FIGURE 1. Estimated coverage with 3 doses of diphtheria-tetanus-pertussis vaccine among children during the first year of life, worldwide and by World Health Organization region, 1980--2009
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Global Routine Vaccination Coverage, 2009
The widespread use of vaccines has greatly improved global public health, preventing millions of childhood hospitalizations and deaths each year. Vaccination of children also is projected to avert adult deaths through the prevention of hepatitis B (HepB) virus--related chronic liver disease and liver cancer (1) and human papilloma virus--related cervical cancer (2). When the World Health Organization (WHO) began the Expanded Programme on Immunization in 1974, <5% of the world's children had been fully vaccinated with bacille Calmette-Guérin (BCG), diphtheria-tetanus-pertussis (DTP) vaccine, oral poliovirus vaccine, and measles-containing vaccine (MCV) during the first year of life (3). Since then, increased vaccination coverage has resulted in substantial reductions in morbidity and mortality, including a >99% decline in polio incidence since 1988 (4), with eradication on the horizon, and a 78% decline in measles-associated mortality from 2000 to 2008 (5). With the introduction of Haemophilus influenzae type b (Hib) vaccine, HepB vaccine, pneumococcal conjugate vaccine (PCV), and rotavirus vaccine into many countries' routine vaccination schedules, further reductions in morbidity and mortality are expected. However, based on an annual global birth cohort of approximately 130 million, an estimated 23 million infants worldwide still do not receive the benefits of routine vaccination (i.e., 3 doses of DTP during the first year of life). The Global Immunization Vision and Strategy (GIVS), developed in 2005 by WHO and UNICEF, assists countries in strengthening immunization programs and vaccinating more persons. GIVS aims to achieve 90% national 3-dose DTP (DTP3) coverage by age 12 months in all countries, and 80% coverage in every district or equivalent administrative unit by 2010 (and to sustain these levels through 2015 [6]). This report summarizes global routine vaccination coverage during 2000--2009 and progress toward achieving GIVS goals.
Methods for Estimating Routine Vaccination Coverage
Routine vaccination coverage levels indicate recent immunization program performance and population immunity. Coverage usually is assessed based on the percentage of children who received a specified number of doses of a recommended vaccine during the first year of life. This is in contrast to mass vaccination campaigns or other supplemental vaccine activities that do not record vaccine doses administered. Vaccination coverage is estimated using a number of methods. Administrative vaccination coverage is calculated by dividing vaccine doses reported to have been administered to the target population by the total estimated target population. Aggregated administrative data are analyzed at the national level, and national coverage data are reported annually to WHO and UNICEF on the Joint Reporting Form on Immunization (JRF), a standard questionnaire that was developed in 1998 and is sent to all 193 WHO member states.* Vaccination coverage estimates are then reported on the WHO website.†
In many countries, household vaccination coverage surveys are conducted to validate administratively reported data. A representative sample of households is selected, and vaccination coverage is determined by examining the child's immunization card or by parental recall. WHO and UNICEF systematically review data from sources including government JRF reports, published and unpublished reports, coverage surveys, and consultation with local experts, to derive annual estimates of national coverage with recommended vaccines§ (7). DTP3 coverage by age 12 months is the agreed-upon indicator of immunization program performance. In addition to DTP3, coverage with the first MCV dose (MCV1) is an indicator used to monitor progress toward the fourth Millennium Development Goal of reducing mortality among children aged <5 years by two thirds (from 1990 levels) by 2015.¶
Estimated Routine Vaccination Coverage, 2009
Estimated global DTP3 coverage in the 193 WHO member states increased from 74% in 2000 to 82% in 2009, reflecting the vaccination of 107.1 million infants with 3 doses of DTP vaccine in 2009 (14.6 million more than in 2000). Changes in coverage varied by geographic region, and the overall increase mainly was attributed to improvements in vaccination coverage in the African (+16%), Eastern Mediterranean (+12%), and Western Pacific (+10%) WHO regions (Figure 1). National DTP3 coverage of ≥90% was reported by 122 (63%) countries, but only 48 (25%) reported ≥80% coverage in all districts, and only 55% of low-income countries are on track to achieve 90% coverage by 2015 (UNICEF, unpublished data, 2010). During 2007--2009, 149 (77%) countries had sustained DTP3 coverage of ≥80% (Figure 1); however, coverage in 2009 was <80% in 36 (19%) countries, and six countries failed to achieve 50% DTP3 coverage. Among the 23.2 million children worldwide who did not receive 3 doses of DTP vaccine during the first year of life in 2009, 70% live in 10 countries, with approximately half in India (37%) and Nigeria (14%) (Figure 2).
