Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Buprenorphine Prescribing Practices and Exposures Reported to a Poison Center — Utah, 2002–2011
Buprenorphine is an effective medication for the treatment of opioid dependence. Its use has increased in the United States as a result of the Drug Addiction Treatment Act of 2000, which allowed physicians to prescribe certain medications as part of office-based treatment for opioid addiction. In France, widespread use of medication-assisted therapy, primarily buprenorphine treatment, was associated with an 80% decrease in overdose deaths from heroin or cocaine from 465 in 1996 to 89 in 2003 (1). With the expanded use of buprenorphine, an increase in exposures among children and adults has been reported in the United States. These exposures (including unintentional and intentional, therapeutic and nontherapeutic) have resulted in adverse effects and, in a small number of cases, death. To assess statewide increases in buprenorphine use and the number of reported exposures, the Utah Department of Health analyzed data from the Utah Controlled Substance Database (CSD) and the Utah Poison Control Center (PCC). The results of that analysis indicated a statewide increase in the annual number of patients prescribed buprenorphine from 22 in 2002 to 9,793 in 2011, and a concurrent increase in the annual number of prescribers writing buprenorphine prescriptions from 16 to 1,088. Over the same period, the number of exposures to buprenorphine reported annually to the PCC increased from six to 81. However, comparison of the ratios of buprenorphine exposures to patients and prescribers in 2002 with data for 2011 indicated substantial decreases from 6/22 for patients and 6/16 for prescribers in 2002 to 81/9,793 for patients and 81/1,088 for prescribers in 2011. Three of the total 462 buprenorphine exposures reported during 2002–2011 in Utah, in a teen and two adults, were associated with fatal outcomes. Increased buprenorphine prescribing in Utah during 2002–2011 likely represents expanded access to critically needed opioid addiction treatment; however, safeguards should be in place to prevent adverse effects. Prescribers and pharmacists are encouraged to counsel patients carefully regarding the safe use, storage, and disposal of buprenorphine.
The epidemic of opioid addiction and related overdose deaths is a well-described and growing public health problem in the United States (2). Numerous barriers to accessing opioid addiction treatment have been identified.* Buprenorphine was approved by the Food and Drug Administration in 2002 for the treatment (alone or in combination with naloxone) of opioid dependence (3,4). The efficacy of buprenorphine in the treatment of opioid dependence has consistently been demonstrated (5), and its use has been associated with new types of patients receiving addiction treatment. Similar to other opioids, buprenorphine produces euphoria and respiratory depression in a dose-dependent manner. However, unique to buprenorphine, these effects increase until, at moderate doses, the effects reach a plateau and no longer continue to increase, making respiratory depression less likely in a habituated opioid user (4,6). This "ceiling effect" has raised concern that some prescribers and patients might think buprenorphine unlikely to cause any adverse effects (6). Studies have indicated that, in an opioid naïve patient, respiratory depression might occur before reaching this ceiling, especially in young children (6).
For this report, data for 2002–2011 were analyzed from the state's CSD and PCC. The CSD tracks all outpatient (but not inpatient) prescriptions for Schedule II–V drugs dispensed in Utah. CSD is maintained by the Division of Occupational and Professional Licensing within the Utah Department of Commerce. The PCC maintains data on reported human exposures to buprenorphine and other drugs (including intentional and unintentional, therapeutic and nontherapeutic exposures). Standardized information collected for each exposure includes age, sex, substance, route of exposure, reason for exposure, location of exposure, location of caller, therapy provided, clinical effects, management location, and medical outcome.
CSD Findings
During 2002–2011, the number of prescribers writing prescriptions for buprenorphine increased 67-fold, from 16 in 2002 to 1,088 in 2011, and the number of patients filling buprenorphine prescriptions increased 444-fold, from 22 in 2002 to 9,793 in 2011. In 2011, the 106,415 buprenorphine prescriptions recorded in the CSD amounted to 2% of the total 5,291,530 controlled substance prescriptions.
The patients whose prescriptions for buprenorphine were recorded in the CSD during 2002–2011 were predominantly (59.7%) men. The mean age of the persons for whom exposures were reported was 34.7 years (standard deviation: 12.6 years), and the median age was 31 years (range: <1 to 109 years).
