of varicella is uncommon. Second occurrence of varicella may be more likely to occur in people who are immunocompromised. As with other viral infections, re-exposure to natural (wild-type) varicella may lead to re-infection that boosts antibody titers without causing illness or detectable viremia.
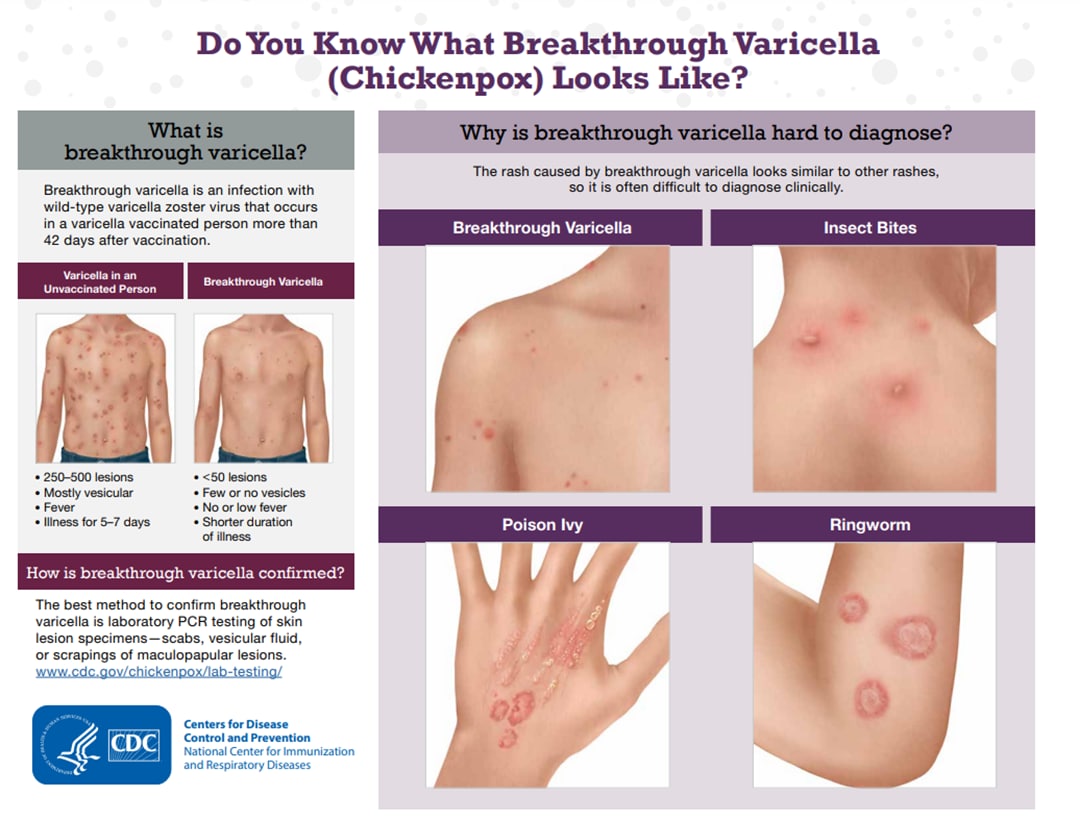
Do You Know What Breakthrough Varicella (Chickenpox) Looks Like?
An infographic that helps identify breakthrough varicella (chickenpox)
Varicella in Vaccinated Persons (Breakthrough Varicella)
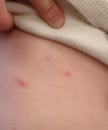
Breakthrough varicella on the abdomen of a vaccinated child.
Breakthrough varicella is infection with wild-type varicella-zoster virus (VZV) occurring in a vaccinated person more than 42 days after varicella vaccination. Breakthrough varicella is usually mild. Patients typically are afebrile or have low fever and develop fewer than 50 skin lesions. They usually have a shorter illness compared to unvaccinated people who get varicella. The rash is more likely to be predominantly maculopapular rather than vesicular. However, 25% to 30% of people vaccinated with one dose who get breakthrough varicella will have clinical features similar to unvaccinated people with varicella.
Since the clinical features of breakthrough varicella are often mild, it can be difficult to make a diagnosis on clinical presentation alone. Laboratory testing is increasingly important for confirming varicella and appropriately managing the patients and their contacts. Breakthrough varicella occurs less frequently among those who have received two doses of vaccine compared with those who have received only one dose; disease may be even milder among two-dose vaccine recipients, although the information about this is limited.
