Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
HIV Transmitted from a Living Organ Donor --- New York City, 2009
Routine screening of organ donors for human immunodeficiency virus (HIV) infection has made transmission of HIV through organ transplantation rare in the United States. However, despite routine screening, transmission of HIV can be an uncommon complication of organ transplantation and is a public health concern. In 2010, the New York City (NYC) Department of Health and Mental Hygiene (NYC DOHMH) was notified of a potential transplant-related HIV infection. This report summarizes the results of the subsequent public health investigation, which confirmed HIV transmission through transplantation of an organ from a living donor. To reduce the risk for transmission of HIV through living-donor organ transplantation, transplant centers should screen living donors for HIV as close to the time of organ recovery and transplantation as possible, using sensitive tests for both chronic and acute infections, namely, serology and nucleic acid testing (NAT). Furthermore, clinicians should inform transplant candidates of the potential risks for disease transmission and advise donors during evaluation of their obligation to avoid behaviors that would put them at risk for acquiring HIV before organ donation.
Recipient Illness
An adult with hemodialysis-dependent renal failure received a kidney transplant from a living donor at hospital A in NYC in 2009. The recipient did not have any history of sexually transmitted infections (STIs), injection drug use, sex with injection drug users, or other high-risk sexual activity. The recipient received blood transfusions in 2006, but none previously. The recipient tested negative for HIV infection by enzyme immunoassay (EIA) 12 days before the transplant (Figure). The posttransplant clinical course was complicated by multiple hospitalizations for febrile illness, episodes of renal insufficiency, and evaluation for possible rejection of the transplanted kidney. During the year after kidney transplantation, the recipient did not engage in any behaviors that would increase the risk for acquiring HIV. One year after transplant, the recipient was hospitalized with refractory oral and esophageal candidiasis; screening for HIV infection by EIA was positive, and HIV infection was confirmed with a positive Western blot. The recipient's initial CD4 cell count was <100 cells/µL. The advanced immunosuppression was attributed, in part, to the recipient's induction with antithymocyte globulins (an immunomodulator that depletes T-lymphocytes to prevent graft-versus-host disease) and use of mycophenolic acid (a drug that suppresses lymphocyte proliferation, prescribed to prevent rejection of a transplanted kidney).
Donor Illness
The donor was an adult male who underwent evaluation as a potential living donor for kidney transplantation at hospital A in 2009. Consistent with the hospital's protocol, a multidisciplinary team (consisting of a living donor coordinator, nephrologist, transplant surgeon, psychiatrist, social worker, and nutritionist) determined the donor's eligibility by assessing his immunologic compatibility with the recipient, his general health, his psychosocial status, and his willingness to donate. His evaluation revealed a previous diagnosis of syphilis and a history of sex with male partners. Laboratory testing conducted during the initial evaluation 79 days before transplant showed no evidence of infection with HIV by EIA, hepatitis B virus (HBV) by HBV surface antigen testing, or hepatitis C virus (HCV) by anti-HCV serology; a rapid plasma reagin test for syphilis was reactive undiluted (1:1) with a fluorescent treponemal antibody absorption test, also positive, consistent with previously treated syphilis. The donor's kidney was removed without complication; no blood products were administered around the time of transplant. Routine medical evaluation by the transplant team 6 months after the transplant was unremarkable. Approximately 1 year after the transplant, the donor visited his primary-care physician requesting repeat screening for STIs. Serologic testing for HIV antibody by EIA with confirmatory Western blot was positive. The transplant team learned of the living donor's new HIV diagnosis during his follow-up visit 1 year after the transplant.
The diagnosis of HIV in both the donor and recipient raised the possibility of transplant-transmitted HIV infection. NYC DOHMH was notified of the possible transmission, and a public health investigation was initiated.
Public Health Investigation
The donor and recipient each were interviewed in person using a standardized case interview form. Medical records were reviewed, focusing on relevant medical history before and after kidney transplantation, history of HIV testing and evidence of infection, pretransplant evaluation, posttransplant course, blood product transfusion history, other past medical history, history of substance use, sexual history, and other risk factors for HIV infection. The recipient's transplant coordinator, nephrologist, and HIV physician were interviewed separately to review the transplant evaluation and medical course. The donor's primary-care physician and transplant nephrologist also were interviewed.
During the public health investigation, the donor reported unprotected sex with one male partner during the 1 year before the transplant, including the time between his initial evaluation and organ recovery. He did not know his partner's HIV status. He did not report any history of injection drug use, tattoos, or blood transfusions.
Two samples of frozen leukocyte specimens collected from the organ donor 57 and 11 days pretransplant and two frozen serum specimens collected from the recipient 11 days pretransplant and 12 days posttransplant were sent to CDC for HIV testing. HIV NAT on the donor leukocytes collected 57 days pretransplant was negative; however, DNA sequences for three HIV genes (envelope gp41, polymerase, and group-specific antigen p17) were amplified from donor leukocytes collected 11 days pretransplant and sequenced at CDC. Recipient serum collected 11 days pretransplant was nonreactive for HIV-1 RNA by Aptima (Gen-Probe). Recipient serum collected 12 days post transplant was reactive for HIV RNA by Aptima.