From 2000 to 2009, estimated global MCV1 coverage increased from 71% to 82%, and 136 (70%) countries added a second MCV dose to their routine vaccination schedules. Three-dose coverage with HepB vaccine (HepB3) increased from 30% to 70% during this period, and 3-dose coverage with Hib vaccine (Hib3) increased from 13% to 38%. In countries where Hib vaccine had been introduced, Hib3 coverage was similar to DTP3 coverage; however, a commensurate increase in global coverage did not occur because several large countries (e.g., China, India, Indonesia, and Nigeria) had not yet introduced Hib vaccine (Figure 3).
Reported by
Dept of Immunization, Vaccines, and Biologicals, World Health Organization, Geneva, Switzerland. United Nations Children's Fund, New York, New York. Global Immunization Div, National Center for Immunization and Respiratory Diseases, CDC.
Editorial Note
In 2009, more children than ever benefitted from vaccination worldwide. However, the global increase in vaccination coverage can obscure regional and local deficits in access to health services resulting from weak health systems, poor planning and resource management, limited outreach, inadequate supervision, and ineffective use of data. A recent review of published literature found that immunization program weaknesses were the leading reasons that children did not complete the DTP3 vaccination series (CDC, unpublished data, 2010). Countries such as Nigeria and India, home to approximately half the world's children who are not fully protected with 3 doses of DTP, will need to establish efficient, effective vaccination services that reach underserved populations to achieve vaccination targets.
In 2005, WHO and UNICEF created GIVS to guide countries in expanding the breadth and scope of vaccination and in using vaccination infrastructure to provide other essential health interventions (e.g., vitamin A, antihelminthics, and insecticide-treated bed nets) (6). In 2002, WHO, UNICEF, and other partners developed the Reaching Every District (RED) approach (8) to remove common obstacles to vaccination and build district-level capacity. Components of the RED approach include effective planning and management of resources, outreach to underserved communities, providing supportive supervision and training, linking health services with communities, and promoting the use of district-level data for decision making. By 2009, 164 (85%) countries reported implementing at least one RED component, and 111 (58%) reported implementing at least four components. Periodic Intensification of Routine Immunization (PIRI)** activities, such as Child Health Days and Immunization Weeks, are being used increasingly to supplement routine vaccination services, raise coverage, and reduce the number of children who do not complete the vaccination series. In 2009, a total of 109 (56%) countries reported using at least one supplemental vaccination delivery strategy.
Administrative data, although timely, are imperfect. Underreporting of vaccine doses will result in underestimation of coverage; alternatively, if children outside the target age group are vaccinated and included in the numerator, or if outdated census data that underestimate the target population are used in the denominator, coverage might be overestimated. As an alternative to administrative data, surveys can estimate coverage even if the size of the target population is not known, but they are retrospective and cannot provide timely information to immunization program managers. Survey data also might be adversely affected if interviewer training and supervision are inconsistent or if caretaker recall is unreliable (9).
Despite the extraordinary progress made toward reducing vaccine-preventable diseases during the past decade, the immunization agenda is unfinished. Increasing and expanding access to new and underutilized vaccines (e.g., PCV, Hib vaccine, and rotavirus vaccine) has the potential to greatly reduce pneumonia and diarrhea, the two leading causes of death among children aged <5 years in the developing world. As of June 2010, Hib vaccine has been introduced in 164 countries; however, because several large countries have not yet introduced the vaccine, it is only available to 48% of the 2010 global birth cohort. Rotavirus vaccine has been introduced in 23 countries, representing 11% of the birth cohort, and PCV is available in 56 countries, representing 11% of the birth cohort.
One of the greatest challenges facing immunization programs is ensuring continued funding for vaccination of children in some of the poorest and most remote places on earth. Introducing new vaccines in low-income countries presents additional funding challenges. In 2000, the GAVI Alliance, a global health partnership representing private and public stakeholders, was established to improve vaccination services in poor countries†† and to improve coverage with new and underutilized vaccines. Since 2000, $2.2 billion has been disbursed to approximately 70 countries to support vaccination services, injection safety, new vaccine introduction, and the strengthening of health service systems. Nonetheless, if the GIVS goal for global coverage is to be met, strategies known to be effective must be prioritized and implemented in all countries.