PCC Findings
From 2002 to 2011, the number of exposures to buprenorphine reported annually to the PCC increased approximately 13-fold, from six to 81. The number of exposures to buprenorphine began to rise significantly in 2004 overall, whereas a similar rise among children aged ≤5 years did not begin until 2006 (Figure). However, comparison of the ratios of buprenorphine exposures to patients and prescribers in 2002 with data for 2011 indicated substantial decreases from 6/22 for patients and 6/16 for prescribers in 2002 to 81/9,793 for patients and 81/1,088 for prescribers in 2011. Of the 462 exposures recorded in the PCC database during 2002–2011, 250 (54.1%) were among adults aged ≥20 years, 179 (38.7%) were among children aged ≤5 years, and 33 (7.1%) were among persons aged 6–19 years (Figure). Nearly all (94%) of the exposures among children aged ≤5 years were to sublingual tablets rather than to the buprenorphine film product, which was not approved until 2010.
The most common clinical effects reported in children aged ≤5 years were drowsiness in 105 (58.6%), vomiting in 47 (26.2%), respiratory depression in 34 (19.0%), miosis in 27 (15.1%), agitation in 18 (10.1%), and tachycardia in 15 (8.4%). Respiratory arrest was reported in three (1.7%) children. In adults, the most common clinical effects included drowsiness in 72 (28.8%), vomiting in 53 (21.2%), agitation in 52 (20.8%), nausea in 49 (19.6%), confusion in 28 (11.2%), dizziness in 28 (11.2%), diaphoresis in 21 (8.4%), tachycardia in 17 (6.8%), respiratory depression in 14 (5.6%), ataxia in 13 (5.2%), and diarrhea in 13 (5.2%). Respiratory arrest was noted in two (0.8%) adults.
Among adults, 33 exposures (13.2%) were unintentional, and 126 (50.4%) were intentional (suicidal intent or intentional misuse or abuse of the medication). Of the 250 adult exposures, 22 (8.8%) were related to withdrawal and 57 (22.8%) to adverse reaction to the medication. A known outcome was documented in 164 (91.6%) children aged ≤5 years, and 42 (25.6%) of those outcomes had a moderate or major effect. In adults, a known outcome was documented in 220 (88.0%) exposures, and 47 (21.4%) of those outcomes had a moderate, major, or fatal effect. Three fatal outcomes were reported, including two in adults and one in a teen.
The majority of the 462 persons with exposures were treated in a health-care facility (247; 53.5%). Of the 247, a total of 127 (51.4%) were treated and released from an emergency department, and the remainder were admitted for medical care. A higher proportion of children aged ≤5 years (137; 76.5%) were treated in a health-care facility, compared with adults (103; 41.2%).
Reported by
Karen C. Thomas, PhD, Marty Malheiro, MS, Barbara I. Crouch, PharmD, Utah Poison Control Center; Christina A. Porucznik, PhD, Dept of Family and Preventive Medicine, University of Utah. Corresponding contributor: Christina A. Porucznik, christy.porucznik@utah.edu, 801-581-4330.
Editorial Note
As use of buprenorphine increased rapidly since 2002, poison control centers throughout the United States observed increases in the number of buprenorphine exposures (7). Increased therapeutic use of buprenorphine likely will help reduce prescription opioid abuse and misuse; however, nontherapeutic or inappropriate use of buprenorphine can cause serious and potentially life-threatening effects among children and adults. Young children exploring their environments might lick or ingest this medication, resulting in vomiting, respiratory depression, coma, or death (6). Results of a study in an Appalachian community suggest that improved access to buprenorphine treatment might help reduce sharing of the medication among adults and teens or diversion for nontherapeutic use that could cause adverse effects (8).
The increase in buprenorphine use has expanded access to opioid addiction treatment in the office-based setting. This presents new opportunities for health-care providers to reduce morbidity and mortality related to opioid addiction and to mitigate risks associated with nontherapeutic use of this drug. Prescriber and pharmacist counseling of patients regarding the safe use, storage, and disposal of this medication can help prevent adverse consequences from unintentional exposure among children or diversion and experimentation among teens and adults who are opioid naïve. In certain cases, health-care provider counseling might prevent a fatality. In several cases, investigators found that help was not sought immediately after exposure, likely because a child was not observed to have swallowed a tablet or placed packaging in its mouth, only to be found later with respiratory effects. Counseling by health-care providers about the potential dangers to children from licking a buprenorphine film package or holding a sublingual tablet in the mouth, even briefly, might help caregivers learn the importance of early intervention in any buprenorphine exposure.
The findings in this report are subject to at least two limitations. The CSD limits identifiers and does not include information on diagnosis; therefore, it is not possible to determine the reason for prescribing buprenorphine or to evaluate the appropriateness of the prescription. The PCC did not add a code for the buprenorphine film product until October 2010; therefore, some exposures to the film product might have been attributed to the sublingual tablet.