On posttransplant day 404, whole blood specimens were obtained from the donor and recipient for phylogenetic analysis at CDC (Figure). The donor had initiated antiretroviral therapy 2 weeks before the specimen was obtained, whereas the recipient had not yet initiated antiretroviral therapy. HIV DNA sequences from donor and recipient peripheral blood lymphocytes collected on day 404 were amplified and sequenced. Sequences from these two specimens were analyzed phylogenetically together with HIV sequences obtained from the donor's frozen leukocyte specimen collected 11 days pretransplant. The gp41, polymerase, and p17 sequences from the donor and recipient were nearly identical, with greater than 98% identity and tight phylogenetic clustering, suggesting that the two viruses are highly related.
Reported by
MA Bernard, MD, J Eavey, MSPH, HW Gortakowski, MPH, C Sabharwal, MD, C Shepard, MD, L Torian, PhD, New York City Dept of Health and Mental Hygiene; L McMurdo, LC Smith, MD, K Valente, New York State Dept of Health. JT Brooks, MD, WM Heneine, PhD, MP Joyce, MD, SM Owen, PhD, A Shankar, MS, W Switzer, MPH, Div of HIV/AIDS Prevention, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention; E Farnon, MD, M Kuehnert, MD, D Seem, Div of Healthcare Quality Promotion, National Center for Emerging and Zoonotic Infections; T Al-Samarrai, MD, P Gounder, MD, CK Kwan, MD, EIS officers, CDC.
Editorial Note
This report describes the first confirmed case of HIV transmission through organ transplantation from a living donor reported since 1989 (1) and the first such transmission documented in the United States since laboratory screening for HIV infection became available in 1985. The time sequence in which HIV was isolated from frozen specimens, tight phylogenetic clustering of HIV sequences from the donor and recipient, and lack of other HIV exposure risk in the recipient confirmed that HIV was transmitted by transplantation of a kidney from a living donor who was infected after screening negative for HIV infection during his initial evaluation. This case highlights the need for repeat HIV screening for all living donors using a combination of HIV serology and NAT, as close to the time of organ donation as logistically feasible, to rule out acute or recent HIV infection in living donors before organ donation.
Reports of confirmed, donor-derived HIV transmission are rare but notable and have important implications for public health when they occur despite screening. The annual number of living donors increased from 1,829 to 6,609 during 1988--2009.* Although the Organ Procurement and Transplantation Network (OPTN) sets national policies for organ allocation, including screening potential donors for HIV and other infections, current OPTN policies do not address screening and counseling for HIV infection in living potential donors.
Deceased organ donors typically are screened for HIV infection at the time of the donor's cardiac death or brain death, which occurs within hours of organ recovery. In contrast, living donors undergo a longer and more comprehensive physical and psychological evaluation (2). In the case described in this report, the time between initial donor evaluation and transplant surgery was 10 weeks. This longer evaluation process for prospective living donors allows potential for acquisition of infections after initial evaluation, emphasizing the need for repeat testing before organ recovery to rule out recently acquired infections. Prospective living donors and transplant candidates frequently have a personal relationship (e.g., they are relatives or friends), which might make a donor less likely to disclose potential risk factors for HIV infection. Therefore, all living donors should be informed about modes of transmission and risk factors for HIV infection and should be counseled to avoid behaviors that would place them at risk for acquiring HIV infection before organ recovery (3). For persons with a history of previous high-risk behavior (e.g., high-risk sexual activity or injection drug use) identified during evaluation (3), individualized counseling and a detailed discussion of specific strategies to avoid risk behaviors should be provided.
The availability of NAT now permits detection of HIV infection before antibodies develop and are detectable by serology. The window between time of HIV infection and time of development of detectable HIV-specific antibodies ranges from 3 to 8 weeks, whereas with NAT, the window (i.e., the "eclipse period" for the time from infection to detection of virus in blood) is estimated to be 8--10 days (4,5). For blood and tissue donation, the combination of HIV NAT and serology is used to screen all donors. However, HIV NAT is not consistently used for organ donor screening. A recent survey of organ procurement organizations (OPO) found that 52% of OPOs report always performing HIV NAT on deceased donors, whereas 24% of OPOs never perform HIV NAT (6). For deceased potential donors who screen negative by HIV serology, NAT can reduce the risk for HIV transmission from an undetected infection during the window period by approximately 68% (7,8). This reduction reflects differences in the timeframe of detection between the two tests. U.S. Public Health Service guidelines have recommended serologic screening for HIV infection in potential organ donors since 1994 (3). A forthcoming updated revision to those guidelines, currently in draft form, likely will include consideration of NAT in addition to serology to screen for HIV infection in all potential organ donors, living and deceased.
