Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Discordant Results from Reverse Sequence Syphilis Screening --- Five Laboratories, United States, 2006--2010
CDC recommends syphilis serologic screening with a nontreponemal test, such as the rapid plasma reagin (RPR) or Venereal Disease Research Laboratory (VDRL) test, to identify persons with possible untreated infection; this screening is followed by confirmation using one of several treponemal tests. Recently, the availability of automatable treponemal enzyme and chemiluminescence immunoassays (EIA/CIA) has led some laboratories to adopt a reverse sequence of screening in which a treponemal EIA/CIA is performed first, followed by testing of reactive sera with a nontreponemal test. To better understand the performance of reverse sequence screening for syphilis, CDC analyzed data from five laboratories that used reverse sequence screening during 2006--2010. This report describes the results of that analysis, which indicated that among sera reactive on initial screening with a treponemal EIA/CIA, 56.7% had a nonreactive RPR test. Among these discordant sera, 31.6% also were nonreactive by treponemal testing using Treponema pallidum particle agglutination (TP-PA) or fluorescent treponemal antibody absorbed (FTA-ABS) tests. Among discordant sera, the rate of nonreactive confirmatory treponemal tests was 2.9 times higher in a population with low prevalence of syphilis, suggesting that the low-prevalence population had a higher percentage of false-positive test results. Although CDC continues to recommend the traditional algorithm with reactive nontreponemal tests confirmed by treponemal testing, in this report CDC offers additional recommendations if reverse sequence syphilis screening is used.
Treponema pallidum, the bacterium that causes syphilis, cannot be cultured. As a result, serologic testing is the method most often used to diagnose syphilis in patients with suspected disease. Because syphilis can be asymptomatic, serologic screening is recommended for 1) persons at high risk, to detect latent infections; 2) pregnant women, to prevent congenital syphilis; and 3) blood donors, to prevent transmission through transfusion. Serodiagnosis of syphilis involves the detection of two distinct types of antibodies: 1) nontreponemal antibodies directed against lipoidal antigens released from damaged host cells and possibly from the treponemes themselves and 2) treponemal antibodies directed against T. pallidum proteins. Nontreponemal antibody tests can be nonreactive early in the course of infection and in late stages of disease, and often become nonreactive (serorevert) after treatment of early infection (1). Treponemal antibodies appear earlier than nontreponemal antibodies and usually remain detectable for life, even after successful treatment.
To reduce the time and labor required for syphilis screening, some laboratories have adopted reverse sequence screening in which sera are tested first by a treponemal EIA/CIA that permits automation for high throughput testing, followed by nontreponemal testing of reactive specimens. This reverse sequence can result in identification of discordant sera that are reactive with a treponemal test but nonreactive with a nontreponemal test. This result does not occur with the traditional algorithm because only nontreponemal-reactive sera are tested with a treponemal test. Discordant testing results could be caused by 1) previous syphilis infection, treated or untreated, with persistence of treponemal antibodies but seroreversion of nontreponemal antibodies, 2) a false-positive treponemal test result, or 3) early primary syphilis in a person who has yet to develop nontreponemal antibodies.
In 2008, a CDC report found that among 6,548 sera that were reactive with a screening EIA, 3,664 (56.0%) were nonreactive by reflex nontreponemal testing (2). Among the 3,664 discordant sera, 2,512 were tested with a TP-PA or FTA-ABS, of which 433 (17.2%) were nonreactive, suggesting false-positive EIA treponemal test results. In that analysis, results from four different laboratories using two different commercial EIAs and different testing protocols were combined without subanalyses. The report recommended a confirmatory treponemal test for discordant sera (using a test other than EIA or CIA) to identify persons who might require treatment (2).
