FIGURE 1. Recommended immunization schedule for persons aged 0 through 6 years --- United States, 2011 (for those who fall behind or start late, see the catch-up schedule [Table])
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Recommended Immunization Schedules for Persons Aged 0 Through 18 Years — United States, 2011
Please note: An erratum has been published for this article. To view the erratum, please click here.
Each year, the Advisory Committee on Immunization Practices (ACIP) publishes immunization schedules for persons aged 0 through 18 years. These schedules summarize recommendations for currently licensed vaccines for children aged 18 years and younger and include recommendations in effect as of December 21, 2010. Changes to the previous schedules (1) include the following:
- Guidance has been added for the hepatitis B vaccine schedule for children who did not receive a birth dose (2).
- Information on use of 13-valent pneumococcal conjugate vaccine has been added (3).
- Guidance has been added for administration of 1 or 2 doses of seasonal influenza vaccine based upon the child's history of monovalent 2009 H1N1 vaccination (4).
- Use of tetanus and diphtheria toxoids, and acellular pertussis (Tdap) vaccine among children aged 7 through 10 years who are incompletely vaccinated against pertussis is addressed, and reference to a specified interval between tetanus and diphtheria toxoids (Td) and Tdap vaccination has been removed (5).
- Footnotes for the use of human papillomavirus (HPV) vaccine have been condensed.
- A routine 2-dose schedule of quadrivalent meningococcal conjugate vaccine (MCV4) for certain persons at high risk for meningococcal disease, and recommendations for a booster dose of MCV4 have been added (6).
- Guidance for use of Haemophilus influenzae type b (Hib) vaccine in persons aged 5 years and older in the catch-up schedule has been condensed.
The National Childhood Vaccine Injury Act requires that health-care providers provide parents or patients with copies of Vaccine Information Statements before administering each dose of the vaccines listed in the schedules. Additional information is available from state health departments and from CDC at http://www.cdc.gov/vaccines/pubs/vis/default.htm.
Detailed recommendations for using vaccines are available from ACIP statements (available at http://www.cdc.gov/vaccines/pubs/acip-list.htm) and the 2009 Red Book (7). Guidance regarding the Vaccine Adverse Event Reporting System form is available online (http://www.vaers.hhs.gov) or by telephone (800-822-7967).
References
- CDC. Recommended immunization schedules for persons aged 0--18 years---United States, 2010. MMWR 2009;58(51&52).
- CDC. A comprehensive immunization strategy to eliminate transmission of hepatitis B virus infection in the United States. Recommendations of the Advisory Committee on Immunization Practices (ACIP): part 1: immunization of infants, children, and adolescents. MMWR 2005;54(No. RR-16):10.
- CDC. Prevention of pneumococcal disease among infants and children---use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine. Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR 2010;59(No. RR-11).
- CDC. Prevention and control of influenza with vaccines. Recommendations of the Advisory Committee on Immunization Practices (ACIP), 2010. MMWR 2010;59(No. RR-8).
- CDC. Updated recommendations for use of tetanus toxoid, reduced diphtheria toxoid and acellular pertussis (Tdap) vaccine from the Advisory Committee on Immunization Practices, 2010. MMWR 2011;60:13--5.
- CDC. Updated recommendations for use of meningococcal conjugate vaccines---Advisory Committee on Immunization Practices (ACIP), 2010. MMWR 2011;60:72--6.
- American Academy of Pediatrics. Active and passive immunization. In: Pickering LK, Baker CJ, Kimberlin DW, Long SS, eds. 2009 red book: report of the Committee on Infectious Diseases. 28th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2009.
The recommended immunization schedules for persons aged 0 through 18 years and the catch-up immunization schedule for 2011 have been approved by the Advisory Committee on Immunization Practices, the American Academy of Pediatrics, and the American Academy of Family Physicians.
Suggested citation: Centers for Disease Control and Prevention. Recommended immunization schedules for persons aged 0--18 years---United States, 2011. MMWR 2011;60(5).