Transmission
Varicella is highly contagious. The virus can be spread from person to person by direct contact, inhalation of aerosols from vesicular fluid of skin lesions of acute varicella or zoster, and possibly through infected respiratory secretions that also may be aerosolized. A person with varicella is considered contagious beginning one to two days before rash onset until all the chickenpox lesions have crusted. Vaccinated people may develop lesions that do not crust. These people are considered contagious until no new lesions have appeared for 24 hours.
It takes from 10 to 21 days after exposure to the virus for someone to develop varicella. Based on studies of transmission among household members, about 90% of susceptible close contacts will get varicella after exposure to a person with disease. Although limited data are available to assess the risk of VZV transmission from zoster, one household study found that the risk for VZV transmission from herpes zoster was approximately 20% of the risk for transmission from varicella.
People with breakthrough varicella are also contagious. One study of varicella transmission in household settings found that people with mild breakthrough varicella (<50 lesions) who were vaccinated with one dose of varicella vaccine were one-third as contagious as unvaccinated people with varicella. However, people with breakthrough varicella with 50 or more lesions were just as contagious as unvaccinated people with the disease.
Varicella is less contagious than measles, but more contagious than mumps and rubella.
Complications
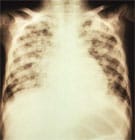
X-ray of pneumonia caused by varicella.
The most common complications from varicella are:
- In children: Bacterial infections of the skin and soft tissues
- In adults: Pneumonia
Severe complications caused by the virus include cerebellar ataxia, encephalitis, viral pneumonia, and hemorrhagic conditions. Other severe complications are due to bacterial infections and include:
- Septicemia
- Toxic shock syndrome
- Necrotizing fasciitis
- Osteomyelitis
- Bacterial pneumonia
- Septic arthritis
People at High Risk for Severe Varicella
People at risk for severe varicella include:
- Immunocompromised people without evidence of immunity to varicella, such as:
- People with leukemia or lymphoma
- People on medications that suppress the immune system, such as high-dose systemic steroids or chemotherapeutic agents
- People with cellular immune-deficiencies or other immune system problems
- Newborns whose mothers have varicella from five days before to two days after delivery
- Premature babies exposed to varicella or herpes zoster, specifically:
- Hospitalized premature infants born at ≥28 weeks of gestation whose mothers do not have evidence of immunity
- Hospitalized premature infants born at <28 weeks of gestation or who weigh ≤1,000 grams at birth regardless of their mothers’ varicella immunity status
- Pregnant women without evidence of immunity to varicella
Immunocompromised People
Immunocompromised people who get varicella are at risk of developing visceral dissemination (VZV infection of internal organs) leading to pneumonia, hepatitis, encephalitis, and disseminated intravascular coagulopathy. They can have an atypical varicella rash with more lesions, and they can be sick longer than immunocompetent people who get varicella. New lesions may continue to develop for more than 7 days, may appear on the palms and soles, and may be hemorrhagic.
People with HIV or AIDS
Children with HIV infection tend to have atypical rash with new crops of lesions presenting for weeks or months. The lesions may initially be typical maculopapular vesicular but can later develop into non-healing ulcers that become necrotic, crusted, and hyperkeratotic. This is more likely to occur in HIV-infected children with low CD4 counts.
Some studies have found that VZV dissemination to the visceral organs is less common in children with HIV than in other immunocompromised people with VZV infection. The rate of complications may also be lower in HIV-infected children on antiretroviral therapy or HIV-infected people with higher CD4 counts at the time of varicella infection. Retinitis can occur among HIV-infected children and adolescents.
Most adults, including those who are HIV-positive, have already had varicella and are VZV seropositive. As a result, varicella is relatively uncommon among HIV-infected adults.
For more information about vaccinating immunocompromised people, including some groups with HIV-infection, see Special Considerations for Vaccination (Vaccination of HIV Infected Persons) in Prevention of Varicella: Recommendations of the Advisory Committee on Immunization Practices (ACIP).
Pregnant Women
Pregnant women who get varicella are at risk for serious complications, primarily pneumonia, and in some cases, may die as a result of varicella. Some studies have suggested that both the frequency and severity of VZV pneumonia are higher when varicella is acquired during the third trimester, although other studies have not supported this observation.
