FIGURE 1. Number of laboratory-confirmed cases by wild poliovirus (WPV) type or vaccine-derived poliovirus type 2 (VDPV2) and month of onset, type of supplementary immunization activity (SIA),* and type of vaccine administered --- Nigeria, January 2009--June 2011
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Progress Toward Poliomyelitis Eradication --- Nigeria, January 2010--June 2011
The Global Polio Eradication Initiative (GPEI) was launched by the World Health Assembly in 1988. By 2006, transmission of indigenous wild poliovirus (WPV) was interrupted in all countries except Nigeria, Afghanistan, Pakistan, and India (1). Among the 36 states and Federal Capital Territory of Nigeria, WPV transmission has persisted in eight northern states considered at high risk; in addition, four other northern states have been considered at high risk for WPV transmission (2). In these 12 high-risk states, type 2 circulating vaccine-derived poliovirus (cVDPV2) transmission also was observed during 2005--2011 (3,4). This report updates GPEI progress in Nigeria during January 2010--June 2011 (1,2) and describes activities required to interrupt transmission. In Nigeria, confirmed WPV cases decreased 95%, from 388 in 2009 to 21 in 2010; cVDPV2 cases decreased 82%, from 154 in 2009 to 27 in 2010. However, as of July 26, 2011, Nigeria had reported 24 WPV cases (including one WPV/cVDPV2 coinfection) and 11 cVDPV2 cases during January--June 2011, compared with six WPV cases and 10 cVDPV2 cases during January--June 2010. Despite substantial progress, immunization activities and surveillance sensitivity will need to be enhanced further to interrupt WPV transmission in Nigeria by the end of 2011.
Immunization Activities
The Nigeria routine immunization schedule recommends doses of trivalent OPV types 1, 2, and 3 (tOPV) at birth and, together with diphtheria-tetanus-pertussis vaccine (DTP), at ages 6, 10, and 14 weeks. Because reported OPV coverage can include doses administered during supplementary immunization activities (SIAs), coverage with DTP is a more accurate indicator of OPV administered through routine immunization. Nationally, the proportion of children aged 1 year who had received 3 doses of DTP (DTP3) was 40% in 2006 and 69% in 2010 as estimated by the World Health Organization (WHO) and the United Nations Children's Fund (UNICEF)* using administrative data and multiple surveys. DTP3 coverage in National Immunization Coverage Surveys (NICS) in the eight states† with persistent WPV transmission in 2006 and 2010 was 30% (range: 9%--52%) and 47% (range: 26%--89%), respectively, compared with national coverage of 54% in 2006 and 68% in 2010.
Bivalent OPV types 1 and 3 (bOPV) became available in 2010 and has largely replaced monovalent OPV type 1 (mOPV1) and type 3 (mOPV3) use in SIAs during 2010--2011. Three national SIAs were conducted in 2010 and two during January--June 2011. In the northern states, five subnational SIAs were conducted in 2010 and three during January--June 2011. Various combinations of mOPV1, mOPV3, bOPV, or tOPV were used during these SIAs (Figure 1).
The effectiveness of SIA implementation in the 12 high-risk states§ is monitored by surveys to look for children missed by SIAs in high-risk wards (i.e., subdistricts). The proportion of wards with >10% children missed by SIAs during January 2011--June 2011 was consistently >15% in six states (Kaduna, Kano, Katsina, Kebbi, Niger, and Yobe). The majority of children missed by SIAs lived in households not visited by SIA teams or were not present during vaccination team visits.
Vaccination recall histories of children with nonpolio acute flaccid paralysis (NPAFP) are used to estimate OPV coverage from routine immunization and SIAs among children aged 6--35 months. The proportion of children with NPAFP reported to have never received an OPV dose (i.e., zero-dose children) declined from 30.9% in early 2006 to 10.8% in early 2009 in the eight states with persistent transmission (5). During 2010--2011, downward trends continued (Table), but the overall proportion has not fallen below 5% and ranges as high as 16.7% in Borno. The proportion of children aged 6--35 months with NPAFP who have received ≥3 doses of OPV increased from 24% in early 2006 to 82% in early 2011(5). The targets for the 12 high-risk states are <10% zero-dose children and >80% children with NPAFP with ≥3 OPV doses. In 2011, nine states (Bauchi, Gombe, Jigawa, Kaduna, Katsina, Kebbi, Niger, Sokoto, and Zamfara) met both targets; Kano and Yobe met only the <10% zero-dose target; Borno met neither target.¶
AFP Surveillance
Polio eradication relies on acute flaccid paralysis (AFP) surveillance to identify and confirm poliomyelitis cases by viral isolation. Surveillance performance is monitored using WHO targets for case detection and adequate stool specimen collection (6). NPAFP detection rates meeting the target of at least two cases per 100,000 were achieved in all states during January 2010--June 2011.The national NPAFP detection rate among children aged <15 years was 7.8 per 100,000 during 2010 and an annualized 7.7 per 100,000 during January--June 2011.