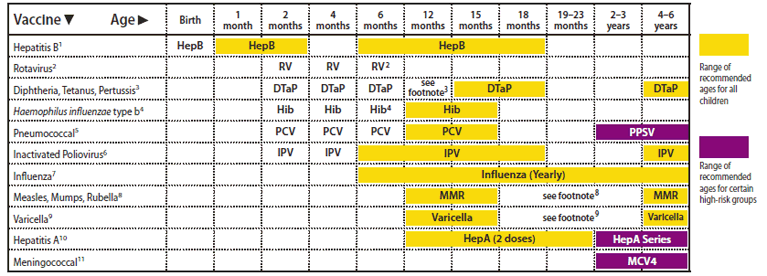
Alternate Text: The figure above shows the recommended immunization schedule for 2011 for persons aged 0 through 6 years in the United States
This schedule includes recommendations in effect as of December 21, 2010. Any dose not administered at the recommended age should be administered at a subsequent visit, when indicated and feasible. The use of a combination vaccine generally is preferred over separate injections of its equivalent component vaccines. Considerations should include provider assessment, patient preference, and the potential for adverse events. Providers should consult the relevant Advisory Committee on Immunization Practices statement for detailed recommendations: http://www.cdc.gov/vaccines/pubs/acip-list.htm. Clinically significant adverse events that follow immunization should be reported to the Vaccine Adverse Event Reporting System (VAERS) at http://www.vaers.hhs.gov or by telephone, 800-822-7967.
1. Hepatitis B vaccine (HepB). (Minimum age: birth)
At birth:
- Administer monovalent HepB to all newborns before hospital discharge.
- If mother is hepatitis B surface antigen (HBsAg)-positive, administer HepB and 0.5 mL of hepatitis B immune globulin (HBIG) within 12 hours of birth.
- If mother's HBsAg status is unknown, administer HepB within 12 hours of birth. Determine mother's HBsAg status as soon as possible and, if HBsAg-positive, administer HBIG (no later than age 1 week).
Doses following the birth dose:
- The second dose should be administered at age 1 or 2 months. Monovalent HepB should be used for doses administered before age 6 weeks.
- Infants born to HBsAg-positive mothers should be tested for HBsAg and antibody to HBsAg 1 to 2 months after completion of at least 3 doses of the HepB series, at age 9 through 18 months (generally at the next well-child visit).
- Administration of 4 doses of HepB to infants is permissible when a combination vaccine containing HepB is administered after the birth dose.
- Infants who did not receive a birth dose should receive 3 doses of HepB on a schedule of 0, 1, and 6 months.
- The final (3rd or 4th) dose in the HepB series should be administered no earlier than age 24 weeks.
2. Rotavirus vaccine (RV). (Minimum age: 6 weeks)
- Administer the first dose at age 6 through 14 weeks (maximum age: 14 weeks 6 days). Vaccination should not be initiated for infants aged 15 weeks 0 days or older.
- The maximum age for the final dose in the series is 8 months 0 days
- If Rotarix is administered at ages 2 and 4 months, a dose at 6 months is not indicated.
3. Diphtheria and tetanus toxoids and acellular pertussis vaccine (DTaP).
(Minimum age: 6 weeks)
- The fourth dose may be administered as early as age 12 months, provided at least 6 months have elapsed since the third dose.
4. Haemophilus influenzae type b conjugate vaccine (Hib). (Minimum age: 6 weeks)
- If PRP-OMP (PedvaxHIB or Comvax [HepB-Hib]) is administered at ages 2 and 4 months, a dose at age 6 months is not indicated.
- Hiberix should not be used for doses at ages 2, 4, or 6 months for the primary series but can be used as the final dose in children aged 12 months through 4 years.
5. Pneumococcal vaccine. (Minimum age: 6 weeks for pneumococcal conjugate vaccine [PCV]; 2 years for pneumococcal polysaccharide vaccine [PPSV])
- PCV is recommended for all children aged younger than 5 years. Administer 1 dose of PCV to all healthy children aged 24 through 59 months who are not completely vaccinated for their age.