In the case described in this report, the donor was screened for HIV infection by EIA 10 weeks before organ procurement but was not rescreened closer to the date of transplant surgery. Despite an initial negative EIA screening and counseling for behavioral risk reduction, living donors can acquire HIV infection between the initial evaluation and organ recovery. To reduce the risk for transplant-transmitted HIV infection, living donors should be rescreened with both HIV serologic tests and NAT as close to the time of organ recovery as logistically feasible, but no longer than 7 days before organ donation (Box). In addition, clinicians should advise living donors of their obligation to avoid behaviors that would put them at risk for acquiring HIV before organ donation. Because NAT cannot detect HIV infections during the eclipse period, all transplant candidates should be informed during the evaluation process that despite donor screening, a very small risk remains that they could acquire HIV or other infections as a result of transplantation. All suspected cases of transplant-associated HIV transmission should be reported to appropriate public health authorities and to OPTN.
References
- Quarto M, Germinario C, Fontana A, Barbuti S. HIV transmission through kidney transplantation from a living related donor. N Engl J Med 1989;320:1754.
- Fischer SA, Avery RK. Screening of donor and recipient prior to solid organ transplantation. Am J Transplant 2009;9 Suppl 4:S7--18.
- CDC. Guidelines for preventing transmission of human immunodeficiency virus through transplantation of human tissue and organs. MMWR 1994;43(No. RR-8).
- Fiebig EW, Wright DJ, Rawal BD, et al. Dynamics of HIV viremia and antibody seroconversion in plasma donors: implications for diagnosis and staging of primary HIV infection. AIDS 2003;17:1871--9.
- Keele BF, Giorgi EE, Salazar-Gonzalez JF, et al. Identification and characterization of transmitted and early founder virus envelopes in primary HIV-1 infection. Proc Natl Acad Sci USA 2008;105:7552--7.
- Kucirka LM, Alexander C, Namuyinga R, Hanrahan C, Montgomery RA, Segev DL. Viral nucleic acid testing (NAT) and OPO-level disposition of high-risk donor organs. Am J Transplant 2009;9:620--8.
- Ellingson K, Seem D, Nowicki M, et al. Estimated risk of human immunodeficiency virus and hepatitis C virus infection among potential organ donors from 17 organ procurement organizations in the United States. Am J Transplant 2011. In press.
- Kucirka LM, Sarathy H, Govindan P, et al. Risk of window period HIV infection in high infectious risk donors: systematic review and meta-analysis. Am J Transplant 2011; March 2 [Epub ahead of print].
* Based on Organ Procurement and Transplantation Network data as of January 14, 2011.
What is already known on this topic?
Routine laboratory screening of organ donors for human immunodeficiency virus (HIV) infection, introduced in 1985, has made transmission of HIV through organ transplantation rare in the United States. However, no national policy exists for the type or timing of HIV screening tests used for living donors.
What is added by this report?
This report describes the first documented case in the United States of HIV transmission through transplantation of an organ from a living donor, despite screening using serologic testing.
What are the implications for public health practice?
All prospective living organ donors should have their initial serologic tests for HIV supplemented with repeat testing with a combination of an HIV serologic test and a nucleic acid test as close to the time of organ donation as possible, but no longer than 7 days before organ donation. Clinicians should advise living donors of their obligation to avoid behaviors that would put them at risk for acquiring HIV before organ donation.
FIGURE. Timeline of events involving HIV transmission from a living organ donor --- New York City, 2009
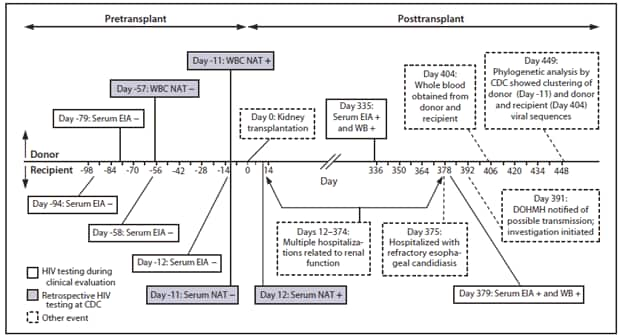
Abbreviations: HIV = human immunodeficiency virus; EIA = HIV enzyme immunoassay; HIV = human immunodeficiency virus; NAT = nucleic acid test; WB = HIV Western blot; WBC = white blood cell; DOHMH = New York City Department of Health and Mental Hygiene.
Alternate Text: The figure above shows a timeline of events involving HIV transmission from a living organ donor in New York City in 2009. An adult with hemodialysis-dependent renal failure received a kidney transplant from a living donor at hospital A during 2009. The recipient did not have any history of sexually transmitted infections, injection drug use, sex with injection drug users, male-male sex, or other high-risk sexual activity. The recipient received blood transfusions in 2006, but none previously. The recipient tested negative for HIV infection by enzyme immunoassay 12 days before the transplant.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


