Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Yellow Fever Vaccine: Recommendations of the Advisory Committee on Immunization Practices (ACIP)
Summary
This report updates CDC's recommendations for using yellow fever (YF) vaccine (CDC. Yellow fever vaccine: recommendations of the Advisory Committee on Immunizations Practices: MMWR 2002;51[No. RR-17]). Since the previous YF vaccine recommendations were published in 2002, new or additional information has become available on the epidemiology of YF, safety profile of the vaccine, and health regulations related to the vaccine. This report summarizes the current epidemiology of YF, describes immunogenicity and safety data for the YF vaccine, and provides recommendations for the use of YF vaccine among travelers and laboratory workers.
YF is a vectorborne disease resulting from the transmission of yellow fever virus (YFV) to a human from the bite of an infected mosquito. It is endemic to sub-Saharan Africa and tropical South America and is estimated to cause 200,000 cases of clinical disease and 30,000 deaths annually. Infection in humans is capable of producing hemorrhagic fever and is fatal in 20%--50% of persons with severe disease. Because no treatment exists for YF disease, prevention is critical to lower disease risk and mortality. A traveler's risk for acquiring YFV is determined by multiple factors, including immunization status, location of travel, season, duration of exposure, occupational and recreational activities while traveling, and local rate of virus transmission at the time of travel.
All travelers to countries in which YF is endemic should be advised of the risks for contracting the disease and available methods to prevent it, including use of personal protective measures and receipt of vaccine. Administration of YF vaccine is recommended for persons aged ≥9 months who are traveling to or living in areas of South America and Africa in which a risk exists for YFV transmission. Because serious adverse events can occur following YF vaccine administration, health-care providers should vaccinate only persons who are at risk for exposure to YFV or who require proof of vaccination for country entry. To minimize the risk for serious adverse events, health-care providers should observe the contraindications, consider the precautions to vaccination before administering vaccine, and issue a medical waiver if indicated.
Introduction
Yellow fever virus (YFV), a mosquito-borne flavivirus, is present in tropical areas of Africa and South America. In humans, the majority of YFV infections are asymptomatic. Clinical disease varies from a mild, undifferentiated febrile illness to severe disease with jaundice and hemorrhagic manifestations (1). After an incubation period of 3--6 days, symptomatic yellow fever (YF) infections typically manifest with an abrupt onset of fever and headache (2). As the illness progresses, other symptoms might occur, including photophobia, myalgias, arthalgias, epigastric pain, anorexia, vomiting, and jaundice (3,4). In some persons, multisystem organ failure with hemorrhagic signs and symptoms occurs. The case-fatality ratio of severe yellow fever with hepatorenal dysfunction is 20%--50% (1). Because no specific antiviral treatment exists for YF, prevention is critical to lower disease risk and mortality.
YFV has three transmissiotn cycles: jungle (sylvatic), intermediate (savannah), and urban (5). All cycles involve the transmission of YFV between primates (nonhuman or human) and mosquitoes. Depending on the transmission cycle and location, different mosquito species are involved, and humans or nonhuman primates serve as the primary reservoir of the virus.
The World Health Organization (WHO) estimates that YFV causes 200,000 cases of clinical disease and 30,000 deaths each year (6). However, the majority of cases and deaths are not recognized because of the predominantly rural nature of the disease and inadequate surveillance and reporting (4,7). In recent decades, hundreds of cases have been reported annually in South America, primarily among men with occupational exposures in forested areas (8). In Africa, the number of cases reported from the continent annually varies substantially (range: 1--5,000 cases). Most of the fluctuation in the annual case numbers from Africa likely is attributable to variations in reporting and recognition. However, because the natural occurrence of disease also can fluctuate, being absent in certain areas for years before reappearing, delineation of affected areas depends on surveillance for animal reservoirs and vectors, accurate diagnosis, and prompt reporting of all human cases.
Evaluation of a traveler's risk should take into account the traveler's immunization status and planned itinerary, including location of travel, season, duration of exposure, occupational and recreational activities, and local rate of virus transmission at the time of travel. All travelers should be provided advice on prevention of mosquito bites. Health-care providers weighing a decision as to whether to administer YF vaccine to travelers must take into account multiple factors (e.g., the risk for travel-associated YFV disease, the high morbidity and mortality when YF does occur, country-entry requirements for YF vaccine, and the risk for serious adverse events following vaccination). This report provides recommendations for use of YF vaccine for prevention of YF among travelers and laboratory workers.
Methods
The Advisory Committee on Immunization Practices (ACIP) Yellow Fever Vaccine Work Group* (the ACIP Work Group) first met in September 2008 to review available information on the risk for YF for travelers and to revise recommendations for the use of the vaccine. In addition to ACIP members, the ACIP Work Group included participants from the American Academy of Family Physicians, CDC, the U.S. Department of Defense, the U.S. Department of State, the Food and Drug Administration (FDA), the Infectious Diseases Society of America, the International Society of Travel Medicine, the Public Health Agency of Canada (PHAC), Boston Medical Center, and the Harvard School of Public Health.
Issues reviewed and considered by the ACIP Work Group included YF epidemiology; incidence of and risk factors for travel-associated YF disease; measures available to prevent YF disease; and YF vaccine immunogenicity, safety, and incidence of vaccine adverse events. Further consideration was given to potential contraindications and precautions to the use of YF vaccine and International Health Regulations (IHRs) for the vaccine. Published, peer-reviewed studies were the primary source of data used. Articles were identified through searches of the PubMed, Global Health, and EMBASE databases; review of relevant bibliographies; and consultation with subject-matter experts. When relevant to issues under discussion, unpublished data available at CDC also were considered.
The ACIP Work Group held monthly conference calls until May 2009. Recommendation options were developed and discussed by the Work Group. When evidence from clinical trials or other research studies was lacking, the recommendations incorporated expert opinion of the ACIP Work Group members. A short overview presentation was made to ACIP at the June 2009 meeting. Proposed recommendations and a draft statement were presented to ACIP and approved at the October 2009 meeting.
Background
Yellow Fever Virus Transmission
YFV is a ribonucleic acid (RNA) virus that belongs to the genus Flavivirus. It is related antigenically to West Nile virus, St. Louis encephalitis virus, and Japanese encephalitis virus (9). YFV is transmitted to humans primarily through the bite of an infected Aedes or Haemagogus species mosquito (10--14). Mosquitoes acquire the virus by feeding on infected nonhuman or human primates and then can transmit the virus to naïve nonhuman or human primates.
YFV has three transmission cycles: jungle (sylvatic), intermediate (savannah), and urban (5) (Figure 1). The jungle (sylvatic) cycle involves transmission of the virus between nonhuman primates (e.g., monkeys) and tree-hole--breeding mosquito species found in the forest canopy (15). The virus is transmitted via mosquitoes from monkey to human when the humans encroach into the jungle during occupational or recreational activities. In Africa, an intermediate (savannah) cycle exists that involves transmission of the YFV from tree-hole--breeding Aedes spp. to humans living or working in jungle border areas (16,17). In this cycle, the virus can be transmitted from monkey to human or from human to human via these mosquitoes. The urban cycle involves anthroponotic transmission of the virus between humans and urban mosquitoes, primarily Ae. aegypti.
Humans infected with YFV experience the highest levels of viremia and are infectious to mosquitoes shortly before the onset of fever and for 3--5 days thereafter (18). Given the high level of viremia attained in humans, bloodborne transmission theoretically could occur through transfusions or needlestick injuries.
Epidemiology of Yellow Fever
Geographic Distribution and Spread
YF occurs in sub-Saharan Africa and tropical South America, where it is endemic and intermittently epidemic (Figure 2). Most YF disease in these areas is attributable to sylvatic or intermediate transmission cycles. However, urban transmission of YF does occur periodically in Africa and sporadically in South America (19--21).
In Africa, the majority of outbreaks have been reported from West Africa; fewer outbreaks have been reported from Central and East Africa (22). The reasons for this are unknown but might include higher densities of humans and Ae. aegypti in the west, genetic differences between YFV strains (genotypes) between the east and west, the presence of antibodies against other flaviviruses contributing to cross-protection, and mosquito species transmitting the disease in East Africa being less likely to bite humans (5,22,23). During West African outbreaks, up to 30% of the population is infected with YFV, and 3%--4% develop clinical disease (24,25). Since 2008, an increased number of YF cases have been reported from Central African countries (e.g., the Central African Republic, Congo, and Chad) (26,27; WHO, unpublished data, 2009). Many of these countries have reported YF cases infrequently in the past, and whether the recent reports of cases in these countries are attributable to improved surveillance or to increased disease activity either locally or via extension from neighboring endemic areas is unclear.
In South America, transmission of YFV occurs predominantly in sparsely populated forested areas rather than in urban areas. Most YF cases are reported from the Orinoco, Amazon, and Araguaia river basins and contiguous grasslands (28). Peru and Bolivia have the highest cumulative incidence of YF over the past 20 years (29). In Brazil, most cases are reported from the Amazon and western Brazil. However, starting in 2007, cases have been identified in more southern and eastern provinces, including Sao Paulo, Rio Grande do Sul, and Goias states (30--32). During this same time, areas contiguous with southern Brazil in northern Argentina and Paraguay also have had confirmed cases of YF, representing the first reemergence of the virus in these regions since the 1970s (31). In addition to the expansion of the endemic zone of the disease in recent years, Ae. aegypti also have reinfested many urban centers in South America, increasing the risk for urban outbreaks of YF in the Americas, as occurred in 2007 in Asunción, Paraguay (21).
Although urban outbreaks of YF occurred in North America and Europe until the early 1900s, autochthonous transmission has not been reported over the past several decades. Local YFV transmission never has been identified in Asia or Australia. However, these areas are at risk for importation, as many urban areas in these regions have Ae. aegypti and have a large susceptible human population (33). As a result, some countries in which the disease is not endemic (e.g., India and Australia) require proof of YF vaccination from travelers arriving from regions in which YFV is endemic (34).
Incidence and Burden of Disease
WHO estimates that 200,000 cases of YF and 30,000 deaths attributable to YF occur annually worldwide (6). However, only a limited percentage of these cases are identified because of underreporting (24,25). During 1988--2007, a total of 26,356 YF cases were reported to WHO, including 23,056 (87%) cases from sub-Saharan Africa and 3,300 (13%) from South America (1,35--37). In Africa, the number of cases reported annually varies widely (range: 1--5,000), which suggests inconsistencies in surveillance and reporting. In South America, as many as 524 cases per year have been reported to WHO. The case-fatality ratio is highly variable but is approximately 20% in Africa and approximately 50% in South America (1). The higher case-fatality ratio in South America is believed to be the result of enhanced diagnostic testing of persons with fatal disease rather than an indication of a more virulent form of the disease (5).
Ecologic and Seasonal Patterns
The ecology of YFV is complex, and multiple factors contribute to its transmission. In general, increased temperature, humidity, and rainfall lead to higher mosquito abundance and consequently an increase in viral circulation (38,39). In South America, YF incidence is highest during months of increased rainfall, humidity, and temperature (January--May) (40). In intermediate zones of West Africa, YFV activity increases during the middle of the rainy season (around August) and peaks during the early dry season (October) (40,41). This period corresponds to the period of maximum longevity of mosquito vectors. Because Ae. aegypti breed in containers used for water storage, mosquito activity and reproduction patterns are less dependent on rainfall (41). As a result, when Ae. aegypti are involved in virus transmission, YF also can occur in the dry season.
Age-Specific Patterns
The age-specific patterns of YF disease in Africa and South America differ and are affected by sex and occupation. In Africa, cases often occur in villages in the savannah region contiguous with the forest. Children are predominantly affected, as older persons in the community frequently have naturally acquired or vaccine-acquired immunity (24). In South America, YF disease occurs most frequently in unvaccinated young men who are exposed to mosquito vectors through their work in forested or jungle areas (42,43). Occupations involving lumbering and forest clearing for building or road construction have been linked to human infection (28). The age and sex distribution of YF cases in urban areas of South America differs from that observed in jungle areas, with a higher prevalence of infections occurring among children and women attributable to Ae. aegypti breeding around homes (21).
Clinical Manifestations and Diagnosis
Signs and Symptoms
In the majority of persons infected with YFV, infections are believed to be asymptomatic (24,25). Clinical disease varies from a mild, undifferentiated febrile illness to severe disease with jaundice and hemorrhagic manifestations. The incubation period is usually 3--6 days (1,2). In its mildest form, YF is a self-limited infection characterized by sudden onset of fever and headache without other symptoms (3). Other patients experience an abrupt onset of a high fever (up to 104°F [40°C]), chills, severe headache, generalized myalgias, lumbosacral pain, anorexia, nausea, vomiting, and dizziness (4,43). The patient appears acutely ill, and examination might demonstrate bradycardia in relation to the elevated body temperature (Faget's sign). The patient is usually viremic during this period, which lasts for approximately 3 days. Many patients have an uneventful recovery, but in approximately 15% of infected persons, the illness recurs in more severe form within 48 hours following the viremic period (44). Symptoms include fever, nausea, vomiting, epigastric pain, jaundice, renal insufficiency, and cardiovascular instability (4). Viremia generally is absent during this phase of symptom recrudescence. A bleeding diathesis can occur, with hematemesis, melena, metrorrhagia, hematuria, petechiae, ecchymoses, epistaxis, and oozing blood from the gingiva and needle-puncture sites. Physical findings include scleral and dermal icterus, hemorrhages, and epigastric tenderness without hepatic enlargement (44).
Clinical Laboratory Findings
Multiple laboratory abnormalities can be observed in patients with YF; these can vary depending on the severity and stage of illness. In the first week of the illness, leukopenia might occur; however, leukocytosis also can occur during the second week of the disease (3). Bleeding dyscrasias also can occur, together with elevated prothrombin and partial thromboplastin times, decreased platelet count, and presence of fibrin-split products (45,46). Hyperbilirubinemia might be present as early as the third day but usually peaks toward the end of the first week of illness. Elevations of serum transaminase levels occur in severe hepatorenal disease and might remain elevated for up to 2 months after onset (47).
Preliminary diagnosis is based on the patient's clinical features, YF vaccination status, and travel history, including destination, time of year, and activities. Mild YF cannot be distinguished clinically from a range of other infections. Cases of YF with jaundice must be differentiated from viral hepatitis, malaria, leptospirosis, Congo-Crimean hemorrhagic fever, Rift Valley fever, typhoid, Q fever, and typhus, as well as surgical, drug-induced, and toxic causes of jaundice (17). The other viral hemorrhagic fevers, which usually manifest without jaundice, include dengue hemorrhagic fever, Lassa fever, Marburg and Ebola virus diseases, and Bolivian, Argentinean, and Venezuelan hemorrhagic fevers (48,49).