- A PCV series begun with 7-valent PCV (PCV7) should be completed with 13-valent PCV (PCV13).
- A single supplemental dose of PCV13 is recommended for all children aged 14 through 59 months who have received an age-appropriate series of PCV7.
- A single supplemental dose of PCV13 is recommended for all children aged 60 through 71 months with underlying medical conditions who have received an age-appropriate series of PCV7.
- The supplemental dose of PCV13 should be administered at least 8 weeks after the previous dose of PCV7. See MMWR 2010:59(No. RR-11).
- Administer PPSV at least 8 weeks after last dose of PCV to children aged 2 years or older with certain underlying medical conditions, including a cochlear implant.
6. Inactivated poliovirus vaccine (IPV). (Minimum age: 6 weeks)
- If 4 or more doses are administered prior to age 4 years an additional dose should be administered at age 4 through 6 years.
- The final dose in the series should be administered on or after the fourth birthday and at least 6 months following the previous dose.
7. Influenza vaccine (seasonal). (Minimum age: 6 months for trivalent inactivated influenza vaccine [TIV]; 2 years for live, attenuated influenza vaccine [LAIV])
- For healthy children aged 2 years and older (i.e., those who do not have underlying medical conditions that predispose them to influenza complications), either LAIV or TIV may be used, except LAIV should not be given to children aged 2 through 4 years who have had wheezing in the past 12 months.
- Administer 2 doses (separated by at least 4 weeks) to children aged 6 months through 8 years who are receiving seasonal influenza vaccine for the first time or who were vaccinated for the first time during the previous influenza season but only received 1 dose.
- Children aged 6 months through 8 years who received no doses of monovalent 2009 H1N1 vaccine should receive 2 doses of 2010--2011 seasonal influenza vaccine. See MMWR 2010;59(No. RR-8):33--34.
8. Measles, mumps, and rubella vaccine (MMR). (Minimum age: 12 months)
- The second dose may be administered before age 4 years, provided at least 4 weeks have elapsed since the first dose.
9. Varicella vaccine. (Minimum age: 12 months)
- The second dose may be administered before age 4 years, provided at least 3 months have elapsed since the first dose.
- For children aged 12 months through 12 years the recommended minimum interval between doses is 3 months. However, if the second dose was administered at least 4 weeks after the first dose, it can be accepted as valid.
10. Hepatitis A vaccine (HepA). (Minimum age: 12 months)
- Administer 2 doses at least 6 months apart.
- HepA is recommended for children aged older than 23 months who live in areas where vaccination programs target older children, who are at increased risk for infection, or for whom immunity against hepatitis A is desired.
11. Meningococcal conjugate vaccine, quadrivalent (MCV4). (Minimum age: 2 years)
- Administer 2 doses of MCV4 at least 8 weeks apart to children aged 2 through 10 years with persistent complement component deficiency and anatomic or functional asplenia, and 1 dose every 5 years thereafter.
- Persons with human immunodeficiency virus (HIV) infection who are vaccinated with MCV4 should receive 2 doses at least 8 weeks apart.
- Administer 1 dose of MCV4 to children aged 2 through 10 years who travel to countries with highly endemic or epidemic disease and during outbreaks caused by a vaccine serogroup.
- Administer MCV4 to children at continued risk for meningococcal disease who were previously vaccinated with MCV4 or meningococcal polysaccharide vaccine after 3 years if the first dose was administered at age 2 through 6 years.
