Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Vital Signs: Risk for Overdose from Methadone Used for Pain Relief — United States, 1999–2010
On July 3, 2012, this report was posted as an MMWR Early Release on the MMWR website (http://www.cdc.gov/mmwr).
Abstract
Background: Vital statistics data suggest that the opioid pain reliever (OPR) methadone is involved in one third of OPR-related overdose deaths, but it accounts for only a few percent of OPR prescriptions.
Methods: CDC analyzed rates of fatal methadone overdoses and sales nationally during 1999–2010 and rates of overdose death for methadone compared with rates for other major opioids in 13 states for 2009.
Results: Methadone overdose deaths and sales rates in the United States peaked in 2007. In 2010, methadone accounted for between 4.5% and 18.5% of the opioids distributed by state. Methadone was involved in 31.4% of OPR deaths in the 13 states. It accounted for 39.8% of single-drug OPR deaths. The overdose death rate for methadone was significantly greater than that for other OPR for multidrug and single-drug deaths.
Conclusions: Methadone remains a drug that contributes disproportionately to the excessive number of opioid pain reliever overdoses and associated medical and societal costs.
Implications for Public Health Practice: Health-care providers who choose to prescribe methadone should have substantial experience with its use and follow consensus guidelines for appropriate opioid prescribing. Providers should use methadone as an analgesic only for conditions where benefit outweighs risk to patients and society. Methadone and other extended-release opioids should not be used for mild pain, acute pain, "breakthrough" pain, or on an as-needed basis. For chronic noncancer pain, methadone should not be considered a drug of first choice by prescribers or insurers.
Introduction
U.S. physicians have used the synthetic opioid methadone as a treatment for heroin addiction since the 1960s and increasingly as a treatment for chronic noncancer pain since the mid-1990s (1). Individual states began to report increasing numbers of overdose deaths involving methadone in 2003 (2). Subsequently, rates of deaths and emergency department (ED) visits involving methadone have increased nationwide (3,4). Studies using medical examiner data suggested that more than three quarters of methadone overdoses involved persons who were not enrolled in programs treating opioid addiction with methadone and that most persons who overdosed were using it without a prescription (3). In November 2006, the Food and Drug Administration (FDA) issued a warning regarding careful prescribing of methadone because of the sharp rise in overdose deaths among patients receiving methadone for pain (5). FDA also revised the interval for the recommended starting dosage from 2.5–10 mg every 3–4 hours to 2.5–10 mg every 8–12 hours. In January 2008, on request of the Drug Enforcement Administration (DEA), manufacturers voluntarily limited distribution of the largest (40 mg) formulation of methadone to authorized opioid addiction treatment programs and hospitals only, because this formulation was not approved for the treatment of pain (6).
Recent analyses have shown that methadone was involved in one in three opioid-related deaths in 2008 (7). Moreover, the involvement of methadone in drug overdose deaths, in toxic exposures quantified by poison centers, and in diversion to nonpatients is disproportionate to the number of methadone prescriptions for pain when compared with other opioid pain relievers (3,8). Analysis of ED data indicates that the estimated number of ED visits resulting from nonmedical use of methadone alone or in combination with other drugs in 2009 (n = 63,031) was significantly greater than the estimated number in 2004 (n = 36,806) (4). CDC reviewed national data on trends in methadone use and mortality and data from medical examiners on methadone mortality to determine whether additional recommendations for its safe use for pain treatment are necessary.
Methods
For this report, national death rates during 1999–2009 are based on the National Vital Statistics System multiple cause of death files (9). Methadone-related deaths were defined as those with an underlying cause of death classified by the International Classification of Diseases, 10th Revision (ICD-10) external cause of injury codes as X40-X44, X60-X64, X85, or Y10-Y14 and an ICD-10 code (T40.3) for methadone poisoning. Methadone might have been listed alone or in combination with other drugs.
The amounts of opioid pain relievers distributed for 1999–2010 nationally and by state were obtained from the DEA's Automation of Reports and Consolidated Orders System (ARCOS).* Distributions of methadone to opioid treatment programs were not included. Annual numbers of prescriptions dispensed for methadone and other opioids in outpatient settings for 1999–2009 came from an analysis conducted by FDA in 2010 using a commercial prescription and patient measurement service (Vector One: National [VONA]) that can estimate the number of prescriptions for drugs dispensed by outpatient retail pharmacies in the United States (10).