The adequate stool specimen target of ≥80% was attained in all states during January 2010--June 2011. Among AFP cases reported nationally, adequate stool specimens were collected from 5,560 (93%) of 6,000 cases during 2010 and 2,788 (93%) of 2,998 cases during January--June 2011. The proportion of districts, or local government areas (LGAs), in the 12 high-risk states meeting both surveillance targets decreased from 89% (254 of 286) in 2009 to 83% (236 of 286) in 2010, to 75% (215 of 286, provisional data) during January--June 2011; many LGAs not meeting both indicators in the high-risk states are contiguous. To supplement laboratory testing of specimens obtained through AFP surveillance, environmental testing of sewage samples for poliovirus began in Kano state in July 2011 (6).
WPV and cVDPV2 Incidence
Eight WPV type 1 (WPV1) cases and 13 WPV type 3 (WPV3) cases were reported during January--December 2010; 16 WPV1 cases (including one WPV1/ cVDPV2 coinfection) and eight WPV3 cases were reported during January--June 2011 (compared with three each during January--June 2010) (Figure 1, Figure 2). During January--June 2011, the WPV1 cases occurred in six persistent-transmission states and the WPV3 cases occurred in three persistent-transmission states (Figure 2). Of 45 WPV cases reported with onset during January 2010--June 2011, 70% occurred in children aged <3 years; 32% were in children reported to have received ≥3 doses OPV, and 27% were in zero-dose children.
Concurrent outbreaks of cVDPV2, which began in Nigeria in 2005 with the use of alternate OPV formulations and were identified with enhanced poliovirus surveillance sensitivity and laboratory screening, resulted in 361 cases reported as of July 26, 2011 (3,4). During January--December 2010, 27 cVDPV2 cases were reported, and during January--June 2011, 10 cVDPV2 cases, one ambiguous VDPV2, and one WPV1/cVDPV2 coinfection were reported (occurring in eight of the 12 high-risk states) (Figure 2). Of 38 cVDPV2 cases with onset during January 2010--June 2011, 69% occurred in children aged <3 years; 36% were in children reported to have received ≥3 doses OPV, and 13% were in zero-dose children.
WPV and VDPV Genomic Sequence Analysis
Analysis of the nucleotide sequence of the VP1 region of each WPV and VDPV isolate is used to investigate transmission links, track international spread, and estimate duration of circulation (6).** The genetic diversity of WPV1 (reflected by the number of virus chains of transmission and genetic clusters) decreased substantially during 2010--2011 in Nigeria. In 2010, four WPV1 genetic clusters were observed, compared with 19 in 2009. However, 13 (68%) of 19 WPV1 isolates tested had <98.5% identity (much less genetic linkage than expected with sensitive AFP surveillance), and two of the five WPV1 clusters observed in 2011 were not detected by AFP surveillance in 2010. In 2010, four WPV3 clusters were observed compared with 20 in 2009. However, eight (44%) of 18 WPV3 isolates tested during 2010--2011 had <98.5% similarity. Among 36 cVDPV2 isolates tested during 2010--2011, 23 (64%) had <98.5% identity.
Reported by
National Primary Health Care Development Agency and Federal Ministry of Health; Country Office of the World Health Organization, Abuja; Poliovirus Laboratory, Univ of Ibadan, Ibadan; Poliovirus Laboratory, Univ of Maiduguri Teaching Hospital, Maiduguri, Nigeria. African Regional Polio Reference Laboratory, National Institute for Communicable Diseases, Johannesburg, South Africa. Vaccine Preventable Diseases, World Health Organization Regional Office for Africa, Brazzaville, Congo; Polio Eradication Dept, World Health Organization, Geneva, Switzerland. Div of Viral Diseases and Global Immunization Div, National Center for Immunization and Respiratory Diseases, CDC. Corresponding contributor: Margaret Hercules, mhercules@cdc.gov, 404-639-8248.