The Recommended Immunization Schedules for Persons Aged 0 Through 18 Years are approved by the Advisory Committee on Immunization Practices (http://www.cdc.gov/vaccines/recs/acip), the American Academy of Pediatrics (http://www.aap.org), and the American Academy of Family Physicians (http://www.aafp.org). U.S. Department of Health and Human Services • Centers for Disease Control and Prevention
FIGURE 2. Recommended immunization schedule for persons aged 7 through 18 years --- United States, 2011 (for those who fall behind or start late, see the schedule below and the catch-up schedule [Table])
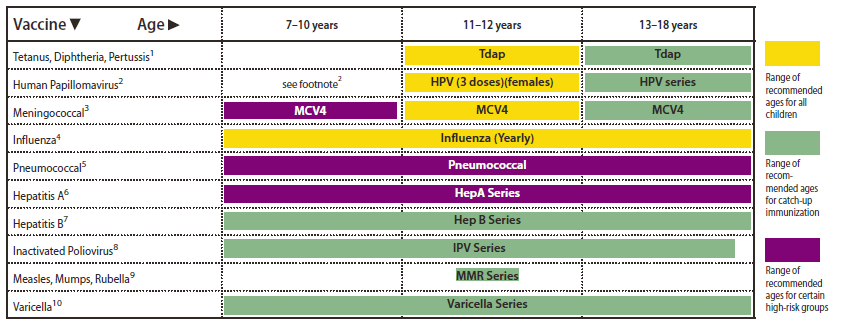
Alternate Text: The figure above shows the recommended immunization schedule for 2011 for persons aged 7 through 18 years in the United States
This schedule includes recommendations in effect as of December 21, 2010. Any dose not administered at the recommended age should be administered at a subsequent visit, when indicated and feasible. The use of a combination vaccine generally is preferred over separate injections of its equivalent component vaccines. Considerations should include provider assessment, patient preference, and the potential for adverse events. Providers should consult the relevant Advisory Committee on Immunization Practices statement for detailed recommendations: http://www.cdc.gov/vaccines/pubs/acip-list.htm. Clinically significant adverse events that follow immunization should be reported to the Vaccine Adverse Event Reporting System (VAERS) at http://www.vaers.hhs.gov or by telephone, 800-822-7967.
1. Tetanus and diphtheria toxoids and acellular pertussis vaccine (Tdap). (Minimum age: 10 years for Boostrix and 11 years for Adacel)
- Persons aged 11 through 18 years who have not received Tdap should receive a dose followed by Td booster doses every 10 years thereafter.
- Persons aged 7 through 10 years who are not fully immunized against pertussis (including those never vaccinated or with unknown pertussis vaccination status) should receive a single dose of Tdap. Refer to the catch-up schedule if additional doses of tetanus and diphtheria toxoid--containing vaccine are needed.
- Tdap can be administered regardless of the interval since the last tetanus and diphtheria toxoid--containing vaccine.
2. Human papillomavirus vaccine (HPV). (Minimum age: 9 years)
- Quadrivalent HPV vaccine (HPV4) or bivalent HPV vaccine (HPV2) is recommended for the prevention of cervical precancers and cancers in females.
- HPV4 is recommended for prevention of cervical precancers, cancers, and genital warts in females.
- HPV4 may be administered in a 3-dose series to males aged 9 through 18 years to reduce their likelihood of genital warts.
- Administer the second dose 1 to 2 months after the first dose and the third dose 6 months after the first dose (at least 24 weeks after the first dose).
3. Meningococcal conjugate vaccine, quadrivalent (MCV4). (Minimum age: 2 years)
- Administer MCV4 at age 11 through 12 years with a booster dose at age 16 years.
- Administer 1 dose at age 13 through 18 years if not previously vaccinated.
- Persons who received their first dose at age 13 through 15 years should receive a booster dose at age 16 through 18 years.
- Administer 1 dose to previously unvaccinated college freshmen living in a dormitory.
- Administer 2 doses at least 8 weeks apart to children aged 2 through 10 years with persistent complement component deficiency and anatomic or functional asplenia, and 1 dose every 5 years thereafter.
- Persons with HIV infection who are vaccinated with MCV4 should receive 2 doses at least 8 weeks apart.
- Administer 1 dose of MCV4 to children aged 2 through 10 years who travel to countries with highly endemic or epidemic disease and during outbreaks caused by a vaccine serogroup.
