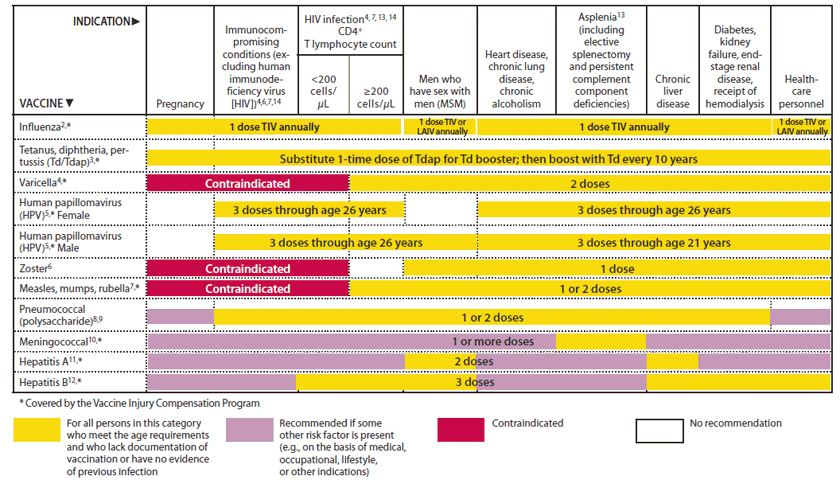
FIGURE 1. Recommended adult immunization schedule, by vaccine and age group1 — United States, 2012
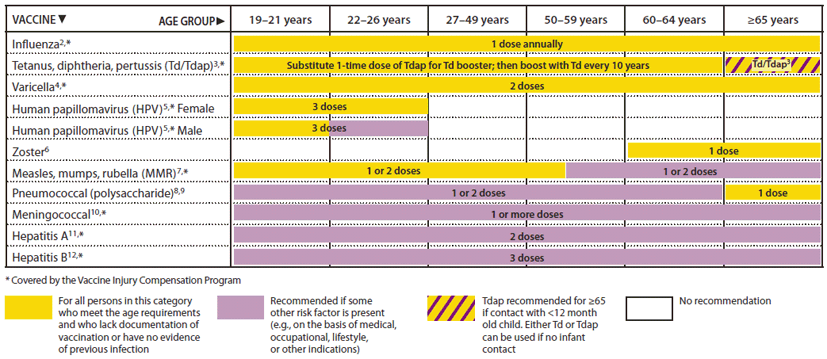
Alternate Text: The figure above shows the recommended adult immunization schedule, by vaccine and age group in the United States for 2012. For Figure 1, the bar for Tdap/Td for persons 65 years and older has been changed to a yellow and purple hashed bar to indicate that persons in this age group should receive 1 dose of Tdap if they are a close contact of an infant younger than 12 months of age. However, other persons 65 and older who are not close contacts of infants may receive either Tdap or Td. The 19-26 years age group was divided into 19-21 years and 22-26 years age groups. The HPV vaccine bar was split into separate bars for females and males. The recommendation for all males 19-21 years to receive HPV is indicated with a yellow bar, and a purple bar is used for 22-26 year old males to indicate that the vaccine is only for certain high-risk groups.
 ShareCompartir
ShareCompartir