Critical Congenital Heart Defects Screening Methods

Newborn screening for critical congenital heart defects (critical CHDs) can identify newborns with these conditions before signs or symptoms are evident and before the newborns are discharged from the birth hospital.
Current published recommendations focus on screening newborns in the well-baby nursery and intermediate care nurseries or other units in which discharge from the hospital is common during a newborn’s first week. Timing the screening around the time of the newborn hearing screening can help improve efficiency. A pulse oximeter is used to measure the percentage of hemoglobin in the blood that is saturated with oxygen.
The following algorithm has been developed to show the steps in screening (Kemper et al., 2011)
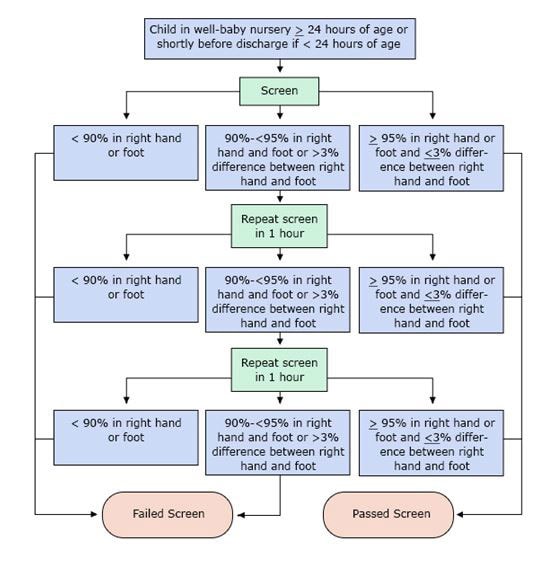
PLEASE NOTE: Percentages refer to oxygen saturation as measured by pulse oximeter.
Pulse oximetry screening should not replace taking a complete family health history and pregnancy history or completing a physical examination, which sometimes can detect a critical CHD before the development of low levels of oxygen (hypoxemia) in the blood.
Screening with pulse oximetry can identify a number of types of critical CHDs, the most common of which are shown in the box below. While not the main targets of screening, many conditions other than critical CHD may present with hypoxemia and thus likewise be detected via pulse oximetry.
- Coarctation of the aorta
- Double outlet right ventricle
- Ebstein anomaly
- Hypoplastic left heart syndrome
- Interrupted aortic arch
- Pulmonary atresia
- Single ventricle
- Tetralogy of Fallot
- Total anomalous pulmonary venous return
- d-Transposition of the great arteries
- Tricuspid artresia
- Truncus ateriosus
- Other critical CHDs requiring treatment in the first year of life
- Hemoglobinopathy
- Hypothermia
- Infection, including sepsis
- Lung disease (congenital or acquired)
- Non-critical congenital heart defect
- Persistent pulmonary hypertension
- Other hypoxic conditions not otherwise specified
Failed Screens
A screen is considered failed if:
- Any oxygen saturation measure is <90% (in the initial screen or in repeat screens),
- Oxygen saturation is <95% in the right hand and foot on three measures, each separated by one hour, or
- A >3% absolute difference exists in oxygen saturation between the right hand and foot on three measures, each separated by one hour.
Any infant who fails the screen should have an evaluation for causes of hypoxemia. In most cases this will include an echocardiogram, but if a reversible cause of hypoxemia is identified and appropriately treated, an echocardiogram may not be necessary. The infant’s pediatrician should be notified immediately and the infant might need to be seen by a cardiologist.
Passed Screens
Any screening with an oxygen saturation measure that is ≥95% in the right hand or foot with a ≤3% absolute difference between the right hand or foot is considered a passed screen and screening would end. Pulse oximetry screening does not detect all critical CHDs, so it is possible for a baby with a passing screening result to still have a critical CHD or other CHD.
Ways to Reduce False Positive Screens
- Screen the newborn while he or she is alert.
- Screen the newborn when he or she is at least 24 hours old.
Other Tools or Resources
American Academy of Pediatrics: Critical Congenital Heart Defect (CCHD) Screening Resource for Primary Care Providers This online resource was developed by doctors for doctors, to help primary care providers navigate the implementation process and provide effective, long-term medical homes for babies with critical CHDs.
American Academy of Pediatrics: Endorsement of Health and Human Services Recommendation for Pulse Oximetry Screening for Critical Congenital Heart Disease [Read article ]
Children’s National Medical Center’s Congenital Heart Disease Screening Program has created videos about critical CHD screening for parents and healthcare professionals.
Congenital Heart Public Health Consortium (CHPHC) The CHPHC is a group of organizations uniting resources and efforts in public health activities to prevent congenital heart defects and improve outcomes for affected children and adults. Their website provides resources for families and providers on heart defects and screening.
Kemper AR, Mahle WT, Martin GR, Cooley WC, Kumar P, Morrow WR, Kelm K, Pearson GD, Glidewell J, Grosse SD, Lloyd-Puryear M, Howell RR. Strategies for implementing screening for critical congenital heart disease. Pediatrics. 2011; 128:e1-8. [Read article]
Knapp AA, Metterville DR, Kemper AR, Prosser L, Perrin JM. Evidence review: Critical congenital cyanotic heart disease. Maternal and Child Health Bureau, Health Resources and Services Administration. 2010. [Read article [524 KB / 46 pages]
Mahle WT, Newburger JW, Matherne GP, Smith FC, Hoke TR, Koppel R, Gidding SS, Beekman RH, 3rd, Grosse, SD. Role of pulse oximetry in examining newborns for congenital heart disease: A scientific statement from the AHA and AAP. Pediatrics. 2009;124:823-36. [Read article]
NewSTEPS This webpage on critical congenital heart defects provides a central location for resources related to these conditions, including webinars, legislative updates, and news.
NIH/NLM Newborn Screening Coding and Terminology Guide This guide provides the data standards for electronic reporting of critical congenital heart defects.
Thangaratinam S, Brown K, Zamora J, Khan KS, Ewer AK. Pulse oximetry screening for critical congenital heart defects in asymptomatic newborn babies: a systematic review and meta-analysis. Lancet. 2012; 379:2459-64. [Read article]
Disclaimer: Linking to a non-federal site does not constitute an endorsement of the sponsors by CDC, HHS, or any of its employees; nor does it constitute an endorsement of the information and products presented on the site.