- Administer MCV4 to children at continued risk for meningococcal disease who were previously vaccinated with MCV4 or meningococcal polysaccharide vaccine after 3 years (if first dose administered at age 2 through 6 years) or after 5 years (if first dose administered at age 7 years or older).
4. Influenza vaccine (seasonal).
- For healthy nonpregnant persons aged 7 through 18 years (i.e., those who do not have underlying medical conditions that predispose them to influenza complications), either LAIV or TIV may be used.
- Administer 2 doses (separated by at least 4 weeks) to children aged 6 months through 8 years who are receiving seasonal influenza vaccine for the first time or who were vaccinated for the first time during the previous influenza season but only received 1 dose.
- Children 6 months through 8 years of age who received no doses of monovalent 2009 H1N1 vaccine should receive 2 doses of 2010-2011 seasonal influenza vaccine. See MMWR 2010;59(No. RR-8):33--34.
5. Pneumococcal vaccines.
- A single dose of 13-valent pneumococcal conjugate vaccine (PCV13) may be administered to children aged 6 through 18 years who have functional or anatomic asplenia, HIV infection or other immunocompromising condition, cochlear implant or CSF leak. See MMWR 2010;59(No. RR-11).
- The dose of PCV13 should be administered at least 8 weeks after the previous dose of PCV7.
- Administer pneumococcal polysaccharide vaccine at least 8 weeks after the last dose of PCV to children aged 2 years or older with certain underlying medical conditions, including a cochlear implant. A single revaccination should be administered after 5 years to children with functional or anatomic asplenia or an immunocompromising condition.
6. Hepatitis A vaccine (HepA).
- Administer 2 doses at least 6 months apart.
- HepA is recommended for children aged older than 23 months who live in areas where vaccination programs target older children, or who are at increased risk for infection, or for whom immunity against hepatitis A is desired.
7. Hepatitis B vaccine (HepB).
- Administer the 3-dose series to those not previously vaccinated. For those with incomplete vaccination, follow the catch-up recommendations (Table).
- A 2-dose series (separated by at least 4 months) of adult formulation Recombivax HB is licensed for children aged 11 through 15 years.
8. Inactivated poliovirus vaccine (IPV).
- The final dose in the series should be administered on or after the fourth birthday and at least 6 months following the previous dose.
- If both OPV and IPV were administered as part of a series, a total of 4 doses should be administered, regardless of the child's current age.
9. Measles, mumps, and rubella vaccine (MMR).
- The minimum interval between the 2 doses of MMR is 4 weeks.
10. Varicella vaccine.
- For persons aged 7 through 18 years without evidence of immunity (see MMWR 2007;56[No. RR-4]), administer 2 doses if not previously vaccinated or the second dose if only 1 dose has been administered.
- For persons aged 7 through 12 years, the recommended minimum interval between doses is 3 months. However, if the second dose was administered at least 4 weeks after the first dose, it can be accepted as valid.
- For persons aged 13 years and older, the minimum interval between doses is 4 weeks.
The Recommended Immunization Schedules for Persons Aged 0 Through 18 Years are approved by the Advisory Committee on Immunization Practices (http://www.cdc.gov/vaccines/recs/acip), the American Academy of Pediatrics (http://www.aap.org), and the American Academy of Family Physicians (http://www.aafp.org). U.S. Department of Health and Human Services • Centers for Disease Control and Prevention
TABLE. Catch-up immunization schedule for persons aged 4 months through 18 years who start late or who are more than 1 month behind --- United States, 2011
The table below provides catch-up schedules and minimum intervals between doses for children whose vaccinations have been delayed. A vaccine series does not need to be restarted, regardless of the time that has elapsed between doses. Use the section appropriate for the child's age.