If a pregnant woman gets varicella in her first or early second trimester, her baby has a small risk (0.4 to 2.0%) of being born with congenital varicella syndrome. The baby may have scarring on the skin; abnormalities in limbs, brain, and eyes, and low birth weight.
If a woman develops varicella rash from 5 days before to 2 days after delivery, the newborn will be at risk for neonatal varicella. Historically, the mortality rate for neonatal varicella was reported to be about 30%, but the availability of VZV immune globulin and intensive supportive care have reduced the mortality to about 7%.
The vaccine is contraindicated for pregnant women. See Guidelines for Vaccinating Pregnant Women: Varicella.
Managing People at High Risk for Severe Varicella
Varicella-Zoster Immune Globulin
For people exposed to varicella or herpes zoster who cannot receive varicella vaccine, varicella-zoster immune globulin can prevent varicella from developing or lessen the severity of the disease. Varicella-zoster immune globulin is recommended for people who cannot receive the vaccine and 1) who lack evidence of immunity to varicella, 2) whose exposure is likely to result in infection, and 3) are at high risk for severe varicella.
The varicella-zoster immune globulin product licensed for use in the United States is VariZIG™. VariZIG should be given as soon as possible after exposure to VZV; it can be given within 10 days of exposure. For more information on the recommendations for VariZIG use, see the Morbidity and Mortality Weekly Report article on Updated Recommendations for Use of VariZIG — United States, 2013. VariZIG is commercially available from a broad network of specialty distributors in the United States (list available at www.varizig.com).
Acyclovir Treatment
The American Academy of Pediatrics (AAP) recommends that certain groups at increased risk for moderate to severe varicella be considered for oral acyclovir or valacyclovir treatment. These high risk groups include:
- Healthy people older than 12 years of age
- People with chronic cutaneous or pulmonary disorders
- People receiving long-term salicylate therapy
- People receiving short, intermittent, or aerosolized courses of corticosteroids
Some healthcare providers may elect to use oral acyclovir or valacyclovir for secondary cases within a household. For maximum benefit, oral acyclovir or valacyclovir therapy should be given within the first 24 hours after the varicella rash starts.
Oral acyclovir or valacyclovir therapy is not recommended by AAP for use in otherwise healthy children experiencing typical varicella without complications. Acyclovir is a category B drug based on the U.S. Food and Drug Administration’s Drug Risk Classification in pregnancy. Some experts recommend oral acyclovir or valacyclovir for pregnant women with varicella, especially during the second and third trimesters. Intravenous acyclovir is recommended for the pregnant patient with serious, viral-mediated complications of varicella, such as pneumonia.
Intravenous acyclovir therapy is recommended for severe disease (e.g., disseminated VZV such as pneumonia, encephalitis, thrombocytopenia, severe hepatitis) and for varicella in immunocompromised patients (including patients being treated with high-dose corticosteroid therapy for >14 days).
Famciclovir is available for treatment of VZV infections in adults, but its efficacy and safety have not been established for children. In cases of infections caused by acyclovir-resistant VZV strains, which usually occur in immunocompromised people, Foscarnet should be used to treat the VZV infection, but consultation with an infectious disease specialist is recommended.
Assessing Immunity to Varicella
Two doses of varicella vaccine are recommended for all children, adolescents, and adults without evidence of immunity to varicella. Those who previously received one dose of varicella vaccine should receive their second dose for best protection against the disease.
Evidence of immunity to varicella includes any of the following:
- Documentation of age-appropriate varicella vaccination
- Preschool-age children (i.e., age 12 months through 3 years): one dose
- School-age children, adolescents, and adults: two doses
- Laboratory evidence of immunity or laboratory confirmation of disease*
- Birth in the United States before 1980 (should not be considered evidence of immunity for healthcare personnel, pregnant women, and immunocompromised people)
- Diagnosis or verification of a history of varicella or herpes zoster by a healthcare provider
*Commercial assays can be used to assess disease-induced immunity, but they lack sensitivity to detect vaccine-induced immunity (i.e., they might yield false-negative results).
To verify a history of varicella, healthcare providers should inquire about:
- An epidemiologic link to another typical varicella case or to a laboratory confirmed case, or
- Evidence of laboratory confirmation, if testing was performed at the time of acute disease
People who have neither an epidemiologic link nor laboratory confirmation of varicella should not be considered as having a valid history of disease. For these people, a second dose of vaccine is recommended if they previously received only one dose. If a healthcare provider verifies the diagnosis based on the above criteria, then vaccination is not needed.