Since the 2008 CDC report, confusion has persisted among clinicians, laboratorians, and public health practitioners regarding testing and treatment decisions and partner notification when using the reverse sequence for syphilis screening (3). Management decisions for patients with discordant sera and nonreactive confirmatory treponemal tests are especially difficult. To evaluate reverse sequence screening in populations with high and low syphilis prevalence and to evaluate the use of TP-PA and FTA-ABS tests for identifying false-positive EIA/CIA screening tests, CDC analyzed syphilis screening data from five clinical laboratories. Three sites served patient populations with low prevalence of syphilis (large managed-care organizations), and two sites served patient populations with high prevalence (including men who have sex with men and patients with human immunodeficiency virus infection).
A total of 140,176 sera screened with a treponemal EIA/CIA were included in the analyses (4--7; SM Novak-Weekley, Southern California Permanente Group Regional Reference Laboratories, personal communication, 2010). Data from sera with equivocal test results were not included as reactive tests. For each site and overall, the following percentages were calculated: 1) reactive EIA/CIAs among all sera, 2) discordant sera among those with reactive EIA/CIAs (i.e., those with negative nontreponemal test results), and 3) nonreactive confirmatory TP-PA or FTA-ABS tests among discordant sera. The same calculations were performed for the populations with low prevalence and high prevalence of syphilis.
Among the 140,176 specimens screened with an EIA/CIA, 4,834 (3.4%) had a reactive test result (Table). Among these 4,834 EIA/CIA-reactive sera, 2,743 (56.7%) were RPR-nonreactive, of which 866 (31.6%) were nonreactive by TP-PA or FTA-ABS testing, suggesting that the initial EIA/CIA result was a false-positive. The percentage of reactive EIA/CIAs was 6.3 times higher (14.5%) in the population with high prevalence of syphilis than the population with low prevalence (2.3%). The percentage with discordant results was higher in the low-prevalence population than in the high-prevalence population (60.6% versus 50.6%), but among the discordant sera, the percentage with nonreactive TP-PA or FTA-ABS tests was 2.9 times greater in the low-prevalence population than the high-prevalence population (40.8% versus 14.1%).
Reported by
JD Radolf, MD, Univ of Connecticut Health Center. G Bolan, MD, IU Park, MD, JM Chow, DrPH, California Dept of Public Health. JA Schillinger, MD, P Pathela, DrPH, S Blank, MD, New York City Dept of Health and Mental Hygiene, New York. SN Zanto, MPH, Montana Dept of Public Health and Human Svcs Laboratory Svcs Bureau. KW Hoover, MD, KA Workowski, MD, DL Cox, PhD, RC Ballard, PhD, Div of STD Prevention, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, CDC.
Editorial Note
For this report, data from five studies of reverse sequence syphilis screening were analyzed to determine whether CDC should provide additional recommendations for serologic testing and patient management when reverse screening is used. The finding that 56.7% of specimens with a reactive EIA/CIA screening test had a nonreactive nontreponemal (RPR) test corroborated the high percentage of discordance described in the 2008 CDC report (2). In addition, an even higher overall percentage of nonreactive confirmatory treponemal tests (31.6%) was found in this analysis, compared with the earlier report (17.2%). That the percentage of patients with nonreactive screening treponemal tests in the low-prevalence population was 2.9 times that of the high-prevalence population suggests that these EIA/CIAs were false-positive results.
CDC continues to recommend that nontreponemal tests be used to screen for syphilis and that treponemal testing be used to confirm syphilis as the cause of nontreponemal reactivity. The traditional algorithm performs well in identifying persons with active infection who require further evaluation and treatment, while minimizing false-positive results in low prevalence populations (8).
However, if reverse sequence screening is used, CDC recommends that a specimen with reactive EIA/CIA results be tested reflexively with a quantitative nontreponemal test (e.g., RPR or VDRL) (Figure). If test results are discordant, the specimen should be tested reflexively using the TP-PA test as a confirmatory treponemal test. Results from all serologic testing should be reported promptly and concurrently to the clinician and public health department. Patients with discordant serologic results by EIA/CIA and RPR/VDRL testing whose sera are reactive by TP-PA testing are considered to have past or present syphilis; if sera is TP-PA nonreactive, syphilis is unlikely (Figure).