|
PERSONS AGED 4 MONTHS THROUGH 6 YEARS |
|||||
|
Vaccine |
Minimum Age for Dose 1 |
Minimum Interval Between Doses |
|||
|
Dose 1 to Dose 2 |
Dose 2 to Dose 3 |
Dose 3 to Dose 4 |
Dose 4 to Dose 5 |
||
|
Hepatitis B1 |
Birth |
4 weeks |
8 weeks (and at least 16 weeks after first dose) |
||
|
Rotavirus2 |
6 wks |
4 weeks |
4 weeks2 |
||
|
Diphtheria, Tetanus, Pertussis3 |
6 wks |
4 weeks |
4 weeks |
6 months |
6 months3 |
|
Haemophilus influenzae type b4 |
6 wks |
4 weeks if first dose administered at younger than age 12 months 8 weeks (as final dose) if first dose administered at age 12--14 months No further doses needed if first dose administered at age 15 months or older |
4 weeks4 if current age is younger than 12 months 8 weeks (as final dose)4 if current age is 12 months or older and first dose administered at younger than age 12 months and second dose administered at younger than 15 months No further doses needed if previous dose administered at age 15 months or older |
8 weeks (as final dose) This dose only necessary for children aged 12 months through 59 months who received 3 doses before age 12 months |
|
|
Pneumococcal5 |
6 wks |
4 weeks if first dose administered at younger than age 12 months 8 weeks (as final dose for healthy children) if first dose administered at age 12 months or older or current age 24 through 59 months No further doses needed for healthy children if first dose administered at age 24 months or older |
4 weeks if current age is younger than 12 months 8 weeks (as final dose for healthy children) if current age is 12 months or older No further doses needed for healthy children if previous dose administered at age 24 months or older |
8 weeks (as final dose) This dose only necessary for children aged 12 months through 59 months who received 3 doses before age 12 months or for children at high risk who received 3 doses at any age |
|
|
Inactivated Poliovirus6 |
6 wks |
4 weeks |
4 weeks |
6 months6 |
|
|
Measles, Mumps, Rubella7 |
12 mos |
4 weeks |
|||
|
Varicella8 |
12 mos |
3 months |
|||
|
Hepatitis A9 |
12 mos |
6 months |
|||
|
PERSONS AGED 7 THROUGH 18 YEARS |
|||||
|
Tetanus,Diphtheria/ Tetanus,Diphtheria,Pertussis10 |
7 yrs10 |
4 weeks |
4 weeks if first dose administered at younger than age 12 months 6 months if first dose administered at 12 months or older |
6 months if first dose administered at younger than age 12 months |
|
|
Human Papillomavirus11 |
9 yrs |
Routine dosing intervals are recommended (females)11 |
|||
|
Hepatitis A9 |
12 mos |
6 months |
|||
|
Hepatitis B1 |
Birth |
4 weeks |
8 weeks (and at least 16 weeks after first dose) |
||
|
Inactivated Poliovirus6 |
6 wks |
4 weeks |
4 weeks6 |
6 months6 |
|
|
Measles, Mumps, Rubella7 |
12 mos |
4 weeks |
|||
|
Varicella8 |
12 mos |
3 months if person is younger than age 13 years 4 weeks if person is aged 13 years or older |
|||
1. Hepatitis B vaccine (HepB).
- Administer the 3-dose series to those not previously vaccinated.
- The minimum age for the third dose of HepB is 24 weeks.
- A 2-dose series (separated by at least 4 months) of adult formulation Recombivax HB is licensed for children aged 11 through 15 years.
2. Rotavirus vaccine (RV).
- The maximum age for the first dose is 14 weeks 6 days. Vaccination should not be initiated for infants aged 15 weeks 0 days or older.
- The maximum age for the final dose in the series is 8 months 0 days.
- If Rotarix was administered for the first and second doses, a third dose is not indicated.
3. Diphtheria and tetanus toxoids and acellular pertussis vaccine (DTaP).
- The fifth dose is not necessary if the fourth dose was administered at age 4 years or older.
4. Haemophilus influenzae type b conjugate vaccine (Hib).
- 1 dose of Hib vaccine should be considered for unvaccinated persons aged 5 years or older who have sickle cell disease, leukemia, or HIV infection, or who have had a splenectomy.
