Biosafety Resources & Tools
Through, federal and state agencies, professional societies, and the Division of Laboratory Systems’s (DLS) work with other CDC programs international organizations, DLS supports the development and adoption of standards, guidelines, recommendations, and tools for improved quality and safety in clinical and public health laboratories. The following are useful resources to establish or strengthen biosafety practices in a clinical or public health laboratory.
The Centers for Disease Control and Prevention (CDC), or the Department of Health and Human Services (HHS) cannot attest to the accuracy of a non-federal site and the listing of non-federal resources and tools below does not constitute an endorsement by HHS or any of its employees of the sponsors of the information or products.
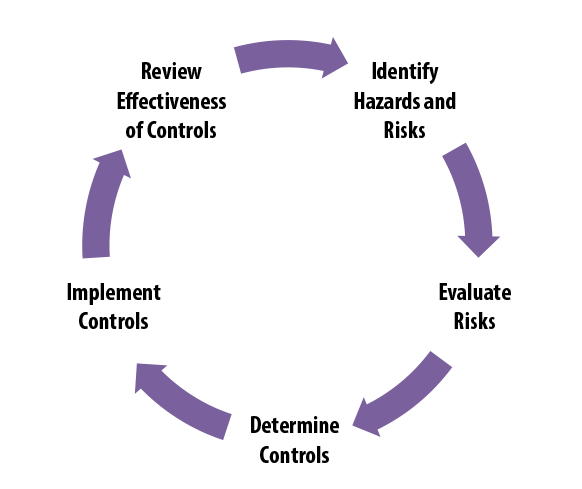
Find DLS’s new biological risk management resource.
- Biological Risk Assessment: General Considerations for Laboratories
- Biosafety in Microbiological and Biomedical Laboratories (BMBL) 6th Edition (CDC/NIH)
- World Health Organization Laboratory Biosafety Manual – Fourth Edition (WHO)
- Guidelines for Safe Work Practices in Human and Animal Medical Diagnostic Laboratories (CDC)
- CDC Prevention Guidelines Database (Archive)
- Guidelines for Biosafety Laboratory Competency (CDC/APHL)
- Laboratory Biosafety Guidance for Research with SARS-CoV-2 and IBC Requirements (NIH)
- FAQs to Lower Minimum Required Biosafety Containment Level for Research Subject to Guidelines (NIH)
Contact us at DLSinquiries@cdc.gov