Editorial Note
Northern Nigeria has had ongoing WPV transmission (and more recently, cVDPV2 transmission) because of a weak health-system infrastructure and programmatic limitations such as poor implementation of SIAs, compounded by a loss of public confidence in OPV during 2003 (2,5,7). Beginning in 2009, Nigeria has shown substantial progress in implementation of GPEI activities. Enhanced collaboration with traditional, religious, and political leaders has furthered community acceptance of SIAs; OPV coverage has increased in routine immunization services, and the implementation of SIAs has improved; AFP surveillance indicators have continued to be met in all states; and the number of WPV cases and cVDPV2 cases has markedly decreased (1,2,5--7). However, despite this progress, virologic data indicate that surveillance is not sufficiently sensitive to detect all chains of WPV transmission in a timely manner. The number of WPV cases has increased in 2011; multiple foci of WPV transmission remain endemic, and cVDPV2 transmission persists (1--4).
Nigeria has been a major reservoir for WPV transmission in other countries. Since 2003, WPV of Nigerian origin has been imported into 25 countries, and many countries of West and Central Africa have had repeated importations (8,9). Currently, six countries have WPV3 circulation of Nigerian origin, and cVDPV2 importation into two countries (Chad and Niger) occurred in 2010.
The 2010--2012 GPEI strategic plan set goals of interrupting WPV transmission in two of the remaining four countries with indigenous WPV transmission (Nigeria and India) by the end of 2011 and ending all WPV transmission by the end 2012 (10). Currently, India is on track to meet the 2011 target (1).
For Nigeria, NPAFP dose history provides the major indicators for tracking strategic plan progress in each of the 12 high-risk states (10). During 2011, the NPAFP dose targets have been met in only nine of these states. Additionally, SIA monitoring surveys reveal weaknesses in implementation in some states that are not suggested by the statewide NPAFP dose history indicators. Limitations exist in the NPAFP and SIA indicators: 1) the NPAFP dose history is by parental recall, which might be biased by collection during AFP surveillance; 2) type-specific dose histories vary because of multiple OPV preparations used in SIAs; 3) state NPAFP dose averages might mask substantial variability within states; and 4) SIA monitoring surveys (directed toward the highest-risk areas within LGAs) are not fully standardized in implementation. Although NPAFP dose history and SIA monitoring indicators have been fully met in Jigawa and Sokoto, WPV and cVDPV2 circulation have continued in those states.
Genomic sequence analysis indicates surveillance gaps with some chains of WPV transmission during 2010--2011 not detected for more than a year. State AFP surveillance indicators might mask surveillance gaps occurring among individual LGAs; additionally, surveillance might overlook subpopulations, such as nomads and migrant workers in northern Nigeria, who have limited access to immunization activities and health-care providers. Despite ongoing progress, the continued circulation of WPV and cVDPV2 during 2011 in six states and evidence of limitations in AFP surveillance indicate that substantial further improvements are needed in the quality of implementation of both surveillance and immunization activities to interrupt transmission by the end of 2011. A revised emergency action plan for June--December 2011 is being implemented in Nigeria to enhance AFP surveillance by identifying and improving activities in LGAs not meeting performance criteria, target activities in migrant populations, and better address limitations in SIA implementation by further enhancing training, supervision, monitoring, community engagement, and social mobilization.
References
- CDC. Progress toward interruption of wild poliovirus transmission---worldwide, January 2010--March 2011. MMWR 2011;60:582--6.
- CDC. Progress toward poliomyelitis eradication---Nigeria, January 2009--June 2010. MMWR 2010;59:802--7.
- Wassilak S, Pate MA, Wannamuehler K, et al. Outbreak of type 2 vaccine-derived poliovirus in Nigeria: emergence and widespread circulation in an underimmunized population. J Infect Dis 2011;203:898--909.
- CDC. Update on vaccine-derived polioviruses---worldwide, July 2009--March 2011. MMWR 2011;60:846--50.
- CDC. Progress toward poliomyelitis eradication---Nigeria, 2005--2006. MMWR 2007;56:278--81.
- CDC. Tracking progress toward global polio eradication---worldwide, 2009--2010. MMWR 2011;60:441--5.
- CDC. Progress toward poliomyelitis eradication---Nigeria, January 2004--July 2005. MMWR 2005;54:873--7.
- CDC. Outbreaks following wild poliovirus importations---Europe, Africa, and Asia, January 2009--September 2010. MMWR 2010;59:1393--9.
- CDC. Progress toward interrupting wild poliovirus circulation in countries with reestablished transmission---Africa, 2009--2010. MMWR 2011;60:306--11.