Laboratory Confirmation
Laboratory diagnosis generally is accomplished by testing serum to detect virus-specific immunoglobulin M (IgM) and immunoglobulin G (IgG) antibodies by serologic assays (50). Serologic cross-reactions occur with other flaviviruses (e.g., West Nile or dengue viruses), so positive results should be confirmed with a more specific test (e.g., plaque reduction neutralization test) (49). Early in the illness (during the first 3--4 days), YFV or YFV RNA often can be detected in the serum by virus isolation or nucleic acid amplification testing (e.g., reverse transcription-polymerase chain reaction [RT-PCR]). However, by the time overt symptoms are recognized, the virus or viral RNA usually is undetectable. Therefore, virus isolation and RT-PCR should not be used for excluding the diagnosis of YF. Immunohistochemical staining of formalin-fixed material can detect YFV antigen on histopathologic specimens (51). Health-care providers should contact their state or local health department and CDC (at telephone 1-970-221-6400) for assistance with diagnostic testing for YFV infections.
Treatment and Management
Although multiple drugs have been evaluated or used empirically to treat YF disease, to date, none has demonstrated specific benefit (52). Management is supportive and based on symptoms and the organ systems involved. Patients with multisystem organ involvement likely will require critical-care support with possible mechanical ventilation or hemodialysis. Rest, fluids, and nonsteroidal anti-inflammatory drugs or acetaminophen can relieve milder symptoms of fever and myalgias. Aspirin should be avoided because of the risk for hemorrhagic complications. Infected persons should be protected from further mosquito exposure (staying indoors and/or under a mosquito net) during the first few days of illness so they do not contribute to the transmission cycle.
Outcome and Sequelae
The majority of persons with mild YF illness recover without long-term sequelae. For those with severe disease involving hepatorenal dysfunction, the length of illness is variable, and the case-fatality ratio is 20%--50% (1). Risk factors for death include hypotension, shock, renal failure, severe hemorrhagic disease, coma, and convulsions (44). For those who survive, convalescence often is prolonged, lasting several weeks. Rarely, death can occur at the end of convalescence or even weeks after complete recovery from the acute illness. These late deaths are thought to be attributable to myocardial damage and cardiac arrhythmia (53). Secondary bacterial infections (e.g., pneumonia) also can complicate recovery. Jaundice has been observed for up to 3 months after recovery from serologically documented YF (45).
Yellow Fever Among Travelers
During 1970--2009, nine cases of YF were reported in unvaccinated travelers from the United States and Europe who traveled to West Africa (five cases) or South America (four cases) (54--63). Eight of these nine travelers died (19,40). Only one case of YF has been documented in a vaccinated traveler, a female aged 37 years from Spain who visited several West African countries during 1988 (56).
A traveler's risk for acquiring YF is determined by multiple factors, including immunization status, use of personal protection measures against mosquito bites, location of travel, duration of exposure, occupational and recreational activities while traveling, and local rate of virus transmission at the time of travel. In both West Africa and South America, YFV transmission typically is seasonal and is associated with the mid-to-late rainy season (). However, YFV can be transmitted by Ae. aegypti even during the dry season in both rural and densely settled urban areas (41). Although the number of reported cases of human disease often is used to estimate the crude level of endemic transmission, cases might not be reported because of a low level of transmission, a high level of immunity in the local population (e.g., because of vaccination), or cases not being detected by local surveillance systems. Therefore, a lack of human disease cases in an area does not equate to absence of risk for transmission.
The risk for acquiring YF is difficult to predict because of variations in ecologic determinants of virus transmission. For a 2-week stay, the estimated risks for illness and death attributable to YF for an unvaccinated traveler traveling to an area of West Africa where the disease is endemic are 50 and 10 cases per 100,000 population, respectively; for South America, the risks for illness and death are five cases and one case per 100,000 population, respectively (40). These crude estimates for unvaccinated travelers are based on risk to indigenous populations, often during peak transmission season. Thus, these risk estimates might not reflect accurately the actual risk to travelers, who might have a different immunity profile, take precautions against getting bitten by mosquitoes, and have less outdoor exposure. The risk for acquiring YF in South America might be lower than that in Africa because the mosquitoes that transmit the virus between monkeys in the forest canopy do not come in contact with humans often, and local residents have a relatively high level of immunity secondary to widespread vaccine use.
Yellow Fever Vaccine
YF vaccine was first developed in the 1930s after successful attenuation of the Asibi strain of YFV (64). Two vaccines were established: the 17D and the French neurotropic vaccines. The French neurotropic vaccine was manufactured until 1982, when production was stopped because of higher rates of neurologic adverse events reported following use of the vaccine (1).
Two 17D substrain vaccines are manufactured today: the 17DD and 17D-204 YF vaccines. The YFV strains in these two vaccines share 99.9% sequence homology (65). The 17DD YF vaccine is manufactured in Brazil and is used in Brazil and many other South American countries. The 17D-204 vaccine is manufactured and used outside of Brazil, including in the United States. Studies comparing the various 17D YF vaccines produced by multiple manufacturers suggest that the reactogenicity or immune responses generated by these vaccines do not differ (66--70). Thus persons who receive YF vaccines in countries other than the United States should be considered protected against YF.
Vaccine Composition, Storage, and Handling
The 17D-204 strain vaccine, YF-VAX (manufactured by sanofi pasteur, Swiftwater, Pennsylvania), is a freeze-dried supernatant of centrifuged embryo homogenate, packaged in 1-dose and 5-dose vials for domestic use. The vaccine should be stored at temperatures of 35ºF--46ºF (2ºC--8ºC) until it is reconstituted by the addition of diluent (sterile, physiologic saline) supplied by the manufacturer. Multidose vials of reconstituted vaccine should be stored at 35ºF--46ºF (2ºC--8ºC) for up to 1 hour. Any unused vaccine that is not used within 1 hour of reconstitution must be discarded (71).
Correlates of Protection
No human studies have been performed to determine the correlates of protection for YFV infection. However, dose-response studies conducted in rhesus monkeys have established the minimal level of neutralizing antibodies needed to protect the monkeys against virulent YFV. Testing was conducted with constant amounts of serum and varying dilutions of virus to establish a log10 neutralization index (LNI). These studies demonstrated that LNI >0.7 was correlated strongly with protection (1,72). Although the amount of serum needed for LNI testing is suitable for animal studies or clinical trials, it precludes routine screening among humans (1). Therefore, a similar test, plaque reduction neutralization test (PRNT), which uses a constant amount of virus and varying dilutions of serum, is used most frequently in diagnostic tests to determine the absence or presence of neutralizing antibodies and the specific serum antibody titer.
Efficacy and Immunogenicity
No human efficacy studies have been performed with YF vaccine. However, several observations support YF vaccine being protective in humans, including 1) the reduction of laboratory-associated infections in vaccinated workers, 2) the observation following initial use of the vaccine in Brazil and other South American countries that YF occurred only in unvaccinated persons, and 3) the rapid disappearance of cases during YF vaccination campaigns initiated during epidemics. Unpublished reports comparing YF incidence among vaccinated and unvaccinated populations during a 1986 epidemic in Nigeria estimated vaccine effectiveness to be approximately 85% (1). Worldwide, only five cases of YF have been reported in vaccine recipients since YF vaccine has been in use; whether any or all of them were vaccinated properly with appropriately handled YF vaccine is uncertain (1).
Following YF vaccine administration, primary vaccine recipients often develop a low-level viremia with the vaccine virus. The viremia usually occurs within 3--7 days and persists for 1--3 days, abating as YFV IgM antibodies are developed (73,74). The level of viremia following vaccination is high enough to be transmitted through blood products. Transfusion-related transmission of YF vaccine virus has been documented in three persons who received blood products that were collected from persons vaccinated 4 days before their blood donation (75).
Studies have demonstrated that 80%--100% of vaccinated persons develop neutralizing antibodies by 10 days after vaccination (74,76,77). Most studies indicate that >99% of vaccinated persons developed YFV neutralizing antibodies by 28 days after vaccination (1). Overall, the vaccine-induced antibodies are delayed in their formation and at lower titers compared with antibodies developed in response to wild-type YFV infection (1).
A study of two YF vaccines (YF-VAX and Arilvax) in 1,440 healthy adults found different neutralizing antibody responses to the vaccine, depending on sex and race. For both vaccines, males and Caucasians were noted to have higher LNI levels when compared with females, blacks, and Hispanics (66). The age at which a person is vaccinated with YF vaccine does not appear to affect immunologic response to the vaccine (1).
IHRs allow countries to require proof of YF vaccination as a condition of entry for travelers arriving from certain countries to prevent importation and indigenous transmission of YFV. IHRs stipulate that the vaccination certificate for YF is valid beginning 10 days after administration of YF vaccine for primary vaccine recipients (78). This time period corresponds to the time at which the majority of vaccinees demonstrate immunity. The YF vaccination certificate for international travel is valid for 10 years. This interval was based on published studies indicating that neutralizing antibodies were present in >90% of persons 16--19 years after vaccination (79). Later studies of U.S. military veterans from World War II tested 30--35 years after a single dose of YF vaccine demonstrated that >80% had neutralizing antibody; in certain subgroups, >95% still had neutralizing antibodies (80).
Healthy persons rarely fail to develop neutralizing antibodies following YF vaccination. In controlled clinical trials, the primary failure rate is generally about 1% (66). Persons who fail to develop antibody after their first vaccination can develop antibody upon revaccination (81).
Three host factors have been associated with failure to respond immunologically to YF vaccine: pregnancy, human immunodeficiency virus (HIV) infection, and malnutrition. In one study, only 39% of pregnant women seroconverted after receiving the vaccine in their third trimester (82). This difference was attributed to the altered immune state associated with pregnancy and suggests the need to revaccinate at-risk women who were vaccinated during pregnancy. However, a more recent study indicated that 425 (98%) of 441 women who were vaccinated inadvertently with YF vaccine, primarily during their first trimester, developed YFV-specific neutralizing antibodies (83). Therefore, revaccination might not be necessary in women who received vaccine during pregnancy, but antibody titers should be checked to ensure an appropriate immune response in women still at risk for the disease.
HIV infection has been associated with a reduced immunologic response to a number of inactivated and live, attenuated vaccines, including YF vaccine (84). Some case reports have suggested that vaccinating HIV-infected persons who are not immunosuppressed (CD4+ T-cell count [CD4 count] >500/mm3) results in seroconversion (85). However, a recent retrospective cohort study indicated that significantly fewer HIV-infected persons had YFV neutralizing antibodies at 1 year after vaccination compared with vaccinated uninfected persons (83% and 97%, respectively; p=0.01) (86). The study measured YFV neutralizing antibodies in HIV-infected persons with median baseline CD4 count of 496 cells/mm3 (range: 72--1,730 cells/mm3) and varying levels of HIV RNA detected in their blood (52% had HIV RNA levels ≥50 copies/mL). Among HIV-infected infants in one developing nation, only 17% developed neutralizing antibodies within 10 months of YF vaccine compared with 74% of HIV-uninfected controls matched for age and nutritional status (87). The mechanisms for this observed difference in immune response among HIV-infected persons are uncertain but appear to correlate with HIV RNA level and CD4 count. Further studies are required to assess the relevance of these findings.
Adverse Events
General Events
After YF vaccination, vaccinees have reported mild headaches, myalgia, low-grade fevers, or other minor symptoms for 5--10 days (71). Localized pain, swelling, erythema, or warmth might occur at the injection site for up to a week following vaccination. In one large study of 1,440 vaccine recipients, the most common systemic side effects were headache (33% of subjects), myalgia (25%), malaise (19%), fever (15%), and chills (11%) (66). These side effects generally were mild, and only 1% of vaccinees curtailed their regular activities as a result. In the only placebo-controlled trial of 1,007 adults vaccinated with either YF vaccine (the 17-DD vaccine virus strain) or placebo, 3% of reported local reactions and 7% of systemic reactions were attributed to vaccine (88).
The Vaccine Adverse Event Reporting System (VAERS) is a passive surveillance system for adverse events following vaccination operated collaboratively by CDC and FDA. Adverse event reports can be submitted to VAERS from a variety of sources, including vaccine providers and recipients, medical practitioners, and manufacturers. The rate of any adverse events following YF vaccine as reported to VAERS is 43 adverse events per 100,000 doses distributed (89). The majority of reported adverse events are classified as nonserious (rate: 38 per 100,000 population) and include reports of fever, injection-site pain, injection-site erythema, pruritis, headache, urticaria, and rash. The majority of events occur a median of 1 day after vaccination; roughly 60% occur within the first 2 days after vaccination.
During 2000--2006, a rate of 4.7 serious adverse events per 100,000 doses distributed was derived from VAERS data. Reporting rates were highest among persons aged ≥60 years (8.3 per 100,000 doses). Three well-characterized serious adverse events occur following YF vaccine administration: 1) immediate hypersensitivity or anaphylactic reactions, 2) YF vaccine-associated neurologic disease (YEL-AND), and 3) YF vaccine-associated viscerotropic disease (YEL-AVD).
In 2002, CDC formed the Yellow Fever Vaccine Safety (YFVS) Working Group, consisting of vaccine safety experts from CDC and partner organizations. The YFVS Working Group systematically reviewed VAERS reports for YF vaccine and devised a surveillance case definition for YEL-AND and YEL-AVD (Boxes 1 and 2) to help classify cases and determine rates of these adverse events.
Hypersensitivity and Anaphylactic Reactions
Multiple components of YF vaccine have been cited as possible allergens, including eggs, chicken proteins, gelatin, or latex (found in vial stopper) (71). Immediate hypersensitivity reactions or anaphylaxis, characterized by urticaria and respiratory symptoms (e.g., dyspnea, bronchospasm, or pharyngeal edema) are uncommon and occur principally among persons with histories of allergies to egg or other substances (90). However, anaphylaxis has been reported to occur in persons with no history of reactions to the vaccine's components. Rates of anaphylaxis from VAERS data vary (range: 0.8--1.8 events per 100,000 doses distributed) (89,90).
YEL-AND
YEL-AND is a serious but rarely fatal adverse event. YEL-AND manifests as several distinct clinical syndromes, including meningoencephalitis (neurotropic disease), Guillain-Barré syndrome (GBS), acute disseminated encephalomyelitis (ADEM), and bulbar palsy (91,92). Meningoencephalitis occurs as a result of direct YF vaccine viral invasion of the central nervous system (CNS) with infection of the meninges and/or the brain. The other neurologic syndromes (e.g., GBS and ADEM) represent autoimmune manifestations in which antibodies and/or T-cells produced in response to the vaccine cross-react with neuronal epitopes and lead to central or peripheral nerve damage.