Routine testing for varicella immunity after two doses of vaccine is not recommended. Available commercial assays are not sensitive enough to detect antibodies after vaccination in all instances. Documented receipt of two doses of varicella vaccine supersedes results of subsequent serologic testing.
Preventing Varicella in Healthcare Settings
Nosocomial Transmission of VZV
Nosocomial transmission of VZV is well-recognized and can be life threatening to certain groups of patients. Reports of nosocomial transmission are uncommon in the United States since introduction of varicella vaccine.
Patients, healthcare providers, and visitors with varicella or herpes zoster can spread VZV to susceptible patients and healthcare providers in hospitals, long-term-care facilities, and other healthcare settings. In healthcare settings, transmissions have been attributed to delays in the diagnosis or reporting of varicella and herpes zoster and failures to implement control measures promptly.
Although all susceptible patients in healthcare settings are at risk for severe varicella and complications, certain patients without evidence of immunity are at increased risk:
- Premature infants born to susceptible mothers
- Infants born at less than 28 weeks gestation or who weigh ≤1000 grams, regardless of maternal immune status
- Immunocompromised people, including those who are undergoing immunosuppressive therapy, have malignant disease, or are immunodeficient
- Pregnant women
Management of Patients with Varicella
Healthcare providers should follow standard precautions plus airborne precautions (negative air-flow rooms) and contact precautions until lesions are dry and crusted. If negative air-flow rooms are not available, patients with varicella should be isolated in closed rooms with no contact with people without evidence of immunity. Patients with varicella should be cared for by staff with evidence of immunity.
For more information, see:
- Preventing Varicella-Zoster Virus (VZV) Transmission from Zoster in Healthcare Settings
- References and Resources
Impact of the U.S. Varicella Vaccination Program
Varicella used to be very common in the United States and contributed significantly to the burden of childhood disease. In the early 1990s each year, more than 4 million people got varicella,10,500 to 13,500 were hospitalized, and 100 to 150 died. More than 90% of cases, 70% of hospitalizations, and about half of the deaths occurred in children.
Since the start of the U.S. varicella vaccination program in 1995, chickenpox cases have declined overall by more than 97%. The program dramatically decreased virus circulation and increased community protection. Disease declined in all age groups, including infants who are not recommended to receive the vaccine because of their age and adults who are not routinely vaccinated and who can experience a more severe illness when infected.
Main Findings from 1995-2019
- Varicella hospitalizations and deaths declined 94% and 97%, respectively, among people 50 years old and younger.
- The greatest decline in cases was among people 20 years old and younger who were born during the program, with 97% drop in hospitalizations and more than 99% drop in deaths.
- The risk of shingles is significantly lower among vaccinated children, including those who are immunocompromised.
- There is approximately an 80% lower risk of shingles among healthy vaccinated children compared to unvaccinated children who had wild-type varicella.
- The varicella vaccination is an effective and cost-saving public health intervention, both in terms of disease prevention and dollars saved. During the first 25 years:
- The U.S varicella vaccination program prevented an estimated 91 million cases, 238,000 hospitalizations, 1.1 million hospitalization days, and almost 2,000 deaths; and saved 118,000 life-years, at net societal savings of $23.4 billion.
- For every $1 spent, the estimated return on investment is $1.70; however, the benefits of the investment will continue to accrue beyond 2020.
Latest publication: The Varicella Vaccination Program in the United States: 25 Years of Saving Lives and Preventing Illness | Journal of Infectious Diseases | November 1, 2022.
Infographic: Chickenpox Vaccine Saves Lives Infographic

- Materials for Talking to Parents about Vaccines
- Varicella Vaccination: Recommendations for Healthcare Personnel
- Prevention of Varicella: Recommendations of the Advisory Committee on Immunization Practices (ACIP)
- Varicella-Zoster Virus Infections In Redbook: 2018 Report of the Committee on Infectious Diseases American Academy of Pediatrics
- Updated Recommendations for Use of VariZIG — United States, 2013. MMWR 2013;62:574-576
- Varicella and Breakthrough Varicella: To Test or Not to Test [2.07MB, 14 slides] MS PowerPoint® file available upon email request