- World Health Organization. Global Polio Eradication Initiative: strategic plan 2010--2012. Geneva, Switzerland: World Health Organization; 2010. Available at http://www.polioeradication.org/content/publications/gpei.strategicplan.2010-2012.eng.may.2010.pdf. Accessed August 2, 2011.
* Available at http://www.who.int/immunization_monitoring/data/nga.pdf.
† Borno, Jigawa, Kano, Katsina, Kebbi, Sokoto, Yobe, and Zamfara.
§ The other four states are Bauchi, Gombe, Kaduna, and Niger.
¶ Reports on progress in the 2010--2012 GPEI Strategic Plan are available at http://www.polioeradication.org/dataandmonitoring/polioeradicationtargets.aspx.
** All isolates are sequenced across the interval encoding the major capsid protein (VP1) (approximately 900 nucleotides) and results are analyzed to determine the likely origin (by state and local government area) of the virus. Isolates within a cluster share >95% VPI nucleotide sequence identity.
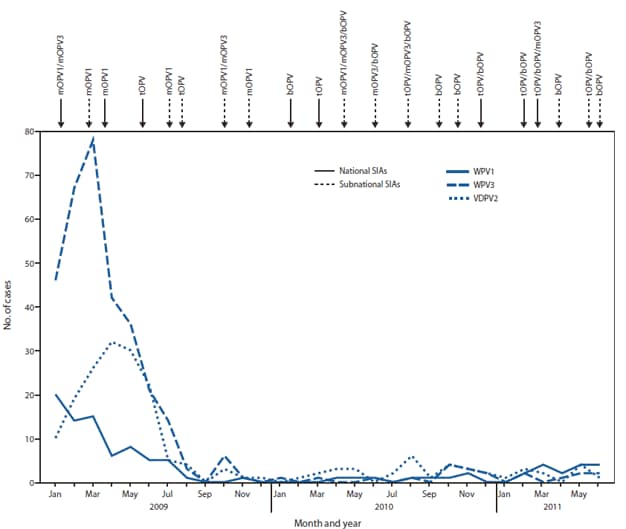
Abbreviations: mOPV1 = monovalent oral polio vaccine (OPV) type 1; mOPV3 = monovalent OPV type 3; tOPV = trivalent OPV; bOPV = bivalent OPV.
* Mass campaign conducted in a short period (days to weeks) during which a dose of OPV is administered to all children aged <5 years, regardless of previous vaccination history. Campaigns can be conducted nationally or in portions of the country.
Alternate Text: The figure above shows the number of laboratory-confirmed cases by wild poliovirus (WPV) type or vaccine-derived poliovirus type 2 (VDPV2) and month of onset, type of supplementary immunization activity (SIA), and type of vaccine administered in Nigeria, during January 2009-June 2011.
FIGURE 2. Wild poliovirus (WPV) types and circulating vaccine-derived poliovirus type 2 (cVDVP2) cases, by period --- Nigeria, January 2010--June 2011
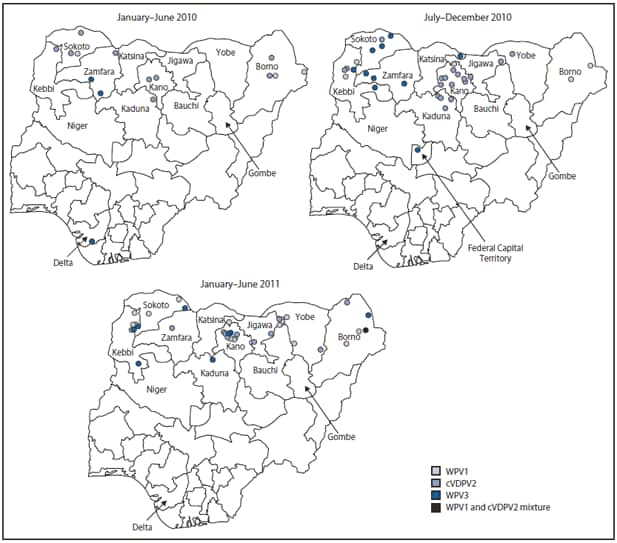
Alternate Text: The figure above shows wild poliovirus (WPV) types and circulating vaccine-derived poliovirus type 2 (cVDVP2) cases, by period in Nigeria during January 2010-June 2011. Eight WPV type 1 (WPV1) cases and 13 WPV type 3 (WPV3) cases were reported during January-December 2010; 16 WPV1 cases (including one WPV1/ cVDPV2 coinfection) and eight WPV3 cases were reported during January-June 2011 (compared with three each during January-June 2010).
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