Through the attenuation process, YF vaccine virus strains were determined to develop neurovirulence (93,94). Early in the vaccine's history, multiple cases of encephalitis associated with vaccine administration were noted and determined to be attributable to too few or too many passages of the virus during the attenuation process. This observation led to the establishment of a vaccine seed lot system in 1945 that clearly defined the number of passages allowed for certain strains of the vaccine (95). However, even after the implementation of the seed lot system, additional cases of encephalitis associated with YF vaccine continued to be reported. Studies conducted during the early 1950s identified four cases of encephalitis out of 1,000 children aged <6 months vaccinated with YF vaccine (96). An additional 10 cases of encephalitis associated with YF vaccine administered to infants aged <4 months were reported worldwide during the 1950s (1). Consequently, recommendations were made in the 1960s restricting the use of YF vaccine to infants aged ≥6 months (97). Subsequently, the number of cases of encephalitis reported in infants diminished.
A total of 29 cases of YEL-AND (nine of meningoencephalitis, eight of GBS, three of ADEM, one of bulbar palsy, and eight cases with insufficient data to classify) that occurred during 1990--2006 were reviewed retrospectively or prospectively by the YFVS Working Group (91,92,98,99). The 29 cases had a varied time from vaccination to symptom onset (range: 3--28 days). For the 25 cases with data available on age, there again was a wide variation in age range (range: 6--78 years). All cases were reported to occur in first-time vaccine recipients. One death was reported in a male aged 53 years who had unrecognized asymptomatic human immunodeficiency virus (HIV) infection and CD4 count <200/mm3 (98). The patient developed fever and malaise 3 days after his vaccination and became encephalopathic at 5 days postvaccination. He died at 9 days following vaccination.
Data regarding the specific age and illness onset for the different neurologic presentations can be derived from VAERS reports (91). Of six reported encephalitis cases, the median age was 54 years (range: 16--78 years), and median time to onset after vaccination was 14 days (range: 5--24 days). For the six GBS cases, the median age was 53 years (range: 17--68 years), and median time to onset after vaccination was 13 days (range: 7--27 days). Finally, for the three ADEM cases, the median age was 19 years (range: 18--61 years), and median time to onset after vaccination was 15 days (range: 7--20 days).
A recently published report has included the first report of a case of longitudinal myelitis in a male aged 56 years who received no other vaccines (100). YFV IgM antibodies were detected in his cerebrospinal fluid (CSF) and serologic testing for other regional flaviviruses was negative. However, the time to symptom onset was 45 days after vaccination, which is longer than that reported for all other YEL-AND cases. A second recently published report concerned a male aged 23 years who presented originally with meningoencephalitis but was later determined to have ADEM (101). The diagnosis of ADEM was determined by magnetic resonance imaging of the brain.
The diagnostic testing for YEL-AND is specific to the clinical presentation. For cases with meningitis or encephalitis, the detection of the YF vaccine virus in CSF either by culture or nucleic acid amplification is diagnostic. The detection of YFV IgM and specific neutralizing antibodies in the CSF also supports the diagnosis. YFV IgM antibodies are not believed to cross the blood-brain barrier normally, so their production in CSF is considered indicative of local CNS infection. For autoimmune mediated events, no specific YF testing is available. The diagnosis of these conditions should be made using appropriate studies (e.g., neuroimaging, electroencephalography, electromyelography, and nerve conduction studies). Because no confirmatory YFV-specific laboratory test exists for the autoimmune syndromes of ADEM and GBS, the YFVS Working Group case definition only specifies "suspect" and "probable" levels of causality. If no evidence of another diagnosis exists, a case is classified further depending on whether YF vaccine was the only vaccine administered. Diagnostic testing for suspected YEL-AND cases is available from CDC at http://www.cdc.gov/ncidod/dvbid/yellowfever/vaccine/special-testing.htm.
Treatment for YEL-AND also depends on the particular clinical syndrome. Treatment for meningoencephalitis is supportive, and manifestations such as seizures or autonomic dysfunction should be managed according to acceptable medical standards for each disorder. For autoimmune neurologic manifestations associated with YF vaccine, treatment often includes intravenous immune globulin (IVIG) or plasmapheresis for GBS, and corticosteroids, IVIG, or plasmapheresis for ADEM (102,103).
YEL-AND rarely is fatal. In addition to the case described previously of fatal encephalitis in a person with HIV, four other deaths have been reported attributable to YEL-AND (1,104). One fatal case of encephalitis occurred in a girl aged 3 years who was vaccinated in 1965 (104). Molecular studies of the YF vaccine virus isolated from the brain of this patient demonstrated that the virus had mutated and become more neurovirulent as documented in monkey studies (105). This is the only case in which a mutation of the vaccine virus has been linked to an adverse outcome in a recipient. Three other fatal YEL-AND cases were reported unofficially from a hospital-based surveillance system for postvaccine encephalitis in Kenya after a mass vaccination campaign in 1993 (1).
The incidence rate of YF vaccine-associated encephalitis in very young infants has been estimated on the basis of reports from the time period before institution of the age restriction for vaccination to be 50--400 cases per 100,000 population (96,106). From VAERS data, the reporting rate for YEL-AND is 0.4--0.8 cases per 100,000 doses distributed (89,107). The reporting rate was higher for persons aged ≥60 years, with a rate of 1.6 cases per 100,000 doses distributed in persons aged 60--69 years and 1.1--2.3 cases per 100,000 doses distributed for persons aged ≥70 years. These estimates most likely are low given underreporting. In countries in which YF is endemic, a national vaccine adverse event surveillance system might not exist or, if one does exist, be consistent and sensitive enough to detect the majority of YEL-AND cases that do occur. However, an unofficial report from a hospital-based surveillance system for postvaccine encephalitis in Kenya after a mass vaccination campaign estimated the incidence of YEL-AND to be 0.6 cases per 100,000 doses (1).
YEL-AVD
In 2001, a previously unrecognized serious adverse reaction among recipients of YF vaccine was first described (48,108--113). This syndrome initially was termed febrile multiple organ system failure; the name subsequently was changed to YEL-AVD. YEL-AVD mimics naturally acquired YF disease, with the vaccine virus proliferating and disseminating throughout the host's tissues.
As of February 2010, a total of 57 cases of YEL-AVD have been reported to CDC from 14 countries (Australia, Belgium, Brazil, China, Colombia, Ecuador, France, Germany, Japan, Peru, Spain, Switzerland, the United Kingdom, and the United States). The cases have occurred in association with the different substrains of YF vaccine produced by several manufacturers. On the basis of an analysis of cases for which information is available, YEL-AVD has occurred only following a recipient's first YF vaccination; no cases of YEL-AVD occurring in persons receiving booster doses of the vaccine have been reported. Of the 53 persons with YEL-AVD cases whose sex is known, 32 (60%) were male. The overall median age was 50 years (range: 3--81 years) but differed by sex, with a median age of 62 years (range: 4--81 years) in males and 24 years (range: 3--79 years) in females. The median time from vaccination until symptom onset was 3 days (range: 1--8 days). Of 57 patients, 37 (65%) died, with a higher case-fatality ratio in females (19 [90%]) than males (16 [50%]) (sex was unknown in two fatal cases). The median time from YF vaccination until death was 10 days (range: 7--30 days). Whether age and case-fatality ratio differences between sexes reflect sex-specific host factors or are a function of surveillance or reporting bias is unclear.
Patients with YEL-AVD typically develop fever and other nonspecific signs and symptoms (including headache, malaise, myalgias, nausea, vomiting, or diarrhea) within 1 week of vaccination. As the illness progresses, jaundice and laboratory abnormalities (e.g., thrombocytopenia, elevations of hepatic transaminases, total bilirubin, and creatinine) might occur. Leukopenia or leukocytosis might be present, depending on the stage and severity of the illness. Persons with severe cases can develop hypotension, hemorrhage, renal failure requiring hemodialysis, and respiratory failure requiring mechanical ventilation. Less frequent complications include rhabdomyolysis and disseminated intravascular coagulation. No specific therapy exists for YEL-AVD, and, assuming other diseases have been excluded, treatment is supportive.
YF vaccine-specific laboratory testing can assist in making the diagnosis. Documentation of high levels of YF vaccine virus in the serum of patients within the first 7 days of vaccination or prolonged presence of replicating YF vaccine virus in blood >7 days postvaccination is consistent with YEL-AVD (probable case). In fatal cases, when an autopsy has been performed, immunohistochemistry can be performed on fixed tissues to detect YFV antigen, and RT-PCR can be performed on RNA extracted from frozen tissues to verify the presence of YF vaccine virus versus wild-type YFV. Liver tissue can have characteristic histopathologic findings (e.g., midzonal necrosis, microvesicular fatty changes, and Councilman bodies) (114). Diagnostic testing for suspect YEL-AVD cases is available from CDC at http://www.cdc.gov/ncidod/dvbid/yellowfever/vaccine/special-testing.htm.
Several risk estimates for YEL-AVD have been published. On the basis of VAERS data, the reporting rate of YEL-AVD is 0.3--0.4 cases per 100,000 doses distributed (89,107). Similar to YEL-AND, reporting rates also are highest among persons aged ≥60 years, with rates of 1.0--1.1 cases per 100,000 doses distributed in persons aged 60--69 years and 2.3--3.2 cases per 100,000 doses distributed for persons aged ≥70 years. An analysis of YEL-AVD cases that occurred in Europe during 1996--2003 yielded an overall incidence of 0.3 cases per 100,000 doses distributed (92). A Brazilian analysis based on four fatal cases of YEL-AVD derived risk estimates of fatal YEL-AVD ranging from <0.01 to 0.21 per 100,000 doses administered (115). However, more recent risk estimates from Brazil have identified overall YEL-AVD rates similar to those from the United States and Europe (CDC, unpublished data, 2010).
Almost all cases of YEL-AVD have occurred in persons who received different lots of vaccine. However, at least one known cluster has been reported involving five cases of YEL-AVD associated with the same lot of YF vaccine (116). This cluster occurred in 2007 after a vaccination campaign in an area of Peru in which the disease was not endemic. Four patients died of confirmed YEL-AVD. The overall incidence of YEL-AVD in this campaign was 7.9 cases per 100,000 doses distributed, compared with a lot-specific incidence of 11.7 cases per 100,000 doses distributed for the implicated lot. A thorough investigation detected no abnormalities of the implicated vaccine lot and no common risk factors among the five case patients. The incidence of YEL-AVD associated with this campaign in Peru is substantially higher than previous risk estimates from other countries and remains unexplained.
To date, two specific risk factors for YEL-AVD have been identified: older age and a history of thymus disease or thymectomy. The two analyses of VAERS reports of YEL-AVD in the United States yielded reporting rates of YEL-AVD for persons aged ≥60 years of 1.4--1.8 cases per 100,000 doses distributed (89,107). This age-specific rate is several-fold higher than the overall reported rate of 0.3--0.4 cases per 100,000 doses distributed. Four (17%) of the first 23 patients with YEL-AVD identified had a history of thymectomy for either a benign or malignant thymoma (117). The details of these cases suggest that a history of thymus disease is a risk factor for YEL-AVD. In 2003, the package insert for YF vaccine available in the United States was updated to include a history of thymus disorder as a contraindication to vaccine administration (71). No additional cases of YEL-AVD identified since 2004 have involved a history of thymus disease.
A number of cases of YEL-AVD have involved a history of autoimmune disease or diseases with potential autoimmune etiology. These include three patients with systemic lupus erythematosis, two with Addison's disease, one with Crohn's disease in remission, one with a history of polymyalgia rheumatica and hypothyroidism, one with ulcerative colitis, and one with myasthenia gravis (108,116,117; CDC, unpublished data, 2010). These cases suggest that autoimmune disease might be a risk factor for YEL-AVD. However, four patients with underlying autoimmune disease were aged >60 years, two of whom had a history of thymectomy for a thymoma; both of these factors are associated with increased risk for YEL-AVD. Therefore, more information is needed regarding the number of patients with underlying autoimmune disease who are vaccinated safely to better address the potential risk for YEL-AVD in persons with autoimmune disease.
Although studies on certain cases of YEL-AVD have identified potential genetic risk factors (118), no common genetic abnormality has been demonstrated. No conclusive evidence has been reported that demonstrates that YEL-AVD has been caused by a reversion to increased virulence in the vaccine (114,119).
Vaccination of Women During Pregnancy and Breastfeeding
No data are available on the YF disease risk for pregnant or breastfeeding women and their fetuses or infants. However, on the basis of surveillance and outbreak data, pregnant and breastfeeding women do not appear to be at risk for more severe YF disease.
The use of YF vaccine during pregnancy has not been studied in a large prospective trial (FDA Pregnancy Category C). Limited data are available from a number of studies in which pregnant women either were vaccinated inadvertently or administered the vaccine in outbreak settings. Two studies involving 81 infants whose mothers had received YF vaccine identified YFV IgM antibodies in the cord blood of one infant shortly after birth (82,120). These findings suggest that infection of the fetus with YF vaccine can occur, albeit at a low incidence (i.e., one of 81). A study of 304 infants born to women who were vaccinated with YF vaccine early in their pregnancies found no increased risk for major malformations (121). An increased risk was identified for minor malformations (e.g., pigmented nevi), but the finding could have resulted from assessment bias. One study of 39 pregnant women who were vaccinated with YF vaccine identified a higher rate of spontaneous abortions in pregnant women receiving the vaccine, with a relative risk of 2.3, but the difference was not statistically significant (95% confidence interval = 0.7--8.0; p=0.2) (122). A more recent study of 441 women inadvertently vaccinated early in their pregnancy did not find an increased risk for fetal death (7.4/1,000 in vaccinated women versus 18.5/1,000 unvaccinated women in the general population) (83).
The proportion of women vaccinated during pregnancy who develop YFV antibodies is variable and might be related to the trimester in which they received vaccine. Of 101 pregnant women receiving YF vaccine predominantly in their third trimester, 39% had evidence of seroconversion to YFV (82). Of 433 women vaccinated predominantly in the first trimester, 425 (98%) developed YFV-specific neutralizing antibodies (83). Because pregnancy might affect immunologic function, serologic testing to document an immune response to the vaccine should be considered.
No evidence exists that breastfeeding mothers have an altered response to YF vaccine. Very limited safety data are available on the use of YF vaccine in breastfeeding women and their infants. One probable and one confirmed case of YEL-AND have been reported in infants whose mothers were vaccinated with YF vaccine (PHAC, unpublished data, 2008; 123). Both infants were breastfed exclusively, were aged <1 month at the time of the exposure, and had not received the vaccine themselves. YF vaccine virus was recovered from the CSF of the confirmed case patient, and YFV-specific IgM antibodies were detected in the probable case-patient's CSF. Testing was not performed on the breast milk in either case to confirm the presence of vaccine virus. RNA from West Nile virus, a related flavivirus, has been identified in breast milk, which suggests that this is a plausible mode of transmission (124,125). Further research is needed to document the risk for potential vaccine exposure through breastfeeding.