Population-based counts of drug-related deaths for methadone and other opioids in 2009 came from 13 states in the Medical Examiner component of the Drug Abuse Warning Network (DAWN): Delaware, Massachusetts, Maryland, Maine, New Hampshire, New Mexico, Oklahoma, Oregon, Rhode Island, Utah, Virginia, Vermont, and West Virginia.† State medical examiners provided information on all drug-related deaths, and CDC analyzed the deaths involving an opioid, whether in combination with other drugs or by itself. Opioid distribution data for these states were available from the ARCOS system and converted to morphine milligram equivalents (MME) using a standard reference (11).
Comparison of methadone to other major opioids in DAWN data was based on rates of death per 100 kg of opioid analgesic in MME. Drug-specific rates were compared using rate ratios and 95% confidence intervals with the rates for methadone as the referents.
Results
The rate of overdose deaths involving methadone in the United States in 2009 was 5.5 times the rate in 1999 (Figure 1). The mortality rate peaked at 1.8 deaths per 100,000 persons in 2007 and then declined in parallel with the amount of methadone being distributed nationally in 2008 and 2009. The annual rate of methadone prescriptions for pain rose to 1.5 per 100 persons by 2008 and did not increase further in 2009. Methadone accounted for 4.4 million (1.7%) of the 257 million opioid prescriptions in 2009. However, in 2010, methadone accounted for 9.0% of all the MME of all major opioids tracked by ARCOS other than buprenorphine. This proportion varied by state from 4.5% in New Jersey to 18.5% in Washington (Figure 2).
Among the 13 DAWN Medical Examiner states, methadone accounted for 9.8% of the MME tracked by ARCOS. Methadone was involved in 31.4% of the 3,294 deaths involving these opioids, more than any opioid other than oxycodone in 2009 (Table). Among the 748 single-drug deaths, methadone was involved in 298 (39.8%), twice as many as any other opioid. The rate of methadone deaths per 100 kg sold in MME was significantly higher than that for any other opioid for both all deaths and single-drug deaths. The difference between methadone and other opioids was more pronounced in the analysis of single-drug deaths. Even if some of these deaths (e.g., 25%) had been attributable to methadone dispensed from opioid treatment programs, the differences between methadone and other opioids would remain significant. The methadone death rate was still significantly higher than the rate for any other opioid in both comparisons.
Conclusions and Comment
The primary advantages of using methadone over other opioids for pain treatment are its long duration of action, relatively low cost, and availability in liquid formulation for oral use. Its primary disadvantages are its long and unpredictable half-life and associated risk for accumulating toxic levels leading to severe respiratory depression; its multiple interactions with other drugs, including frequently abused drugs such as antianxiety agents; and its ability to cause major disturbances of cardiac rhythm (12).
Increased use of methadone since 1999 might have been prompted by growing costs of treating pain with opioids and increasing reports of abuse of other, more expensive, extended-release opioids (1). Overdose reports and interventions by FDA and DEA might have resulted in declines in the amount of methadone distributed and methadone-related fatal overdoses in 2008, although the number of methadone prescriptions did not decline. The parallel trends in the amount of methadone distributed for use as a pain reliever and in the methadone mortality rate are consistent with methadone prescribed as a pain reliever being the primary determinant of methadone mortality rates (1,3).
Data suggest that some of the current uses of methadone for pain might be inappropriate. According to an analysis conducted by FDA, the most common diagnoses associated with methadone use for pain in 2009 were musculoskeletal problems (such as back pain and arthritis) (46%), headaches (17%), cancer (11%), and trauma (5%). Most methadone prescriptions were written by primary care providers or mid-level practitioners (e.g., nurse practitioners) rather than pain specialists. Nearly a third of prescriptions appear to have been dispensed to patients with no opioid prescriptions in the previous month (i.e., opioid-naïve patients) (10).
The findings in this report are subject to at least five limitations. First, vital statistics underestimate the number of overdose deaths from specific drugs because the type of drug is not specified on many death certificates. Second, medical examiners in the DAWN system might have varying definitions of drug-related deaths. However, individual medical examiners likely apply the same definitions to all types of opioid analgesics. Third, assigning responsibility to any single drug in multidrug overdoses is difficult. However, this is not an issue in single-drug deaths, among which the highest risks for methadone were observed. Fourth, some deaths might have resulted from methadone provided in take-home doses by opioid treatment programs, but adjusting for such deaths in this analysis did not change the overall results. Finally, ARCOS data reflect distributions to retail outlets by state, but some drugs might have been used by residents of neighboring states.
This study and others suggest that methadone remains a drug that contributes disproportionately to opioid pain reliever overdoses and associated medical and societal costs. Additional warnings to prescribers about dosage are likely to have limited effect, given the high prevalence of use without a prescription among persons who overdose. The public health goal now should be to mount a concerted effort to reserve methadone for those pain-related conditions for which the benefits likely outweigh the risks to patients and society, such as use for cancer-related pain or palliative care. This will reduce the amount of methadone available for diversion and nonmedical use.