Traditionally, the FTA-ABS test has been considered the gold standard treponemal test and still is used by some laboratories. However, the FTA-ABS test has lower specificity than other treponemal tests and probably lower sensitivity (9). In addition to inherent subjectivity, the FTA-ABS test also requires trained personnel and a dedicated fluorescence microscope. For these reasons, CDC recommends that the FTA-ABS test not be used to confirm discordant treponemal screening results. Based on published sensitivity and specificity data, the TP-PA test currently is considered to be the most suitable confirmatory treponemal test (10).
When making management decisions, clinicians always should consider data other than the results of serodiagnostic tests. An assessment is needed of the patient's sexual risk factors and medical history, especially history of previous treatment for syphilis. A physical examination also should be performed to assess for evidence of syphilis, especially primary disease (e.g., ulcerative genital or anal lesions). If the traditional algorithm is used and the initial nontreponemal test is nonreactive, patients with suspected primary syphilis should be treated and then retested with a nontreponemal test in several weeks. Previously untreated patients with discordant sera and a reactive TP-PA should be treated according to CDC's 2010 STD Treatment Guidelines.*
The findings in this report are subject to at least one limitation. The specimens were tested in five different laboratories and were not assessed using the same screening immunoassay or the same confirmatory treponemal test. CDC plans to conduct studies to compare head-to-head the performances of EIAs, CIAs, the TP-PA test, the FTA-ABS test, and a new treponemal test that utilizes an alternative format (i.e., microbead immunoassay), using specimens from well-defined patient populations whose clinical histories and syphilis risk are known.
Additional studies are expected to provide a better understanding of serologic syphilis testing practices. Studies are planned by CDC to characterize discordant sera with nonreactive confirmatory treponemal tests by immunoblotting to define their reactivities with T. pallidum antigens and identify the causes of unconfirmed reactive treponemal tests. Comparative studies are of particular importance in populations with a low prevalence of syphilis, including pregnant women who are screened as part of routine prenatal care, to guide clinical management of women and infants born to mothers with discordant sera. Additionally, based on the finding that immunoglobulin M (IgM) antibodies are produced within 2 weeks of infection (3), the utility of IgM treponemal testing for diagnosis of primary syphilis and evaluation of infection in asymptomatic, seropositive, untreated persons should be investigated.
References
-
Romanowski B, Sutherland R, Fick GH, Mooney D, Love EJ. Serologic response to treatment of infectious syphilis. Ann Intern Med 1991;114:1005--9.
-
Sena AC, White BL, Sparling PF. Novel Treponema pallidum serologic tests: a paradigm shift in syphilis screening for the 21st century. Clin Infect Dis 2010;51:700--8.
-
Borenstein LA, Spotkov JM, Cox DL, Novak-Weekley SM. High throughput laboratory experience using the Trep-Chek EIA as a screening test for syphilis [C-097]. Presented at the American Society for Microbiology 106th General Meeting. Orlando, FL; May 21--25, 2006.
-
Park IU, Schapiro JM, Chow JM, Stanley M, Shieh J, Bolan G. Treponemal immunoassays for syphilis testing: how should we manage patients with discrepant serology [P2.110]? Presented at the International Society for STD Research. London, England; June 30, 2009.
-
Philips-Rodriguez D, Perlman D, Schillinger J. Past and current syphilis diagnoses among Treponema pallidum EIA+/RPR- patients with a high rate of HIV infection; findings from medical record review, New York City, 2008--2009 [LBe]. Presented at the 2010 National STD Prevention Conference. Atlanta, GA; March 10, 2010.
-
Pohl D, Hotton A, Gratzer B, et al. Discordant syphilis EIA test results: are newer tests better [D4a]? Presented at the 2010 National STD Prevention Conference. Atlanta, GA; March 11, 2010.
-
Pope V. Use of treponemal tests to screen for syphilis. Infect Med 2004;8:399--404.