Summary of Rationale for YF Vaccine Recommendations
YF is a vectorborne disease resulting from the transmission of YFV to a human from the bite of an infected mosquito. It is endemic to sub-Saharan Africa and tropical South America. Infection in humans is capable of producing hemorrhagic fever and is fatal in 20%--50% of persons with severe disease. Because no treatment exists for YF disease, prevention is critical to lower disease risk and mortality.
The risk for acquiring YF for travelers is determined by multiple factors, including vaccination status, location of travel, season, duration of exposure, occupational and recreational activities while traveling, and local rate of virus transmission at the time of travel. Although only 10 cases of imported YF in travelers from Europe (seven cases) or the United States (three cases) have been reported since 1970, the low number of cases is likely, at least in part, to be a result of the routine use of the vaccine. In attempts to minimize the potential importation and spread of the disease into new areas, many countries enforce the IHR and have vaccine requirements for entry.
Decisions regarding the use of YF vaccine for travelers must take into account the overall risk for travel-associated YFV disease, the high mortality when it does occur, vaccine requirements per IHR, contraindications or precautions for vaccination, and risk for serious adverse events following YF vaccine administration.
Recommendations for the Use of YF Vaccine in Laboratory Workers
YF vaccine is recommended for laboratory personnel who might be exposed to virulent YFV or to concentrated preparations of YF vaccine virus strains by direct or indirect contact or by aerosols.
Recommendations for Prevention of YF Among Travelers
All travelers to countries in which YF is endemic should be advised of the risks for the disease and available methods to prevent it, including personal protective measures and vaccine.
Personal Protective Measures
All travelers should take precautions to avoid mosquito bites to reduce the risk for YF and other vector-borne infectious diseases. These precautions include using insect repellent, wearing permethrin-impregnated clothing, and staying in accommodations with screened or air-conditioned rooms. Additional information on protection against mosquitoes and other arthropods is available at http://wwwnc.cdc.gov/travel/yellowbook/2010/chapter-2/protection-against-mosquitoes-ticks-insects-arthropods.aspx.
Recommendations for the Use of YF Vaccine in Travlers
YF vaccine is recommended for persons aged ≥9 months who are traveling to or living in areas at risk for YFV transmission in South America and Africa. Countries with risk for YFV transmission have been listed (Table 1). For many of these countries, only a portion of the country is at risk for YFV transmission (Figure 2). Given that changes in the designation of endemic regions can occur, travelers and health-care providers should obtain updated information from CDC, available at http://wwwnc.cdc.gov/travel/yellowbook/2010/chapter-2/yellow-fever.aspx.
YF vaccine may be required for entry into certain countries. A list of country-specific requirements is available at http://wwwnc.cdc.gov/travel/yellowbook/2010/chapter-2/yellow-fever-vaccine-requirements-and-recommendations.aspx. Requirements are mandatory and are intended primarily to prevent importation into and transmission of YFV within a given country. However, IHRs stipulate that a medical provider may issue a waiver of YF vaccination to a traveler to fulfill these requirements if the provider judges that YF vaccination is medically contraindicated.
Because of the risk for serious adverse events that can occur following YF vaccine administration, health-care providers should vaccinate only persons who are at risk for exposure to YF virus or require proof of vaccination for country entry. To minimize further the risk for serious adverse events, health-care providers should observe the contraindications carefully and consider the precautions to vaccination before administration of YF vaccine and provide a medical waiver when needed (Table 2).
Requirements for Vaccination Before International Travel
Certain countries require evidence of vaccination from all entering travelers, which includes direct travel from the United States (34). Travelers who arrive in a country with a YF vaccination entry requirement without proof of vaccination may be quarantined for up to 6 days.
Country-entry requirements for proof of YF vaccination under IHRs are different from ACIP or CDC recommendations. Requirements are mandatory and are intended primarily to prevent importation into and transmission of YFV within a given country. CDC's recommendations are public health advice provided to travelers on the basis of the best available epidemiologic data to prevent YFV infection among travelers visiting countries with a risk for YFV transmission.
For purposes of international travel, YF vaccines must be administered at an approved YF vaccination center. In the United States, state and territorial health departments have the authority to designate nonfederal vaccination centers and to issue YF vaccination stamps to those centers. A list of designated vaccine centers is available from CDC at http://wwwnc.cdc.gov/travel or can be obtained from state or local health departments.
As proof of receipt of YF vaccine, all vaccinees should possess a completed International Certificate of Vaccination or Prophylaxis (ICVP), validated with the provider's signature and official YF vaccination center stamp (Figure 3). An ICVP must be complete in every detail; if it is incomplete or inaccurate, it is not valid. Failure to secure validations can cause a traveler to be quarantined, denied entry, or possibly revaccinated at the point of entry to a country. Some countries do not require an ICVP for infants younger than a certain age (e.g. aged <6 months, <9 months, or <1 year, depending on the country). Information on age requirements for vaccination by individual countries is available at http://wwwnc.cdc.gov/travel.
The certificate of vaccination is valid beginning 10 days after the date of vaccination and extending for a period of 10 years. When a booster dose of the vaccine is administered within this 10-year period, the certificate is considered valid from the day of the most recent vaccination.
IHRs stipulate that a medical provider may issue a waiver of YF vaccination to a traveler if the provider judges that YF vaccination is medically contraindicated (see Contraindications and Precautions). In this case, the physician should fill out and sign the "Medical Contraindications to Vaccination" section of the ICVP (Figure 4), provide a signed and dated letter on letterhead stationary clearly stating the contraindication, and bearing that center's official YF vaccination stamp. The provider should inform the traveler of any increased risk for YF infection associated with nonvaccination and how to minimize this risk. Reasons other than medical contraindications are not acceptable for exemption from vaccination. The traveler also should be advised of the possibility that the medical waiver might not be accepted by the destination country.
Because requirements and recommendations might change, all travelers should seek up-to-date information before travel from health departments, CDC, and WHO. Travel agencies, international airlines, or shipping lines also might have up-to-date information. Updated information on requirements and recommendations for vaccination are available at http://wwwnc.cdc.gov/travel.
Administration of Yellow Fever Vaccine
Dosage and Administration
For persons of all ages for whom vaccination is indicated, a single subcutaneous injection of 0.5 mL of reconstituted vaccine is used. According to the package insert, the vaccine needs to be administered within 1 hour of reconstitution (71). Once reconstituted, a multidose vial should be maintained at 35°F--46°F (2°C--8°C), and the remaining doses should be used or discarded within 1 hour.
Although limited data suggest YF vaccine might retain its potency following the expiration date (126), the package insert states that the vaccine should not be used after its expiration date. According to ACIP's General Recommendations on Immunization, a dose of an expired vaccine should not be counted as valid, and the dose should be repeated with a nonexpired vaccine following a 28-day interval (127). For more guidance on the inadvertent administration of an expired vaccine, health-care providers should contact the vaccine manufacturer.
If the vaccine is administered inadvertently via the intramuscular route, the response to vaccine probably will not be affected. Repeating doses of vaccine administered by the intramuscular route rather than by the subcutaneous route is not necessary (127).
Anaphylaxis has been reported to occur in persons with no history of reactions to the components of the vaccine. Given this, all persons should be observed for at least 15 minutes following the administration of the vaccine, and epinephrine injection (1:1,000) should be readily available in case of a serious allergic reaction. Furthermore, because reactions have been delayed up to several hours following YF vaccine, all patients should be advised of signs and symptoms of an allergic reaction (e.g., urticaria, angioedema, rash, dyspnea, bronchospasm, pharyngeal edema, wheezing, and throat tightness). In addition, vaccinated persons should be advised to seek immediate medical care if any symptoms of an allergic reaction develop following vaccination.
Transfusion-related transmission of yellow fever vaccine virus has been documented among persons receiving blood products that were collected from recently vaccinated persons (75). Blood donation centers should screen blood donors to determine if they received a yellow fever vaccine within the preceding 2 weeks and defer these donors. However, practices vary between blood collection centers, and questions about vaccination history might not be asked routinely. Therefore, health-care providers should advise persons to defer blood donation for 2 weeks after receiving YF vaccine.
Booster Doses
IHRs require revaccination at intervals of 10 years to boost antibody titer. Evidence from multiple studies demonstrates that YF vaccine immunity persists for many decades and might provide life-long protection (79,80,128,129).
To minimize the occurrence of adverse events and optimize the immune response, efforts should be taken to observe a 10-year interval between YF vaccine doses. However, limited data suggest that revaccination occurring <10 years after the previous dose does not increase the risk for adverse events (74), particularly as serious adverse events occur primarily after the initial vaccination of YF vaccine. Although the level of YFV-specific antibodies formed following revaccination appears to be correlated inversely to the amount of preexisting antibodies (130), no data suggest that earlier revaccination will have a negative impact on seroprotection. If the date of the most recent vaccination cannot be ascertained, and the patient requires vaccination, a booster dose should be administered.
Simultaneous Administration of Other Vaccines or Drugs
Determination of whether to administer YF vaccine and other immunobiologics simultaneously (i.e., administration on the same day but at a different injection site) should be made on the basis of convenience to the traveler in completing the desired vaccinations before travel and on information regarding possible immune interference.
No evidence exists that inactivated vaccines and YF vaccine interfere with the immune response to the vaccine. Therefore, inactivated vaccines can be administered either simultaneously or at any time before or after YF vaccination (127). YF vaccine should be administered either simultaneously or 30 days apart from other live viral vaccines because the immune response to one live virus vaccine might be impaired if administered within 30 days of another live-virus vaccine (131,132). Limited data suggest that the serologic response to YF vaccine is not inhibited by administration of measles vaccine nonsimultaneously within 30 days of YF vaccine administration (133). However, the serologic response to measles vaccine was not assessed in this study. Oral Ty21a typhoid vaccine can be administered simultaneously or at any interval before or after YF vaccine attributable to the different routes of administration (34).
Vaccines that have been administered simultaneously with YF vaccine without interfering in the immune response to the vaccines or resulting in unusual safety profiles include Bacillius Calmette-Guerin (134), diphtheria (135,136), hepatitis A (137,138), hepatitis B (139), influenza (140), measles (133), meningococcal (Menomune) (141), pertussis (136), polio (135), smallpox (136,142), tetanus (135,136), and typhoid (both injectable [138] and oral [143,144]). No data exist regarding possible interference between YF vaccine and rabies, human papillomavirus, Japanese encephalitis, live attenuated influenza, or varicella virus vaccines (145).
Studies of persons administered YF vaccine and immune globulin simultaneously found no alteration of the immunologic response to YF vaccine when compared with controls (i.e., YF vaccine alone) (146,147). Although chloroquine inhibits replication of YFV in vitro, it does not affect antibody responses to YF vaccine adversely among persons receiving antimalarial prophylaxis (148).
No data exist for YF vaccine and the potential suppression of the tuberculin skin test (TST) response. However, because the use of live attenuated measles vaccine theoretically can suppress TST reactivity resulting in a false-negative reaction, ACIP's General Recommendations on Immunization suggest that TST should be administered at the same time as YF vaccine or 4 weeks after receipt of YF vaccine (127). Alternatively, TST screening can be performed and read before administering YF vaccine. However, if YF vaccine has been administered recently, TST screening should be delayed for at least 4 weeks after vaccination (127).
Contraindications
Allergy to Vaccine Components
YF vaccine is contraindicated for persons with a history of hypersensitivity to any of the vaccine components, including eggs, egg products, chicken proteins, or gelatin. The stopper used in vials of vaccine also contains dry latex rubber, which might cause an allergic reaction (71).
According to the manufacturer, persons who are able to eat eggs or egg products may receive the vaccine. However, potential hypersensitivity reactions might occur in persons with a history of minor reactions to eggs. For egg-sensitive persons, a scratch test or intradermal test can be performed before administering the vaccine to check for reactivity. If a person has a severe egg-sensitivity or has a positive skin test to the vaccine, but the vaccination is recommended because of their travel destination-specific risk, desensitization can be performed under direct supervision of a physician experienced in the management of anaphylaxis. The desensitization procedure is detailed in the product insert (71).
Given the risk for anaphylaxis, even among persons with no history of reactions to components of the vaccine, all persons should be observed for at least 15 minutes following the administration of the vaccine, and epinephrine (1:1,000) should be readily available in case of a serious allergic reaction. Vaccinated persons should be advised of symptoms of an allergic reaction and should be advised to seek immediate medical care if any symptoms of an allergic reaction develop following vaccination.
Infants Aged <6 Months
YF vaccine is contraindicated for infants aged <6 months. This contraindication was instituted in the late 1960s in response to a high rate of YEL-AND documented in vaccinated young infants (97). The mechanism of increased neurovirulence in infants is unknown but might be attributable to the immaturity of the blood-brain barrier, higher or more prolonged viremia, or immune system immaturity.
Altered Immune Status
Thymus Disorder
YF vaccine is contraindicated for persons with a thymus disorder that is associated with abnormal immune cell function (e.g., thymoma or myasthenia gravis) (149). Four (17%) of the initial 23 YEL-AVD reported cases were noted to occur in persons who had had thymectomies performed for thymomas (117). In 2003, thymus disorder was added to the YF vaccine package insert as a contraindication. To date, no evidence has been identified of immune dysfunction or increased risk for YF vaccine-associated serious adverse events in persons who have undergone incidental surgical removal of their thymus or have had indirect radiation therapy in the distant past. Therefore, YF vaccine can be administered, if indicated based on their destination-specific YF risk, in persons who underwent incidental surgical removal of the thymus or have a remote history of radiation therapy to the thymus.
AIDS and HIV Infection with Severe Immune Suppression
YF vaccine is contraindicated for persons with acquired immunodeficiency syndrome (AIDS) or other clinical manifestations of HIV (150,151), including persons with CD4 counts <200 per mm3 or <15% of total lymphocytes for children aged <6 years. This recommendation is based on a theoretic increased risk for encephalitis in this population. One fatal YEL-AND case has been reported in a person with a previously undiagnosed HIV infection and a CD4 count of 108 cells/mm3 who was vaccinated with YF vaccine (98). No large prospective, randomized trials have been performed to address the safety and efficacy of YF vaccine among this group adequately. Several retrospective and prospective studies that included a total of approximately 450 HIV-infected persons who received YF vaccine reported no additional serious adverse events. However, these studies included a limited subset of adults (n = 10) with a CD4 counts <200 per mm3 (84--87,98,152--155).
If travel to an area in which YF is endemic cannot be avoided in a person with severe immune suppression based on CD4 counts (<200 per mm3 or <15% total) or symptomatic HIV, a medical waiver should be provided, and counseling on protective measures against mosquito bites should be emphasized. (See Precautions for other HIV-infected persons not meeting these criteria.)