References
- Goldstein ST, Zhou F, Hadler SC, Bell BP, Mast EE, Margolis HS. A mathematical model to estimate global hepatitis B disease burden and vaccination impact. Int J Epidemiol 2005;34:1329--39.
- Garland SM, Smith JS. Human papillomavirus vaccines: current status and future prospects. Drugs 2010;70:1079--98.
- Keja K, Chan C, Hayden G, Henderson RH. Expanded Programme on Immunization. World Health Stat Q 1988;41:59--63.
- CDC. Progress toward interruption of wild poliovirus transmission worldwide, 2009. MMWR 2010;59:545--50.
- CDC. Global measles mortality 2000--2008. MMWR 2009;58:1321--6.
- World Health Organization, UNICEF. Global Immunization Vision and Strategy 2006--2015. Geneva, Switzerland: World Health Organization; 2005. Available at http://www.who.int/vaccines-documents/docspdf05/givs_final_en.pdf. Accessed October 21, 2010.
- Burton A, Monasch R, Lautenbach B, et al. WHO and UNICEF estimates of national infant immunization coverage: methods and processes. Bull World Health Organ 2009;87:535--41.
- World Health Organization. Implementing RED approach: a guide for district health management teams. Brazzaville, Republic of Congo: World Health Organization Regional Office for Africa; 2004. Available at http://www.who.int/immunization_delivery/systems_policy/AFRO-RED-guide.pdf. Accessed October 21, 2010.
- Murray CJL, Shengelia B, Gupta N, Moussavi S, Tandon A, Thieren M. Validity of reported vaccination coverage in 45 countries. Lancet 2003;362:1022--7.
* Additional information available at http://www.who.int/entity/immunization_monitoring/routine/WHO_UNICEF_JRF_10_EN.xls.
† Additional information available at http://apps.who.int/immunization_monitoring/en/globalsummary/timeseries/tswucoveragedtp3.htm.
§ Estimates include BCG, first and third doses of DTP, third dose of polio vaccine (inactivated poliovirus vaccine or oral poliovirus vaccine), third dose of Hib vaccine, third dose of HepB vaccine, and first dose of MCV.
¶ Additional information available at http://www.un.org/millenniumgoals/childhealth.shtml.
** Vaccination activities that share some characteristics of vaccination campaigns, while promoting the goal of raising routine vaccination coverage, rather than focusing primarily on accelerated disease-control activities.
†† Additional information available at http://www.gavialliance.org/media_centre/press_releases/2000_01_31_en_press_release.php.
What is already known on this topic?
Since implementation of the Expanded Programme on Immunization in 1974, widespread use of vaccines has substantially reduced morbidity and mortality worldwide. In 2005, the World Health Organization and UNICEF developed the Global Immunization Vision and Strategy (GIVS), with the goal of protecting more persons against more diseases by increasing vaccination coverage, introducing new vaccines, and linking vaccination with provision of other health services.
What is added by this report?
During the past decade, global coverage of children with 3 doses of diphtheria-tetanus-pertussis (DTP) vaccine during the first year of life has reached 82%. However, substantial regional variations in coverage exist; approximately 23 million children (half of whom live in India and Nigeria) did not receive 3 doses of DTP vaccine during the first year of life, and many low-income countries will not reach 2010 vaccination targets.
What are the implications for public health practice?
Ensuring ongoing funding for existing vaccines, as well as the introduction of new and underutilized vaccines (e.g., Haemophilus influenzae type b, pneumococcal conjugate, and rotavirus vaccines) that have the potential to reduce childhood morbidity and mortality through prevention of many cases of pneumonia and diarrhea, will be a challenge in the next decade. To achieve GIVS goals, it is critical to strengthen immunization systems and prioritize and implement effective strategies in all countries.
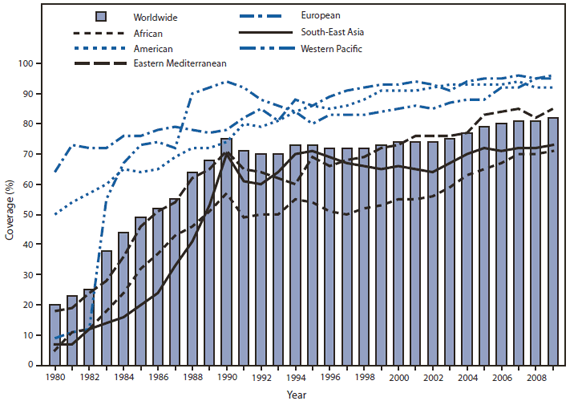
Source: World Health Organization.