-
Marangoni A, Sambri V, Storni E, D'Antuono A, Negosanti M, Cevenini R. Treponema pallidum surface immunofluorescence assay for serologic diagnosis of syphilis. Clin Diagn Lab Immunol 2000;7:417--21.
-
Cole MJ. Comparative evaluation of 15 serological assays for the detection of syphilis infection. Eur J Clin Microbiol Infect Dis 2007;26:705--13.
* Available at http://www.cdc.gov/std/treatment/2010.
What is already known on this topic?
Reverse sequence syphilis screening identifies a large proportion of patients with reactive treponemal enzyme or chemiluminescence immunoassays (EIA/CIA) and nonreactive nontreponemal (e.g., rapid plasma reagin [RPR] or Venereal Disease Research Laboratory [VDRL]) test results, causing uncertainty about patient management.
What is added by this report?
Data from five laboratories that tested 140,176 serum specimens with reverse sequence syphilis screening indicated that, among patients with reactive EIA/CIA results, 56.7% had nonreactive nontreponemal test results and among these discordant sera, 12.2%--60.0% were nonreactive with a second treponemal test, suggesting they were false-positive results.
What are the implications for public health practice?
CDC continues to recommend traditional screening using a nontreponemal test followed by testing of reactive sera with a treponemal test. When reverse sequence screening is used, CDC recommends reflexively testing all sera that produce reactive EIA/CIA results with a quantitative nontreponemal test and reflexively testing sera with discordant results (i.e., reactive EIA/CIA and nonreactive RPR/VDRL test) with a confirmatory Treponema pallidum particle agglutination assay (TP-PA); all test results should be reported promptly and concurrently to the clinician and public health department.
FIGURE. CDC-recommended algorithm for reverse sequence syphilis screening (treponemal test screening followed by nontreponemal test confirmation)*
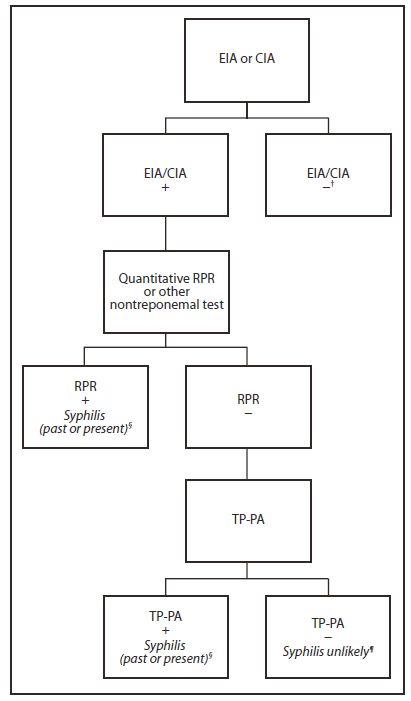
Abbreviations: EIA/CIA = enzyme immunoassay/chemiluminescence immunoassay; RPR = rapid plasma reagin; TP-PA = Treponema pallidum particle agglutination.
* Despite these recommendations for reverse sequence screening, CDC continues to recommend the traditional algorithm with reactive nontreponemal tests confirmed by treponemal testing.
† If incubating or primary syphilis is suspected, treat with benzathine penicillin G 2.4 million units intramuscularly in a single dose.
§ Evaluate clinically, determine whether treated for syphilis in the past, assess risk for infection, and administer therapy according to CDC's 2010 STD Treatment Guidelines (available at http://www.cdc.gov/std/treatment/2010).
¶ If at risk for syphilis, repeat RPR in several weeks.
Alternate Text: The figure above shows the recommended algorithm for reverse sequence syphilis screening (treponemal test screening followed by nontreponemal test confirmation). CDC recommends that a specimen with reactive EIA/CIA results be tested reflexively with a quantitative nontreponemal test (e.g., RPR or VDRL). If test results are discordant, the specimen should be tested reflexively using the TP-PA test as a confirmatory treponemal test.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