Immunodeficiencies Other than Thymus Disorder or HIV Infection
YF vaccine is contraindicated for persons with primary immunodeficiencies, malignant neoplasms, and transplantation (127). Although no data exist on the use of YF vaccine in these persons, they are presumed to have an increased risk for YF vaccine-associated serious adverse events. More specific information on primary immunodeficiencies for which the use of live viral vaccines, such as YF vaccine, is contraindicated will be published (127). In general, solid organ transplant or hematopoietic stem cell transplant recipients within 2 years of transplantation, or persons whose transplants occurred >2 years ago but who are still taking immunosuppressive drugs, are considered to be immunosuppressed (156). Live viral vaccines should be deferred in persons with a history of malignant neoplasm or transplantation until immune function has improved substantially. This is best determined by a physician who is familiar with the patient and the patient's underlying medical condition and treatments in consultation with a YF vaccination center. If a person with an immunodeficiency cannot avoid travel to an area in which YF is endemic, a medical waiver should be provided, and counseling on protective measures against mosquito bites should be emphasized.
Immunosuppressive and Immunomodulatory Therapies
YF vaccine is contraindicated for person whose immunologic response is either suppressed or modulated by current or recent radiation therapies or drugs. Drugs with known immunosuppressive or immunomodulatory properties include high-dose systemic corticosteroids, alkylating drugs, antimetabolites, TNF-α inhibitors (e.g., etanercept), IL-1 blocking agent (e.g., anakinra), and other monoclonal antibodies targeting immune cells (e.g., rituximab, alemtuzumab). No specific data exist on the use of YF vaccine in persons receiving these therapies. However, these persons are presumed to be at an increased risk for YF vaccine-associated serious adverse events, and the use of live attenuated vaccines in these persons is contraindicated according to the package insert for most of these therapies (127).
Although the immunosuppressive effects of corticosteroids can vary, a dose of either ≥2 mg/kg of body weight or a total ≥20 mg/day of prednisone or its equivalent for persons who weigh >10 kg when administered for ≥2 weeks is considered sufficiently immunosuppressive to contraindicate the use of live attenuated vaccines (127). Corticosteroids are not a contraindication when administration is under any of the following circumstances: short-term (i.e., <2 weeks); a low-to-moderate dose (<20 mg of prednisone or its equivalent per day); long-term, alternate-day treatment with short-acting preparations; maintenance physiologic doses (replacement therapy); or administered topically (skin or eyes), inhaled, or by intra-articular, bursal, or tendon injection (127).
Administration of live viral vaccines should be deferred in persons who have discontinued these therapies until immune function has improved. This is best determined by a physician who is familiar with the patient's underlying medical conditions and by the patient's pharmacist, who can assist in determining the specific half-lives of the immunosuppressive drugs and the potential duration of immune suppression. If someone receiving immunosuppressive or immunomodulatory therapies cannot avoid travel to an area in which YF is endemic , a medical waiver should be provided, and counseling on protective measures against mosquito bites should be emphasized.
Precautions
Infants Aged 6--8 Months
Age 6--8 months is a precaution for YF vaccine administration. In infants aged <6 months, the rates of YEL-AND are substantially elevated (50--400 cases per 100,000 infants vaccinated) (96,106). Two cases of YEL-AND have been reported among infants aged 6--8 months (96). By age 9 months, the risk for YEL-AND is believed to be substantially lower. In general, whenever possible, travel of children aged 6--8 months to countries in which YF is endemic should be postponed or avoided. If travel is unavoidable, the decision of whether to vaccinate these infants needs to balance the risks for YFV exposure with the risk for adverse events following vaccination.
Adults Aged ≥60 Years
Age ≥60 years is a precaution for YF vaccine administration, particularly if the first dose of the YF vaccine is to be administered. A recent analysis of adverse events reported to VAERS during 2000--2006 indicates that persons aged ≥60 years are at increased risk for any serious adverse events after vaccination, compared with younger persons (89). The rate of serious adverse events in persons aged ≥60 years was 8.3 events per 100,000 doses distributed compared with 4.7 events per 100,000 doses distributed for all YF vaccine recipients. This reinforces findings from VAERS data from 1990--1998 that reported the rate of serious adverse events was 7.5 times higher in persons aged ≥60 years than in the reference group (persons aged 19--29 years) (107). The risk for YEL-AND and YEL-AVD also are increased in this age group, at 1.8 and 1.4 reported cases per 100,000 doses distributed, respectively, compared with 0.8 and 0.4 reported cases per 100,000 doses distributed for all YF vaccine recipients. Given that YEL-AND and YEL-AVD are seen almost exclusively in primary vaccine recipients, caution should be exercised with older travelers who might be receiving YF vaccine for the first time. If travel is unavoidable, the decision to vaccinate travelers aged ≥60 years needs to weigh the risks and benefits of the vaccination in the context of their destination-specific risk for exposure to YFV.
Asymptomatic HIV Infection with Moderate Immune Suppression
Asymptomatic HIV infection with moderate immune suppression (i.e., CD4 counts of 200--499 per mm3 for persons aged ≥6 years or 15%--24% of total lymphocytes for children aged <6 years) is a precaution for YF vaccine administration. Large prospective randomized trials have not been performed to address the safety and efficacy of YF vaccine adequately among this group. Several retrospective and prospective studies including approximately 450 persons infected with HIV have reported no serious adverse events among patients considered moderately immunosuppressed based on the their CD4 counts (84--87,152--155).
HIV infection has been associated with a reduced immunologic response to a number of inactivated and live-attenuated vaccines, including YF vaccine (84). In a recent retrospective cohort study, 65 (83%) of 78 HIV-infected persons developed specific antibodies against YFV in the first year after vaccination; however, this was significantly lower than vaccinated persons without HIV infection (97% [64/66]; p=0.01) (86). Among HIV-infected infants in developing nations, only three (17%) of 18 developed YFV-specific neutralizing antibodies within 10 months of vaccination compared with 42 (74%) of 57 HIV-uninfected controls matched for age and nutritional status (87). The mechanisms for the diminished immune response in HIV-infected persons are uncertain but appear to be correlated with HIV RNA levels and CD4 counts. Because vaccination of asymptomatic HIV-infected persons might be less effective than that for persons not infected with HIV, measurement of their neutralizing antibody response to vaccination should be considered before travel. Health-care providers should contact the appropriate state health department or CDC (at telephone 1-970 221-6400) to discuss serologic testing further.
For HIV-infected persons who experience immune reconstitution in response to antiretroviral therapy, their current CD4 count and symptoms of HIV infection (if stable over 3 month time period) rather than a CD4 count nadir and a history of prior opportunistic infections/symptomatic HIV should be used to categorize their HIV status (156). Of note, one YEL-AVD case was reported in a person determined to have a genetic polymorphisms in chemokine receptor CCR5 (118). Consequently, concern has been raised that despite having adequate immune function, persons with HIV infection who are receiving an antiretroviral regimen containing a CCR5-receptor antagonist could be at increased risk for adverse events after YF vaccination (157). Further research is needed to address this concern.
If an asymptomatic HIV-infected person with moderate immune suppression is traveling to an area in which YF is endemic, vaccination can be considered. Vaccinated persons should be monitored closely after vaccination for evidence of adverse events. If an adverse event occurs, a VAERS report should be filed, and the state health department or CDC should be notified to obtain technical support and assistance with diagnostic testing. If international travel requirements rather than an increased risk for acquiring YFV infection are the only reason to vaccinate HIV-infected persons, the person should be excused from vaccination and issued a medical waiver to fulfill health regulations. If an asymptomatic HIV-infected person has no evidence of immune suppression based on CD4 counts (CD4 count ≥500 per mm3 or ≥25% of total lymphocytes for children aged <6 years), YF vaccine can be administered, if recommended on the basis of their destination-specific YF risk. (See Contraindications for other HIV-infected persons not meeting these criteria.)
Pregnancy
Pregnancy is a precaution for YF vaccine administration, compared with most other live vaccines, which are contraindicated in pregnancy. If travel is unavoidable, and the risks for YFV exposure are felt to outweigh the vaccination risks, a pregnant woman should be vaccinated. If the risks for vaccination are felt to outweigh the risks for YFV exposure, pregnant women should be issued a medical waiver to fulfill health regulations. Although no specific data are available, a woman should wait 4 weeks after receiving YF vaccine before conceiving. (See Vaccination of Women During Pregnancy and Breastfeeding.)
Breastfeeding
Breastfeeding is a precaution for YF vaccine administration. Two serious adverse events have been reported in exclusively breastfed infants whose mothers were vaccinated with YF vaccine (123; PHAC, unpublished data, 2008). Further research is needed to document the risk for potential vaccine exposure through breastfeeding. Until more information is available, YF vaccine should be avoided in breastfeeding women. However, when travel of nursing mothers to a YF endemic area cannot be avoided or postponed, these women should be vaccinated. (See Vaccination of Women During Pregnancy and Breastfeeding.)
Special Populations
Age
YF vaccine is approved for use in persons aged ≥9 months. The vaccine is contraindicated for infants aged <6 months. (See Contraindications for more information.) Age 6--8 months and adults aged ≥60 years are precautions for YF vaccine administration. (See Precautions.)
Pregnancy
Limited data are available regarding the safety and immunogenicity of YF vaccine in pregnancy. Pregnancy is a precaution for YF vaccine administration. (See Vaccination of Women During Pregnancy and Breastfeeding and Precautions.)
Breastfeeding Women
Although no data are available on the immune response in breastfeeding mothers, no alteration to the immune response in these women are suspected. Limited data are available regarding the safety of YF vaccine in breastfed infants. Breastfeeding is a precaution for YF vaccine administration. (See Vaccination of Women During Pregnancy and Breastfeeding and Precautions.)
Altered Immune Status
The following conditions are a contraindication for YF vaccine administration: 1) thymus disorders associated with abnormal immune cell function, 2) symptomatic HIV-infection or CD4+ T-lymphocyte values <200 per mm3 or <15% of total lymphocytes for children aged <6 years, 3) primary immunodeficiencies, 4) malignant neoplasms, 5) transplantation, and 6) immune suppression or modulation attributable to current or recent radiation therapies or drugs. The following condition with altered immune status is a precaution for YF vaccine administration: asymptomatic HIV-infection with CD4+ T-lymphocyte values 200--499 per mm3 or 15%--24% of total lymphocytes for children aged < 6 years. (See Contraindications and Precautions.)
Complete details are not available regarding what medications the nine patients with reported YEL-AVD who had a history of autoimmune disease or diseases with potential autoimmune etiology were taking; these might have included immunosuppressive medications. Four of the nine patients were aged >60 years, including two who also had a history of thymectomy for thymoma (see YEL-AVD). Older age and history of thymoma are both risk factors for YEL-AVD. Nonetheless, the fact that nine (16%) of 57 persons with reported cases of YEL-AVD had a history of autoimmune disease is a concern and suggests that autoimmune disease, either by itself of in conjunction with other risk factors, including immunosuppressive medication, could increase the risk for YEL-AVD. Definitive data are lacking to guide decision-making about YF vaccination in these patients. Health-care providers should consider the possibility of diminished immune function resulting from the autoimmune disease state itself and/or the medication used in deciding whether or not to administer YF vaccine to these patients.
Reporting of Vaccine Adverse Events
Even if a causal relation to vaccination is not certain, all adverse events following receipt of vaccine that are thought to be clinically significant by health-care providers or vaccine recipients should be reported to VAERS at http://vaers.hhs.gov or by telephone at 1-800-822-7967. To promote better timeliness and quality of safety data, secure web-based reporting is available and health-care providers are encouraged to report electronically at https://vaers.hhs.gov/esub/step1.
Surveillance to monitor, characterize, and quantify YF vaccine-specific adverse outcomes are ongoing. Because current reporting mechanisms are strictly passive, detection of of YEL-AVD and YEL-AND in recently vaccinated persons who experience new symptoms largely depends on health-care providers being familiar with these conditions and with reporting requirements. An education program regarding the vaccine and possible serious adverse events is being developed and will be made available to all health-care providers by fall 2010 through CDC's Travelers' Health website. State health departments are encouraged to incorporate requirements for completion of these programs into their certification and recertification processes for issuance of YF vaccination stamps to health-care providers.
Future Research
Research focusing on suspected risk factors for serious adverse events or suboptimal response to YF vaccine for which few or no data exist should be designed and implemented. This should include HIV, immunosuppressive diseases, autoimmune diseases, and use of immunosuppressive and immunomodulatory drugs. Ethical considerations might limit these to observational studies. If possible, studies geared to collecting population-based data would be the most useful, though also the most difficult to implement. Further research also is needed to determine if YF vaccine virus is present in breast milk and the exact route of transmission involved (e.g., ingested milk or blood).
Studies addressing the duration of immune response to the vaccine, in particular the duration of IgM antibodies following vaccination and the duration of protective antibody levels, are warranted. These studies would help resolve issues related to possible YF disease in vaccinated travelers as well as provide updated evidence on the dosing interval needed for booster doses of the vaccine. Finally, studies examining the immune response to an inactivated yellow fever vaccine are underway. If successful, the inactivated vaccine potentially could be administered to persons with contraindications to the use of a live viral vaccine.
Additional Information
Information about YF for travelers and health-care providers has been published previously (34). Additional information about YF is available at http://www.cdc.gov/ncidod/dvbid/yellowfever/index.html. Additional licensure information for the YF vaccine that is approved in the United States is available at http://www.fda.gov/BiologicsBloodVaccines/Vaccines/ApprovedProducts/ucm094074.htm.
Acknowledgments
This report is based in part on contributions by Nicole Lindsey, MPH, Susan Hills, MBBS, Division of Vector-Borne Diseases, National Center for Emerging and Zoonotic Infectious Diseases, CDC.
References
- Monath T, Cetron MS, Teuwen DE. Yellow fever vaccine. In: Plotkin SA, Orenstein WA, Offit PA, eds. Vaccines. 5th ed. Philadelphia, PA: Saunders Elsevier; 2008:959--1055.
- Reed W, Carroll JAS, Agramonte A. The etiology of yellow fever: an additional note. JAMA 1901;36:431--40.
- Berry GP, Kitchen SF. Yellow fever accidentally contracted in the laboratory: a study of seven cases. Am J Trop Med Hyg 1931;11:365--434.
- Beeuwkes H. Clinical manifestations of yellow fever in the west African native as observed during four extensive epidemics of the disease in the Gold Coast and Nigeria. Trans R Soc Trop Med Hyg 1936;30:61--86.
- Barrett AD, Monath TP. Epidemiology and ecology of yellow fever virus. Adv Virus Res 2003;61:291--315.