Methadone and other, extended-release opioids should not be used for mild pain, acute pain, "breakthrough" pain, or on an as-needed basis. For chronic noncancer pain, methadone should not be considered a drug of first choice. This is especially true for conditions for which the benefits of opioids have not been demonstrated, such as headache and low back pain. Only a small fraction of patients with intractable chronic headache treated with opioids experience long-term pain reduction or functional improvement (13). Evidence that any opioids are effective in chronic low back pain is limited (14). Additionally, methadone should not be prescribed to opioid-naïve patients, and, whenever possible, should not be prescribed to patients taking benzodiazepine antianxiety agents because of an increased risk for severe respiratory depression. Health-care providers who choose to prescribe methadone should have substantial experience with its use and follow consensus guidelines for appropriate opioid prescribing (15). Providers should instruct patients about the potential risks of methadone and how to store and dispose of it properly.
Public and private insurers and health-care systems can ensure that prescribers of methadone follow dosage guidelines by requiring authorization for starting doses for pain that exceed the recommended upper limit of 30 mg per day (5). Insurance formularies should not list methadone as a preferred drug for the treatment of chronic noncancer pain. Pharmaceutical companies should introduce a 2.5-mg formulation of methadone to facilitate treatment with the lowest recommended dosage.
Although interventions related to methadone use are urgently needed, government agencies, health-care providers, insurers, and other stakeholders must combine these interventions with measures that will address the problems of misuse and abuse of all opioid pain relievers. Interventions such as the use of prescription drug monitoring programs, appropriate screening and monitoring before prescribing opioid pain relievers, regulatory and law enforcement efforts, and state policies (e.g., "pill mill" laws) aimed at providers and patients involved in diversion of these drugs continue to be essential elements in addressing this public health emergency.
Reported by
Leonard J. Paulozzi, MD, Karin A. Mack, PhD, Christopher M. Jones, PharmD, Div of Unintentional Injury Prevention, National Center for Injury Prevention and Control, CDC. Corresponding contributor: Leonard J. Paulozzi, lpaulozzi@cdc.gov.
Acknowledgment
Elizabeth Crane, PhD, Center for Behavioral Health Statistics and Quality, Substance Abuse and Mental Health Svcs Admin.
References
- US Government Accountability Office. Methadone-associated overdose deaths: factors contributing to increased deaths and efforts to prevent them. [GAO-09-341] Washington, DC: US Government Accountability Office; 2009. Available at http://www.gao.gov/products/gao-09-341. Accessed May 15, 2012.
- Ballesteros MF, Budnitz DS, Sanford CP, Gilchrist J, Agyekum GA, Butts J. Increase in deaths due to methadone in North Carolina. JAMA 2003;290:40.
- Substance Abuse and Mental Health Services Administration. Data summary: methadone mortality, a 2010 reassessment. Rockville, MD: US Department of Health and Human Services, Substance Abuse and Mental Health Services Administration; 2010. Available at http://www.dpt.samhsa.gov/pdf/methadone_mortality_data_2010.pdf. Accessed April 17, 2012.
- Substance Abuse and Mental Health Services Administration Center for Behavioral Statistics and Quality. The DAWN report: methadone-related emergency department visits involving nonmedical use. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2012. Available at http://www.samhsa.gov/data/2k12/web_dawn_022/methadone_er_nonmedical.pdf. Accessed April 16, 2012.
- Food and Drug Administration. Public health advisory: methadone use for pain control may result in death and life-threatening changes in breathing and heart beat. Rockville, MD: Food and Drug Administration; 2006. Available at http://www.fda.gov/drugs/drugsafety/postmarketdrugsafetyinformationforpatientsandproviders/drugsafetyinformationforheathcareprofessionals/publichealthadvisories/ucm124346.htm. Accessed May 17, 2012.
- Drug Enforcement Administration. Advisory: methadone hydrochloride tables USP 40 mg (dispersible). Washington, DC: Drug Enforcement Administration; 2008. Available at http://www.deadiversion.usdoj.gov/pubs/advisories/methadone_advisory.htm. Accessed May 15, 2012.
- Warner M, Chen L, Makuc D, Anderson R, Miniño A. Drug poisoning deaths in the United States, 1980–2008. NCHS Data Brief, no 81. Hyattsville, MD: National Center for Health Statistics; 2011. Available at http://www.cdc.gov/nchs/data/databriefs/db81.htm. Accessed April 16, 2012.