- If the first 2 doses were PRP-OMP (PedvaxHIB or Comvax), and administered at age 11 months or younger, the third (and final) dose should be administered at age 12 through 15 months and at least 8 weeks after the second dose.
- If the first dose was administered at age 7 through 11 months, administer the second dose at least 4 weeks later and a final dose at age 12 through 15 months.
5. Pneumococcal vaccine.
- Administer 1 dose of 13-valent pneumococcal conjugate vaccine (PCV13) to all healthy children aged 24 through 59 months with any incomplete PCV schedule (PCV7 or PCV13).
- For children aged 24 through 71 months with underlying medical conditions, administer 1 dose of PCV13 if 3 doses of PCV were received previously or administer 2 doses of PCV13 at least 8 weeks apart if fewer than 3 doses of PCV were received previously.
- A single dose of PCV13 is recommended for certain children with underlying medical conditions through 18 years of age. See age-specific schedules for details.
- Administer pneumococcal polysaccharide vaccine (PPSV) to children aged 2 years or older with certain underlying medical conditions, including a cochlear implant, at least 8 weeks after the last dose of PCV. A single revaccination should be administered after 5 years to children with functional or anatomic asplenia or an immunocompromising condition. See MMWR 2010;59(No. RR-11).
6. Inactivated poliovirus vaccine (IPV).
- The final dose in the series should be administered on or after the fourth birthday and at least 6 months following the previous dose.
- A fourth dose is not necessary if the third dose was administered at age 4 years or older and at least 6 months following the previous dose.
- In the first 6 months of life, minimum age and minimum intervals are only recommended if the person is at risk for imminent exposure to circulating poliovirus (i.e., travel to a polio-endemic region or during an outbreak).
7. Measles, mumps, and rubella vaccine (MMR).
- Administer the second dose routinely at age 4 through 6 years. The minimum interval between the 2 doses of MMR is 4 weeks.
8. Varicella vaccine.
- Administer the second dose routinely at age 4 through 6 years.
- If the second dose was administered at least 4 weeks after the first dose, it can be accepted as valid.
9. Hepatitis A vaccine (HepA).
- HepA is recommended for children aged older than age 23 months who live in areas where vaccination programs target older children, or who are at increased risk for infection, or for whom immunity against hepatitis A is desired.
10. Tetanus and diphtheria toxoids (Td) and tetanus and diphtheria toxoids and acellular pertussis vaccine (Tdap).
- Doses of DTaP are counted as part of the Td/Tdap series.
- Tdap should be substituted for a single dose of Td in the catch-up series for children aged 7 through 10 years or as a booster for children aged 11 through 18 years; use Td for other doses.
11. Human papillomavirus vaccine (HPV).
- Administer the series to females at age 13 through 18 years if not previously vaccinated or have not completed the vaccine series.
- Quadrivalent HPV vaccine (HPV4) may be administered in a 3-dose series to males aged 9 through 18 years to reduce their likelihood of genital warts.
- Use recommended routine dosing intervals for series catch-up (i.e., the second and third doses should be administered at 1 to 2 and 6 months after the first dose). The minimum interval between the first and second doses is 4 weeks. The minimum interval between the second and third doses is 12 weeks, and the third dose should be administered at least 24 weeks after the first dose.
Information about reporting reactions after immunization is available online at http://www.vaers.hhs.gov or by telephone, 800-822-7967. Suspected cases of vaccine-preventable diseases should be reported to the state or local health department. Additional information, including precautions and contraindications for immunization, is available from the National Center for Immunization and Respiratory Diseases at http://www.cdc.gov/vaccines or telephone, 800-CDC-INFO (800-232-4636).
U.S. Department of Health and Human Services • Centers for Disease Control and Prevention
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of Health and Human Services. |
All MMWR HTML versions of articles are electronic conversions from typeset documents. This conversion might result in character translation or format errors in the HTML version. Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr) and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