- World Health Organization, Division of Epidemiological Surveillance and Health Situation Trend Assessment. Global health situation and projections---estimates. Geneva, Switzerland: World Health Organization; 1992.
- Robertson SE, Hull BP, Tomori O, Bele O, LeDuc JW, Esteves K. Yellow fever: a decade of reemergence. JAMA 1996;276:1157--62.
- Chippaux A, Deubel V, Moreau JP, Reynes JM. [Current situation of yellow fever in Latin America]. Bull Soc Pathol Exot 1993;86:460--4.
- Kuno G, Chang GJ, Tsuchiya KR, Karabatsos N, Cropp CB. Phylogeny of the genus Flavivirus. J Virol 1998;72:73--83.
- Simpson DI, Haddow AJ, Williams MC, Woodall JP. Yellow fever in central Uganda, 1964. IV. Investigations on blood-sucking diptera and monkeys. Trans R Soc Trop Med Hyg 1965;59:449--58.
- Germain M, Cornet M, Mouchet J, et al. [Sylvatic yellow fever in Africa recent advances and present approach]. Med Trop (Mars) 1981;41:31--43.
- Bauer JH. Transmission of yellow fever by mosquitoes other than Aedes aegypti. Am J Trop Med Hyg 1928;8:261--82.
- Shannon RC, Whitman L, Franca M. Yellow fever virus in jungle mosquitoes. Science 1938;88:110--1.
- Vasconcelos PF, Rodrigues SG, Degallier N, et al. An epidemic of sylvatic yellow fever in the southeast region of Maranhao State, Brazil, 1993--1994: epidemiologic and entomologic findings. Am J Trop Med Hyg 1997;57:132--7.
- Soper FL. The newer epidemiology of yellow fever. Am J Public Health 1937;27:1--14.
- Digoutte JP, Cornet M, Deuble V, Downs WG. Yellow fever. In: Porterfield JS, ed. Exotic viral infections: Kass handbook of infectious diseases. London, UK: Chapman & Hall Medical; 1995: 67--102.
- Monath TP. Yellow fever: an update. Lancet Infect Dis 2001;1:11--20.
- Hindle E. The transmission of yellow fever. Lancet 1930;216:835--42.
- Tomori O. Yellow fever: the recurring plague. Crit Rev Clin Lab Sci 2004;41:391--427.
- Van der Stuyft P, Gianella A, Pirard M, et al. Urbanisation of yellow fever in Santa Cruz, Bolivia. Lancet 1999;353:1558--62.
- Pan American Health Organization. Outbreak of yellow fever in Paraguay. Epidemiol Bull 2008;27.
- Ellis BR, Barrett AD. The enigma of yellow fever in East Africa. Rev Med Virol 2008;18:331--46.
- Mutebi JP, Barrett AD. The epidemiology of yellow fever in Africa. Microbes Infect 2002;4:1459--68.
- Monath TP, Craven RB, Adjukiewicz A, et al. Yellow fever in the Gambia, 1978--1979: epidemiologic aspects with observations on the occurrence of Orungo virus infections. Am J Trop Med Hyg 1980;29:912--28.
- Nasidi A, Monath TP, DeCock K, et al. Urban yellow fever epidemic in western Nigeria, 1987. Trans R Soc Trop Med Hyg 1989;83:401--6.
- World Health Organization. Outbreak news: yellow fever, Republic of the Congo. Wkly Epidemiol Rec 2009;84:161.
- World Health Organization. Yellow fever in the Central African Republic, 2009. Available at http://www.who.int/csr/don/2009_12_01/en/print.html. Accessed July 1, 2010.
- Pan American Health Organization. Yellow fever in the Americas. Bull Pan Am Health Organ 1985;19:209--12.
- Vasconcelos PF. [Yellow Fever]. Rev Soc Bras Med Trop 2003;36:275--93.
- CDC. Outbreak notice: yellow fever in Brazil 2009. Available at http://wwwnc.cdc.gov/travel/content/outbreak-notice/yellow-fever-brazil.aspx. Accessed July 1, 2010.
- Pan American Health Organization. Update: situation of yellow fever in the Americas 2009. Available at http://new.paho.org/hq/index.php?option=com_content&task=view&id=568&Itemid=259. Accessed July 1, 2010.
- Brazil Ministry of Health. [Yellow fever situation. Brazil, 2007--2008]. Available at http://portal.saude.gov.br/portal/arquivos/pdf/boletim_svs_febre_amarela_040408.pdf. Accessed July 1, 2010.
- Gubler DJ. The global emergence/resurgence of arboviral diseases as public health problems. Arch Med Res 2002;33:330--42.
- Gershman M, Schroeder B, Staples JE. Yellow fever. In: Brunette GW, Kozarsky PE, Magill AJ, Shlim DR, Whatley AD, eds. CDC health information for international travel 2010. Atlanta, GA: US Department of Health and Human Services, Public Health Service, CDC; 2009.
- World Health Organization. Yellow fever in Africa and South America, 2007. Wkly Epidemiol Rec 2009;84:97--104.
- World Health Organization. Yellow fever in Africa and South America, 2006. Wkly Epidemiol Rec 2008;83:60--76.
- World Health Organization. Yellow fever situation in Africa and South America, 2005. Wkly Epidemiol Rec 2006;81:317--24.
- Monath TP, Wilson DC, Lee VH, Stroh G, Kuteyi K, Smith EA. The 1970 yellow fever epidemic in Okwoga District, Benue Plateau State, Nigeria. 1. Epidemiological observations. Bull World Health Organ 1973;49:113--21.
- Vasconcelos PF, Costa ZG, Travassos Da Rosa ES, et al. Epidemic of jungle yellow fever in Brazil, 2000: implications of climatic alterations in disease spread. J Med Virol 2001;65:598--604.
- Monath TP, Cetron MS. Prevention of yellow fever in persons traveling to the tropics. Clin Infect Dis 2002;34:1369--78.
- Beeuwkes H, Kerr JA, Wethersbee AA. Observations on the bionomics and comparative prevalence of the vectors of yellow fever and other domestic mosquitoes of West Africa, and the epidemiological significance of seasonal variations. Trans R Soc Trop Med Hyg 1933;26:425--47.
- Pinheiro FP, Travassos da Rosa AP, Moraes MA, Almeida Neto JC, Camargo S, Filgueiras JP. An epidemic of yellow fever in central Brazil, 1972--1973. I. Epidemiological studies. Am J Trop Med Hyg 1978;27:125--32.
- Tuboi SH, Costa ZG, da Costa Vasconcelos PF, Hatch D. Clinical and epidemiological characteristics of yellow fever in Brazil: analysis of reported cases 1998--2002. Trans R Soc Trop Med Hyg 2007;101:169--75.
- Monath TP. Yellow fever: a medically neglected disease. Rev Infect Dis 1987;9:165--75.
- Monath TP. Yellow fever. In: Monath TP, ed. The arboviruses: epidemiology and ecology. Boca Raton, FL: CRC Press; 1989.
- Santos F, Pereira Lima C, Paiva M, Caosta e Silva M, Nery de Castro C. Disseminated intravascular coagulation in acute yellow fever: measurement of coagulation factors. Brasilia Med 1973;9:9--15.
- Oudart JL, Rey M. [Proteinuria, proteinemia and transaminases in 23 cases of confirmed yellow fever]. Bull World Health Organ 1970;42:95--102.
- Vasconcelos PF, Luna EJ, Galler R, et al. Serious adverse events associated with yellow fever 17DD vaccine in Brazil: a report of two cases. Lancet 2001;358:91--7.
- American Academy of Pediatrics. Arboviruses. In: Pickering LK, Baker CJ, Kimberlin DW, Long SS, eds. Red book: 2009 report of the Committee on Infectious Diseases. 28th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2009.
- Saluzzo JF, Monath TP, Cornet M, Deubel V, Digoutte JP. [Comparison of different methods used to detect yellow fever virus in human samples and mosquito pools: advantages of an ELISA method for rapid detection]. Ann Inst Pasteur (Paris) 1985;136:115--29.
- Paddock CD, Nicholson WL, Bhatnagar J, et al. Fatal hemorrhagic fever caused by West Nile virus in the United States. Clin Infect Dis 2006;42:1527--35.
- Monath TP. Treatment of yellow fever. Antiviral Res 2008;78:116--24.
- Kirk R. Epidemic of yellow fever in Nuba Mountains, Anglo-Egyptian Sudan. Ann Trop Med 1941;35:67--113.
- McFarland JM, Baddour LM, Nelson JE, et al. Imported yellow fever in a United States citizen. Clin Infect Dis 1997;25:1143--7.
- Digoutte JP, Plassart H, Salaun JJ, Heme G, Ferrara L, Germain M. [3 cases of yellow fever contracted in Senegal]. Bull World Health Organ 1981;59:759--66.
- Nolla-Salas J, Saballs-Radresa J, Bada JL. Imported yellow fever in vaccinated tourist. Lancet 1989;334:1275.
- Rodhain F, Hannoun C, Jousset FX, Ravisse P. [Isolation of the yellow fever virus in Paris from 2 imported human cases]. Bull Soc Pathol Exot Filiales 1979;72:411--5.
- CDC. Fatal yellow fever in a traveler returning from Venezuela, 1999. MMWR 1999;49:303--5.
- Colebunders R, Mariage JL, Coche JC, et al. A Belgian traveler who acquired yellow fever in the Gambia. Clin Infect Dis 2002;35:e113--6.
- CDC. Fatal yellow fever in a traveler returning from Amazonas, Brazil, 2002. MMWR 2002;51:324--5.
- Teichmann D, Grobusch MP, Wesselmann H, et al. A haemorrhagic fever from the Cote d'Ivoire. Lancet 1999;354:1608.
- World Health Organization. Yellow fever 1996--1997. Part 1. Wkly Epidemiol Rec 1998;73:354--9.
- World Health Organization. Imported case of yellow fever in the Netherlands, 2000. Available at http://www.who.int/csr/don/2000_02_25/en. Accessed July 1, 2010.
- Lloyd W, Theiler M, Ricci NI. Modification of the virulence of yellow fever virus by cultivation in tissues in vitro. Trans R Soc Trop Med Hyg 1936;29:481--529.
- Pugachev KV, Ocran SW, Guirakhoo F, Furby D, Monath TP. Heterogeneous nature of the genome of the ARILVAX yellow fever 17D vaccine revealed by consensus sequencing. Vaccine 2002;20:996--9.
- Monath TP, Nichols R, Archambault WT, et al. Comparative safety and immunogenicity of two yellow fever 17D vaccines (ARILVAX and YF-VAX) in a phase III multicenter, double-blind clinical trial. Am J Trop Med Hyg 2002;66:533--41.
- Ripoll C, Ponce A, Wilson MM, et al. Evaluation of two yellow fever vaccines for routine immunization programs in Argentina. Hum Vaccin 2008;4:121--6.
- Pfister M, Kursteiner O, Hilfiker H, et al. Immunogenicity and safety of BERNA-YF compared with two other 17D yellow fever vaccines in a phase 3 clinical trial. Am J Trop Med Hyg 2005;72:339--46.
- Lang J, Zuckerman J, Clarke P, Barrett P, Kirkpatrick C, Blondeau C. Comparison of the immunogenicity and safety of two 17D yellow fever vaccines. Am J Trop Med Hyg 1999;60:1045--50.
- Camacho LA, Freire MS, Leal ML, et al. Immunogenicity of WHO-17D and Brazilian 17DD yellow fever vaccines: a randomized trial. Rev Saude Publica 2004;38:671--8.
- sanofi pasteur. Yellow fever vaccine. YF-VAX.[ Product insert]. Swiftwater, PA: sanofi pasteur; 2005. Available at https://www.vaxserve.com/image.cfm?doc_id=5991&image_type=product_pdf. Accessed July 1, 2010.
- Mason RA, Tauraso NM, Spertzel RO, Ginn RK. Yellow fever vaccine: direct challenge of monkeys given graded doses of 17D vaccine. Appl Microbiol 1973;25:539--44.
- Reinhardt B, Jaspert R, Niedrig M, Kostner C, L'Age-Stehr J. Development of viremia and humoral and cellular parameters of immune activation after vaccination with yellow fever virus strain 17D: a model of human flavivirus infection. J Med Virol 1998;56:159--67.
- Monath TP, McCarthy K, Bedford P, et al. Clinical proof of principle for ChimeriVax: recombinant live, attenuated vaccines against flavivirus infections. Vaccine 2002;20:1004--18.
- CDC. Transfusion-related transmission of yellow fever vaccine virus---California, 2009. MMWR 2010; 59:34--7.
- Monath TP. Neutralizing antibody responses in the major immunoglobulin classes to yellow fever 17D vaccination of humans. Am J Epidemiol 1971;93:122--9.
- Courtois G. Time of appearance and duration of immunity conferred by 17D vaccine. In: Yellow fever vaccine. WHO Series. Geneva, Switzerland: World Health Organization; 1956.
- World Health Organization. International Health Regulations (2005). Geneva, Switzerland; 2008. Available at http://whqlibdoc.who.int/publications/2008/9789241580410_eng.pdf. Accessed July 1, 2010.
- Rosenzweig EC, Babione RW, Wisseman CL, Jr. Immunological studies with group B arthropod-borne viruses. IV. Persistence of yellow fever antibodies following vaccination with 17D strain yellow fever vaccine. Am J Trop Med Hyg 1963;12:230--5.
- Poland JD, Calisher CH, Monath TP, Downs WG, Murphy K. Persistence of neutralizing antibody 30--35 years after immunization with 17D yellow fever vaccine. Bull World Health Organ 1981;59:895--900.
- Bonnevie-Nielsen V, Heron I, Monath TP, Calisher CH. Lymphocytic 2',5'-oligoadenylate synthetase activity increases prior to the appearance of neutralizing antibodies and immunoglobulin M and immunoglobulin G antibodies after primary and secondary immunization with yellow fever vaccine. Clin Diagn Lab Immunol 1995;2:302--6.
- Nasidi A, Monath TP, Vandenberg J, et al. Yellow fever vaccination and pregnancy: a four-year prospective study. Trans R Soc Trop Med Hyg 1993;87:337--9.
- Suzano CE, Amaral E, Sato HK, Papaiordanou PM. The effects of yellow fever immunization (17DD) inadvertently used in early pregnancy during a mass campaign in Brazil. Vaccine 2006;24:1421--6.
- Goujon C, Tohr M, Feuillie V, Coulaud P, Dupont B, Sansonetti P. Good tolerance and efficacy of yellow fever vaccine among carriers of human immunodeficiency virus. Fourth International Conference on Travel Medicine; April 23--27, 1995; Acapulco, Mexico.
- Receveur MC, Thiebaut R, Vedy S, Malvy D, Mercie P, Bras ML. Yellow fever vaccination of human immunodeficiency virus-infected patients: report of 2 cases. Clin Infect Dis 2000;31:E7--8.