- Webster LR, Cochella S, Dasgupta N, et al. An analysis of the root causes for opioid-related overdose deaths in the United States. Pain Med 2011;12:S26–35.
- CDC. WONDER [Database]. Atlanta, GA: US Department of Health and Human Services, CDC; 2012. Available at http://wonder.cdc.gov. Accessed May 23, 2012.
- Governale L. Outpatient methadone utilization in the U.S., years 2000–2009. IMS Vector One: National, and SDI, Physician Drug and Diagnosis Audit. Methadone Mortality: A 2010 Reassessment Meeting; July 29–30, 2010; Washington, DC. Rockville, MD: Food and Drug Administration; 2010.
- VonKorff M, Saunders K, Ray GT, et al. De facto long-term opioid therapy for noncancer pain. Clin J Pain 2008;24:521–7.
- Sandoval JA, Furlan A, Mailis-Gagnon A. Oral methadone for chronic noncancer pain. Clin J Pain 2005;21:503–12.
- Saper JR, Lake A, Bain P, et al. A practice guide for continuous opioid therapy for refractory daily headache: patient selection, physician requirements, and treatment monitoring. Headache 2010;50:1175–93.
- Martell BA, O'Connor P, Kerns R, et al. Systematic review: opioid treatment for chronic low back pain: prevalence, efficacy, and association with addiction. Ann Int Med 2007;146:116–27.
- Chou R, Fanciullo GJ, Fine PG, et al. Clinical guidelines for the use of chronic opioid therapy in chronic noncancer pain. J Pain 2009;10:113–30.
* Information about the DEA's ARCOS system is available at http://www.deadiversion.usdoj.gov/arcos.
† Information about the DAWN Medical Examiner system is available at http://www.samhsa.gov/data/2k11/dawn/2k9dawnme/html/dawn2k9me.htm.
|
Key Points |
|
FIGURE 1. Rates of methadone distribution for pain, methadone-related overdose deaths, and methadone prescriptions for pain — United States, 1999–2010
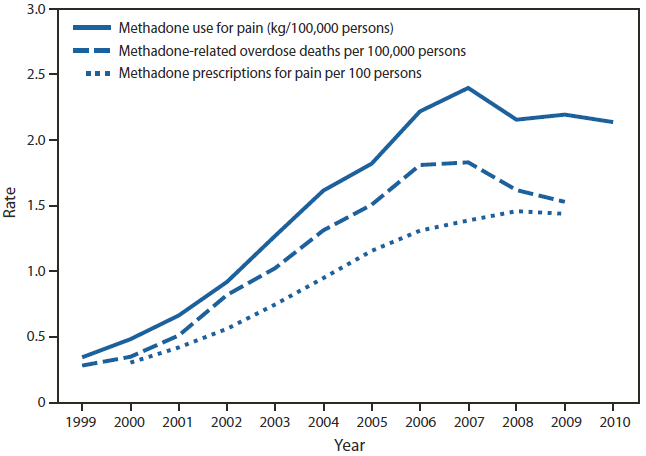
Alternate Text: The figure above shows rates of methadone distribution for pain, methadone-related overdose deaths, and methadone prescriptions for pain in the United States during 1999-2010. The rate of overdose deaths involving methadone in the United States in 2009 was 5.5 times the rate in 1999.
FIGURE 2. Percentage of opioid distribution accounted for by methadone prescribed for pain, by state — United States, 2010
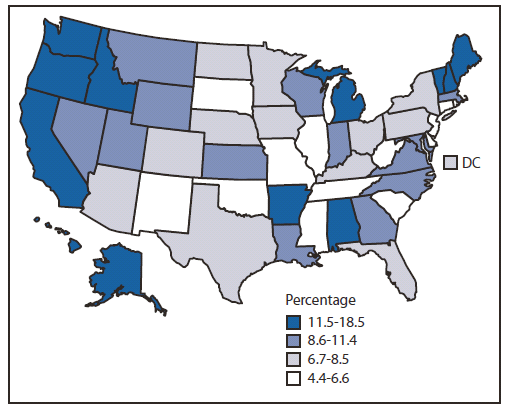
Alternate Text: The figure above shows the percentage of opioid distribution accounted for by methadone prescribed for pain, by state in the United States during 2010. In 2010, methadone accounted for 9.0% of all the morphine milligram equivalents of all major opioids tracked by the Drug Enforcement Administration's Automation of Reports and Consolidated Orders System (ARCOS) other than buprenorphine. This proportion varied by state from 4.5% in New Jersey to 18.5% in Washington.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