Alternate Text: The figure above shows estimated coverage with 3 doses of diphtheria-tetanus-pertussis vaccine among children during the first year of life, worldwide, during 1980-2009. Estimated global DTP3 coverage in the 193 World Health Organization (WHO) member states increased from 74% in 2000 to 82% in 2009, reflecting the vaccination of 107.1 million infants with 3 doses of DTP vaccine in 2009 (14.6 million more than in 2000). Changes in coverage varied by geographic region, and the overall increase mainly was attributed to improvements in vaccination coverage in the African (+16%), Eastern Mediterranean (+12%), and Western Pacific (+10%) regions.
FIGURE 2. Estimated number (in millions) of children who had not received 3 doses of diphtheria-tetanus-pertussis vaccine during the first year of life, by country --- worldwide, 2009
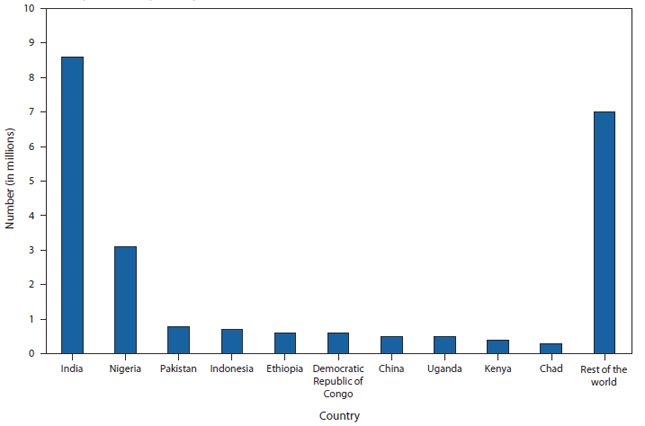
Source: World Health Organization.
Alternate Text: The figure above shows estimated number (in millions) of children who had not received 3 doses of diphtheria-tetanus-pertussis vaccine during the first year of life, by country, in 2009. Among the 23.2 million children worldwide who did not receive 3 doses of DTP vaccine during the first year of life in 2009, 70% live in 10 countries, with approximately half in India (37%) and Nigeria (14%).
FIGURE 3. Estimated vaccination coverage among children by age 12 months, by vaccine dose --- worldwide, 1980--2009
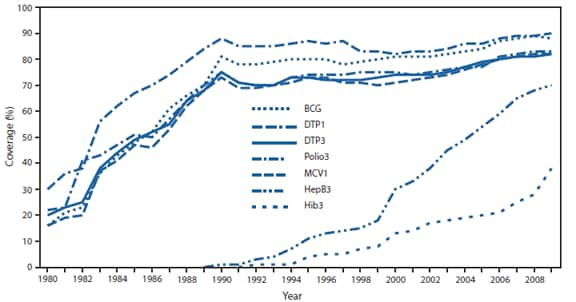
Abbreviations: BCG = bacille Calmette-Guérin; DTP1 = 1 dose of diphtheria-tetanus-pertussis vaccine; DTP3 = 3 doses of diphtheria-tetanus-pertussis vaccine; Polio3 = 3 doses of polio vaccine; MCV1 = 1 dose of measles-containing vaccine; HepB3 = 3 doses of hepatitis B vaccine; Hib3 = 3 doses of Haemophilus influenzae type b (Hib) vaccine.
Source: World Health Organization.
Alternate Text: The figure above shows estimated global vaccination coverage among children by age 12 months, by vaccine dose, during 1980-2009. From 2000 to 2009, estimated coverage with 1 dose of measles-containing vaccine (MCV) increased from 71% to 82%, and 136 (70%) countries added a second MCV dose to their routine vaccination schedules. Three-dose coverage with hepatitis B vaccine increased from 30% to 70% during this period, and 3-dose coverage with Haemophilus influenza vaccine (Hib3) increased from 13% to 38%. In countries where Hib vaccine had been introduced, Hib3 coverage was similar to DTP3 coverage; however, a commensurate increase in global coverage did not occur because several large countries (e.g., China, India, Indonesia, and Nigeria) had not yet introduced Hib vaccine.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