- Veit O, Niedrig M, Chapuis-Taillard C, et al. Immunogenicity and safety of yellow fever vaccination for 102 HIV-infected patients. Clin Infect Dis 2009;48:659--66.
- Sibailly TS, Wiktor SZ, Tsai TF, et al. Poor antibody response to yellow fever vaccination in children infected with human immunodeficiency virus type 1. Pediatr Infect Dis J 1997;16:1177--9.
- Camacho LA, de Aguiar SG, Freire MS, et al. Reactogenicity of yellow fever vaccines in a randomized, placebo-controlled trial. Rev Saude Publica 2005;39:413--20.
- Lindsey NP, Schroeder BA, Miller ER, et al. Adverse event reports following yellow fever vaccination. Vaccine 2008;26:6077--82.
- Kelso JM, Mootrey GT, Tsai TF. Anaphylaxis from yellow fever vaccine. J Allergy Clin Immunol 1999;103:698--701.
- McMahon AW, Eidex RB, Marfin AA, et al. Neurologic disease associated with 17D-204 yellow fever vaccination: a report of 15 cases. Vaccine 2007;25:1727--34.
- Kitchener S. Viscerotropic and neurotropic disease following vaccination with the 17D yellow fever vaccine, ARILVAX. Vaccine 2004;22:2103--5.
- Fox JP, Penna HA. Behavior of 17D yellow fever virus in Rhesus monkeys: relation to substrain, dose, and neural, or extraneural innoculaton. Am J Hyg 1943;38:152--72.
- Nickells J, Cannella M, Droll DA, Liang Y, Wold WS, Chambers TJ. Neuroadapted yellow fever virus strain 17D: a charged locus in domain III of the E protein governs heparin binding activity and neuroinvasiveness in the SCID mouse model. J Virol 2008;82:12510--9.
- World Health Organization Expert Committee on Biological Standardization. Forty-sixth report. WHO technical report series, 1998;872:1--90.
- Stuart G. Reactions following vaccination against yellow fever. In: Yellow fever vaccine. WHO series. Geneva, Switzerland: World Health Organization; 1956.
- Public Health Service. Yellow fever vaccine: recommendation of the U.S. Public Health Service Advisory Committee on Immunization Practices. Ann Intern Med 1969;71:365--7.
- Kengsakul K, Sathirapongsasuti K, Punyagupta S. Fatal myeloencephalitis following yellow fever vaccination in a case with HIV infection. J Med Assoc Thai 2002;85:131--4.
- CDC. Adverse events associated with 17D-derived yellow fever vaccination---United States, 2001--2002. MMWR 2002;51:989--93.
- Chaves M, Riccio P, Patrucco L, Rojas J, Cristiano E. Longitudinal myelitis associated with yellow fever vaccination. J Neurovirol 2009;3:1--3.
- Miravalle A, Biller J, Silva E, Conneely M, O'Keefe P. Acute disseminated encephalomyelitis: yellow fever vaccination. Arq Neuro Psiquiatr 2009;67:710--1.
- Vucic S, Kiernan MC, Cornblath DR. Guillain-Barré syndrome: an update. J Clin Neurosci 2009;16 :733--41.
- Noorbakhsh F, Johnson RT, Emery D, Power C. Acute disseminated encephalomyelitis: clinical and pathogenesis features. Neurol Clin 2008;26:759--80.
- Anonymous. Fatal viral encephalitis following 17D yellow fever vaccine inoculation. Report of a case in a 3-year-old child. JAMA 1966;198:671--2.
- Jennings AD, Gibson CA, Miller BR, et al. Analysis of a yellow fever virus isolated from a fatal case of vaccine-associated human encephalitis. J Infect Dis 1994;169:512--8.
- Louis JJ, Chopard P, Larbre F. [A case of encephalitis after anti-yellow fever vaccination with the 17 D strain]. Pediatrie 1981;36:547--50.
- Khromava AY, Eidex RB, Weld LH, et al. Yellow fever vaccine: an updated assessment of advanced age as a risk factor for serious adverse events. Vaccine 2005;23:3256--63.
- Martin M, Tsai TF, Cropp B, et al. Fever and multisystem organ failure associated with 17D-204 yellow fever vaccination: a report of four cases. Lancet 2001;358:98--104.
- Chan RC, Penney DJ, Little D, Carter IW, Roberts JA, Rawlinson WD. Hepatitis and death following vaccination with 17D-204 yellow fever vaccine. Lancet 2001;358:121--2.
- CDC. Fever, jaundice, and multiple organ system failure associated with 17D-derived yellow fever vaccination, 1996--2001. MMWR 2001;50:643--5.
- Adhiyaman V, Oke A, Cefai C. Effects of yellow fever vaccination. Lancet 2001;358:1907--8.
- Troillet N, Laurencet F. Effects of yellow fever vaccination. Lancet 2001;358:1908--9.
- Werfel U, Popp W. Effects of yellow fever vaccination. Lancet 2001;358:1909.
- Belsher JL, Gay P, Brinton M, et al. Fatal multiorgan failure due to yellow fever vaccine-associated viscerotropic disease. Vaccine 2007;25:8480--5.
- Struchiner CJ, Luz PM, Dourado I, et al. Risk of fatal adverse events associated with 17DD yellow fever vaccine. Epidemiol Infect 2004;132:939--46.
- Whittembury A, Ramirez G, Hernandez H, et al. Viscerotropic disease following yellow fever vaccination in Peru. Vaccine 2009;27:5974--81.
- Barwick R. History of thymoma and yellow fever vaccination. Lancet 2004;364:936.
- Pulendran B, Miller J, Querec TD, et al. Case of yellow fever vaccine--associated viscerotropic disease with prolonged viremia, robust adaptive immune responses, and polymorphisms in CCR5 and RANTES genes. J Infect Dis 2008;198:500--7.
- Hayes EB. Acute viscerotropic disease following vaccination against yellow fever. Trans R Soc Trop Med Hyg 2007;101:967--71.
- Tsai TF, Paul R, Lynberg MC, Letson GW. Congenital yellow fever virus infection after immunization in pregnancy. J Infect Dis 1993;168:1520--3.
- Cavalcanti DP, Salomao MA, Lopez-Camelo J, Pessoto MA. Early exposure to yellow fever vaccine during pregnancy. Trop Med Int Health 2007;12:833--7.
- Nishioka SA, Nunes-Araujo FR, Pires WP, Silva FA, Costa HL. Yellow fever vaccination during pregnancy and spontaneous abortion: a case-control study. Trop Med Int Health 1998;3:29--33.
- CDC. Transmission of yellow fever vaccine virus through breast-feeding---Brazil, 2009. MMWR 2010;59:130--2.
- Theiler RN, Rasmussen SA, Treadwell TA, Jamieson DJ. Emerging and zoonotic infections in women. Infect Dis Clin North Am 2008;22:755--72.
- CDC. Possible West Nile virus transmission to an infant through breast-feeding---Michigan, 2002. MMWR 2002;51:877--8.
- Allen KW, Nguyen-Van-Tam JS, Howells J. Administration of time-expired yellow fever vaccine: public health response and results of a serological investigation. Commun Dis Pub Health 1999;2:141--2.
- CDC. General recommendations on immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR. In press.
- Groot H, Riberiro RB. Neutralizing and haemagglutination-inhibiting antibodies to yellow fever 17 years after vaccination with 17D vaccine. Bull World Health Organ 1962;27:699--707.
- Niedrig M, Lademann M, Emmerich P, Lafrenz M. Assessment of IgG antibodies against yellow fever virus after vaccination with 17D by different assays: neutralization test, haemagglutination inhibition test, immunofluorescence assay and ELISA. Trop Med Int Health 1999;4:867--71.
- Hepburn MJ, Kortepeter MG, Pittman PR, et al. Neutralizing antibody response to booster vaccination with the 17D yellow fever vaccine. Vaccine 2006;24:2843--9.
- Petralli JK, Merigan TC, Wilbur JR. Circulating interferon after measles vaccination. N Engl J Med 1965;273:198--201.
- Petralli JK, Merigan TC, Wilbur JR. Action of endogenous interferon against vaccinia infection in children. Lancet 1965;2:401--5.
- Stefano I, Sato HK, Pannuti CS, et al. Recent immunization against measles does not interfere with the sero-response to yellow fever vaccine. Vaccine 1999;17:1042--6.
- Gateff C, Relyveld EH, Le Gonidec G, et al. [Study of a new pentavalent vaccine combination]. Ann Microbiol (Paris) 1973;124:387--409.
- Wolga J, Rodhain F, Hannoun C, et al. [Evaluation of thermostable yellow fever vaccine from the Pasteur Institute on international travellers]. J Biol Stand 1986;14:289--95.
- Ruben FL, Smith EA, Foster SO, et al. Simultaneous administration of smallpox, measles, yellow fever, and diphtheria-pertussis-tetanus antigens to Nigerian children. Bull World Health Organ 1973;48:175--81.
- Dumas R, Forrat R, Lang J, Farinelli T, Loutan L. Safety and immunogenicity of a new inactivated hepatitis A vaccine in concurrent administration with a typhoid fever vaccine or a typhoid fever + yellow fever vaccine. Adv Ther 1997;14:160--7.
- Jong EC, Kaplan KM, Eves KA, Taddeo CA, Lakkis HD, Kuter BJ. An open randomized study of inactivated hepatitis A vaccine administered concomitantly with typhoid fever and yellow fever vaccines. J Travel Med 2002;9:66--70.
- Yvonnet B, Coursaget P, Deubel V, Diop-Mar I, Digoutte JP, Chiron JP. Simultaneous administration of hepatitis B and yellow fever vaccines. J Med Virol 1986;19:307--11.
- Goullin B, Masseron T, Teyssou R, et al. [Effectiveness of simultaneous vaccination against influenza and yellow fever]. Med Arm 1993;21:115--7.
- Dukes C, Froeschle J, George J, et al. Safety and immunogenicity of simultaneous administration of Typhim Vi (TV), YF-VAX (YF) and Menomune (MV) [Abstract]. Presented at the 36th International Conference on Antimicrobial Agents and Chemotherapy; September 15--18, 1996; New Orleans, Louisiana.
- Tauraso NM, Myers MG, Nau EV, O'Brien TC, Spindel SS, Trimmer RW. Effect of interval between inoculation of live smallpox and yellow-fever vaccines on antigenicity in man. J Infect Dis 1972;126:362--71.
- Kollaritsch H, Que JU, Kunz C, Wiedermann G, Herzog C, Cryz SJ, Jr. Safety and immunogenicity of live oral cholera and typhoid vaccines administered alone or in combination with antimalarial drugs, oral polio vaccine, or yellow fever vaccine. J Infect Dis 1997;175:871--5.
- Tsai TF, Kollaritsch H, Que JU, et al. Compatible concurrent administration of yellow fever 17D vaccine with oral, live, attenuated cholera CVD103-HgR and typhoid ty21a vaccines. J Infect Dis 1999;179:522--4.
- Fletcher MA, Fabre P, Debois H, Saliou P. Vaccines administered simultaneously: directions for new combination vaccines based on an historical review of the literature. Int J Infect Dis 2004;8:328--38.
- Kaplan JE, Nelson DB, Schonberger LB, et al. The effect of immune globulin on the response to trivalent oral poliovirus and yellow fever vaccinations. Bull World Health Organ 1984;62:585--90.
- Edupuganti S, Eidex R, Keyserling H, et al. A randomized, double-blind, controlled trial of the comparative viremia, immunogenicity and safety of live, attenuated 17D yellow fever vaccine given in combination with human immune globulin or placebo. Presented at the 12th Annual Conference on Vaccine Research; April 27--29, 2009; Baltimore; Maryland.
- Tsai TF, Bolin RA, Lazuick JS, Miller KD. Chloroquine does not adversely affect the antibody response to yellow fever vaccine. J Infect Dis 1986;154:726--7.
- Kelleher P, Misbah SA. What is Good's syndrome? Immunological abnormalities in patients with thymoma. J Clin Pathol 2003;56:12--6.
- Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents; Dept Health Human Services; 2008. Available at http://aidsinfo.nih.gov/Guidelines/GuidelineDetail.aspx?MenuItem=Guidelines&Search=Off&GuidelineID=7&ClassID=1. Accessed July 1, 2010.
- Working Group on Antiretroviral Therapy and Medical Management of HIV-Infected Children. Guidelines for the use of antiretroviral agents in pediatric HIV infection, 2009. Available at http://aidsinfo.nih.gov/ContentFiles/PediatricGuidelines.pdf. Accessed July 1, 2010.
- Tattevin P, Depatureaux AG, Chapplain JM, et al. Yellow fever vaccine is safe and effective in HIV-infected patients. AIDS 2004;18:825--7.
- Ho YL, Enohata T, Lopes MH, De Sousa Dos Santos S. Vaccination in Brazilian HIV-infected adults: a cross-sectional study. AIDS Patient Care STDS 2008;22:65--70.
- Wilson ME, von Reyn CF, Fineberg HV. Infections in HIV-infected travelers: risks and prevention. Ann Intern Med 1991;114:582--92.
- Pistone T, Verdiere CH, Receveur MC, Ezzedine K, Lafon ME, Malvy D. [Immunogenicity and tolerance of yellow fever vaccine in travelers with HIV, France, 2005]. BEH Thematique 2007;25--26:238--40.
- Jong EC, Freedman DO. Advising travelers with specific needs. In: Brunette GW, Kozarsky PE, Magill AJ, Shlim DR, Whatley AD, eds. CDC health information for international travel 2010. Atlanta. GA: US Department of Health and Human Services, Public Health Service, CDC; 2009.
- Conesa-Botella A, Colebunders R. Response to "Case of yellow fever vaccine-associated viscerotropic disease with prolonged viremia, robust adaptive immune responses, and polymorphisms in CCR5 and RANTES genes." J Infect Dis 2009;199:601.
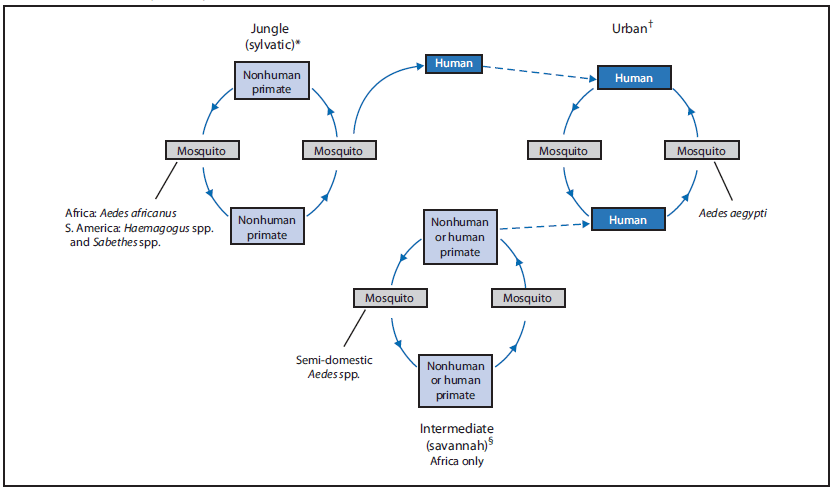
* The jungle (sylatic) transmission cycle involves transmission of the virus between nonhuman primates and mosquito species found in the forest canopy. The virus is transmitted via mosquitoes from nonhuman primates to human when the humans encroach into the jungle during occupational or recreational activities.