The expanded use of buprenorphine as part of office-based treatment is an important tool to reduce morbidity and mortality associated with opioid addiction (9). Education on how to safely use, store, and dispose of buprenorphine is needed to help prevent unintentional exposures. Health-care professionals and members of the public can contact their local poison control center at 800-222-1222 for guidance regarding the adverse consequences of any exposure and the safe use, storage, and disposal of medications such as buprenorphine (Box). Patients and caregivers are encouraged to seek assistance from their pharmacists, prescribers, local poison control centers, and other members of the health-care community for information regarding the safe use of their medications.
Acknowledgments
Jacob Crook, Anna Fondario, MPH, Utah Dept of Health.
References
- Emmanuelli J, Desenclos JC. Harm reduction interventions, behaviours and associated health outcomes in France, 1996–2003. Addiction 2005;100:1690–1700.
- CDC. CDC grand rounds: prescription drug overdoses— a U.S. epidemic. MMWR 2012;61:10–3.
- Gowing L, Ali R, White JM. Buprenorphine for the management of opioid withdrawal. Cochrane Database Syst Rev 2009;3:CD002025.
- Sporer KA. Buprenorphine: a primer for emergency physicians. Ann Emerg Med 2004;43:580–4.
- Mendelson J, Flower K, Pletcher MJ, Galloway GP. Addiction to prescription opioids: characteristics of the emerging epidemic and treatment with buprenorphine. Exp Clin Psychopharmacol 2008;16:435–41.
- Pedapati EV, Bateman ST. Toddlers requiring pediatric intensive care unit admission following at-home exposure to buprenorphine/naloxone. Pediatr Crit Care Med 2011;12:e102–7.
- Durback-Morris LF, Scharman EJ. Are children the unintended victims of changes in buprenorphine prescribing practices [Abstract]? Clin Toxicol 2010;48:616.
- Lofwall MR, Havens JR. Inability to access buprenorphine treatment as a risk factor for using diverted buprenorphine. Drug Alcohol Depend 2012;126:379–83.
- Kresina TF, Lubran R. Improving public health through access to and utilization of medication assisted treatment. Int J Environ Res Public Health 2011;8:4102–17.
* Additional information available at http://www.ncbi.nlm.nih.gov/books/nbk14677.
What is already known on this topic?
Opioid addiction is a well-described and growing public health problem in the United States. In 2002, buprenorphine was approved by the Food and Drug Administration for the treatment (alone or in combination with naloxone) of opioid dependence.
What is added by this report?
In Utah, the annual number of prescribers writing prescriptions for buprenorphine increased 67-fold, from 16 in 2002 to 1,088 in 2011, and the annual number of patients filling buprenorphine prescriptions increased 444-fold, from 22 to 9,793. During the same period, as the number of prescriptions increased, the annual number of buprenorphine exposures increased 13-fold, from six to 81, with exposures primarily among adults aged ≥20 years and children aged ≤5 years. Of 462 exposures, three (<1%) were fatal.
What are the implications for public health practice?
Despite buprenorphine's effectiveness in the treatment of opioid dependence, nontherapeutic use, both misuse and unintentional exposure, can have adverse outcomes. Expanded use of buprenorphine for the treatment of opioid dependence is important to improve public health. Through education and counseling of prescribers, pharmacists, and patients regarding the safe use, storage, and disposal of this drug, adverse effects likely can be reduced.
FIGURE. Reported buprenorphine exposures (N = 462), by age group — Utah Poison Control Center, 2002–2011
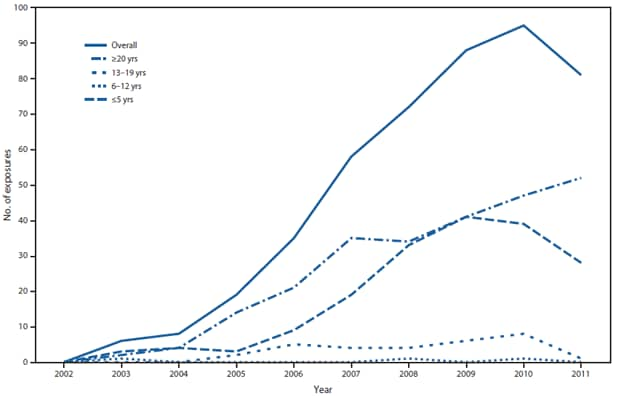
Alternate Text: The figure above shows reported buprenorphine exposures (N = 462), by age group during 2002-2011, according to the Utah Poison Control Center (PCC). From 2002 to 2011, the number of exposures to buprenor-phine reported annually to the PCC increased approximately 13-fold, from six to 81.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