† The urban transmission cycle involves transmission of the virus between human and urban mosquitoes, primarily Ae. aegypti. Viremic humans traveling from one region to another can feed into and serve as a source of infection for mosquitoes in other transmission cycles (dotted line).
§ In Africa, an intermediate (savannah) cycle involves transmission of YFV from tree hole-breeding Aedes spp. to humans living or working in jungle border areas. In this cycle, the virus can be transmitted from nonhuman primate to humans or from human to human via these mosquitoes.
Alternate Text: This figure shows the three yellow fever transmission cycles: jungle (sylvatic), intermediate (savannah), and urban. The jungle (sylatic) transmission cycle involves transmission of the virus between nonhuman primates and mosquito species found in the forest canopy. The virus is transmitted via mosquitoes from nonhuman primates to human when the humans encroach into the jungle during occupational or recreational activities. The urban transmission cycle involves transmission of the virus between human and urban mosquitoes, primarily Aedes aegypti. Viremic humans traveling from one region to another can feed into and serve as a source of infection for mosquitoes in other transmission cycles. In Africa, an intermediate (savannah) cycle involves transmission of YFV from tree-hole-breeding Aedes spp. to humans living or working in jungle border areas. In this cycle, the virus can be transmitted from nonhuman primate to humans or from human to human via these mosquitoes.
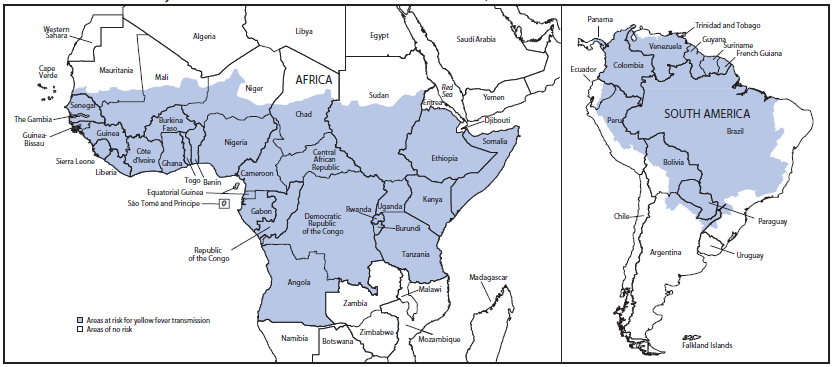
Source: Brunette GW, Kozarsky PE, Magill AJ, Shlim DR, Whatley AD, eds. CDC health information for international travel 2010. Atlanta. GA: US Department of Health and Human Services, Public Health Service, CDC; 2009.
* As defined by the World Health Organization, countries/areas where yellow fever has been reported currently or in the past, plus areas where vectors and animal reservoirs currently exist.
Alternate Text: The figure shows the areas of Africa and South America in which the World Health Organization has indicated that yellow fever has been reported currently or in the past and areas where vectors and animal reservoirs currently exist.
|
TABLE 1. Countries with risk for yellow fever virus (YFV) transmission* |
|||
|---|---|---|---|
|
Africa |
Central and South America |
||
|
Angola Benin Burkina Faso Burundi Cameroon Central African Republic Chad† Congo, Republic of the Côte d'Ivoire Democratic Republic of the Congo Equatorial Guinea Ethiopia Gabon |
The Gambia Ghana Guinea Guinea-Bissau Kenya Liberia Mali† Mauritania† Niger† Nigeria Rwanda Sierra Leone São Tomé and Principe |
Senegal Somalia Sudan† Tanzania Togo Uganda |
Argentina† Bolivia† Brazil† Colombia Ecuador† French Guiana Guyana Panama† Paraguay Peru† Suriname Trinidad and Tobago† Venezuela† |
|
* As defined by the World Health Organization, countries/areas where yellow fever has been reported currently or in the past, plus areas where vectors and animal reservoirs currently exist. † These countries are not holoendemic (i.e., only a portion of the country has risk for YFV transmission). Information about which areas are endemic is available at http://wwwnc.cdc.gov/travel/yellowbook/2010/chapter-2/yellow-fever.aspx. |
|||
|
TABLE 2. Contraindications and precautions to yellow fever vaccine administration |
|
|---|---|
|
Contraindications |
Precautions |
|
Allergy to vaccine component |
Age 6--8 months |
|
Age less than 6 months |
Age ≥60 years† |
|
Symptomatic HIV infection or CD4+ T-lymphocytes <200/mm3 (or <15% of total in children aged <6 years)* |
Asymptomatic HIV infection and CD4+ T-lymphocytes 200--499/mm3 (or 15%--24% of total in children aged <6 years)* |
|
Thymus disorder associated with abnormal immune function† |
Pregnancy |
|
Primary immunodeficiencies |
Breastfeeding |
|
Malignant neoplasms |
|
|
Transplantation |
|
|
Immunosuppressive and immunomodulatory therapies† |
|
|
* Symptoms of HIV have been classified (Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents; US Department of Health and Human Services; 2008. Available at http://aidsinfo.nih.gov/Guidelines/GuidelineDetail.aspx?MenuItem=Guidelines&Search=Off&GuidelineID=7&ClassID=1. Working Group on Antiretroviral Therapy and Medical Management of HIV-Infected Children. Guidelines for the Use of Antiretroviral Agents in Pediatric HIV Infection; 2009. Available at http://aidsinfo.nih.gov/ContentFiles/PediatricGuidelines.pdf.) † New contraindication or precaution since the ACIP's recommendations for the use of yellow fever vaccine were published in 2002 (CDC. Yellow fever vaccine: recommendations of the Advisory Committee on Immunizations Practices: MMWR 2002;51[No. RR-17]). |
|
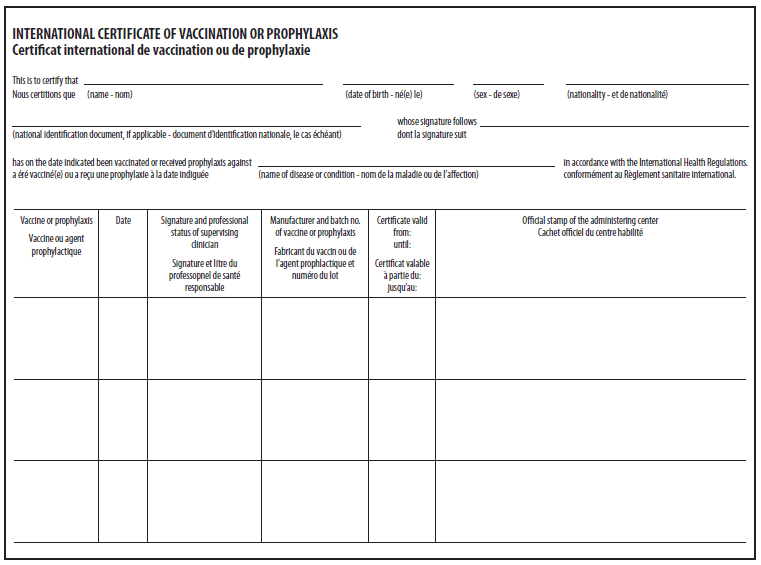
Source: Brunette GW, Kozarsky PE, Magill AJ, Shlim DR, Whatley AD, eds. CDC health information for international travel 2010. Atlanta. GA: US Department of Health and Human Services, Public Health Service, CDC; 2009.
* International Health Regulations allow countries to require proof of yellow fever vaccination for entry and from travelers arriving from certain countries to prevent importation and indigenous transmission of yellow fever vaccine. As proof of vaccination, vaccinees should receive a completed ICVP, validated (stamped and signed) with the stamp of the center where the vaccine was administered.
FIGURE 4. Medical contraindication section of the International Certificate of Vaccination or Prophylaxis (ICVP)*

Source: Brunette GW, Kozarsky PE, Magill AJ, Shlim DR, Whatley AD, eds. CDC health information for international travel 2010. Atlanta. GA: US Department of Health and Human Services, Public Health Service, CDC; 2009.
* A health-care provider who identifies a medical contraindication to yellow fever vaccination and wants to issue a medical waiver should fill out and sign the Medical Contraindications to Vaccination section of the ICVP. The health-care provider also should give the traveler a signed and dated exemption letter.
Advisory Committee on Immunization Practices
Membership List, October 2009
Chair: Carol Baker, MD, Baylor College of Medicine, Houston, Texas.
Executive Secretary: Larry Pickering, MD, National Center for Immunization and Respiratory Diseases, CDC, Atlanta, Georgia.
Members: Lance Chilton, MD, University of New Mexico, Albuquerque, New Mexico; Paul Cieslak, MD, Oregon Public Health Division, Portland, Oregon; Kristen Ehresmann, MPH, Minnesota Department of Health, St. Paul, Minnesota; Janet Englund, MD, University of Washington and Children's Hospital and Regional Medical Center, Seattle, Washington; Franklyn Judson, MD, University of Colorado Health Sciences Center, Denver, Colorado; Wendy Keitel, MD, Baylor College of Medicine, Houston, Texas; Susan Lett, MD, Massachusetts Department of Public Health, Boston, Massachusetts; Michael Marcy, MD, UCLA Center for Vaccine Research, Torrance, California; Cody Meissner, MD, Tufts Medical Center, Boston, Massachusetts; Kathleen Neuzil, MD, University of Washington, Seattle, Washington; Sara Rosenbaum, JD, George Washington University, District of Columbia; Mark Sawyer, MD, University of California - San Diego, California; Ciro Valent Sumaya, MD, Texas A&M Health Science Center, College Station, Texas; Jonathan Temte, MD, University of Wisconsin School of Medicine and Public Health, Madison, Wisconsin.
ExOfficio Members: James E. Cheek, MD, Indian Health Service, Albuquerque, New Mexico; Wayne Hachey, DO, Department of Defense, Falls Church, Virginia; Geoffrey S. Evans, MD, Health Resources and Services Administration, Rockville, Maryland; Bruce Gellin, MD, National Vaccine Program Office, District of Columbia; Linda Murphy, Centers for Medicare and Medicaid Services, Baltimore, Maryland; George T. Curlin, MD, National Institutes of Health, Bethesda, Maryland; Norman Baylor, MD, Food and Drug Administration, Bethesda, Maryland; Linda Kinsinger, MD, Department of Veterans Affairs, Durham, North Carolina.
Liaison Representatives: American Academy of Family Physicians, Doug Campos-Outcalt, MD, Phoenix, Arizona; American Academy of Pediatrics, Joseph Bocchini, MD, Shreveport, Louisiana, David Kimberlin, MD, Birmingham, Alabama; American College Health Association, James C. Turner, MD, Charlottesville, Virginia; American College of Obstetricians and Gynecologists, Stanley Gall, MD, Louisville, Kentucky; American College of Physicians, Gregory Poland, Rochester, Minnesota; American Geriatrics Society, Kenneth Schmader, MD, Durham, North Carolina; America's Health Insurance Plans, Mark Netoskie, MD, Houston, Texas; American Medical Association, Litjen Tan, PhD, Chicago, Illinois; American Osteopathic Association, Stanley Grogg, DO, Tulsa, Oklahoma; American Pharmacists Association, Stephan L. Foster, PharmD, Memphis, Tennessee; Association for Prevention Teaching and Research, W. Paul McKinney, MD, Louisville, Kentucky; Biotechnology Industry Organization, Clement Lewin, PhD, Cambridge, Massachusetts; Canadian National Advisory Committee on Immunization, Joanne Langley, MD, Halifax, Nova Scotia, Canada; Council of State and Territorial Epidemiologists, Christine Hahn, MD, Boise, Idaho; Department of Health, United Kingdom David M. Salisbury, MD, London, United Kingdom; Healthcare Infection Control Practices Advisory Committee, Alexis Elward, MD, St Louis, Missouri; Infectious Diseases Society of America, Samuel L. Katz, MD, Durham, North Carolina; National Association of County and City Health Officials, Jeff Duchin, MD, Seattle, Washington; National Association of Pediatric Nurse Practitioners, Patricia Stinchfield, St Paul, Minnesota; National Foundation for Infectious Diseases, William Schaffner, MD, Nashville, Tennessee; National Immunization Council and Child Health Program, Mexico, Vesta Richardson, MD, Mexico City, Mexico; National Medical Association, Patricia Whitley-Williams, MD, New Brunswick, New Jersey; National Vaccine Advisory Committee, Guthrie Birkhead, MD, Albany, New York; Pharmaceutical Research and Manufacturers of America, Damian A. Braga, Swiftwater, Pennsylvania, Peter Paradiso, PhD, Collegeville, Pennsylvania; Society for Adolescent Medicine, Amy Middleman, MD, Houston, Texas; Society for Healthcare Epidemiology of America, Harry Keyserling, MD, Atlanta, Georgia.
Yellow Fever Vaccine Work Group
Chair: Carol J. Baker, MD, Houston, Texas.
Members: Elizabeth Barnett, MD, Boston, Massachusetts; Louisa Chapman, MD, Atlanta, Georgia; Eileen Farnon, MD, Atlanta, Georgia; Patrick Garmon, PharmD, PhD, Falls Church, Virginia; Mark D. Gershman, MD, Atlanta, Georgia; Christina Greenaway, MD, Toronto, Canada; Kristen B. Janusz, DVM, Fort Collins, Colorado; James F. Jones, MD, Atlanta, Georgia; Franklyn N. Judson, MD, Denver, Colorado; Katrin S. Kohl, MD, Atlanta, Georgia; Nina Marano, DVM, Atlanta, Georgia; Michael E. Neseman, MD, Lima, Peru; Karen O'Brien, MD, Fort Monroe, Virginia; Michele Sabourin, Ottawa, Ontario, Canada; Betsy Schroeder, MPH, Atlanta, Georgia; Barbara A. Slade, MD, Atlanta, Georgia; J. Erin Staples, MD, PhD, Fort Collins, Colorado; Andrea Sutherland, MD, Silver Springs, Maryland; Mary E. Wilson, MD, Boston, Massachusetts.
* A list of the members of the work group appears on page 27 of this report.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


