Technical Report 3: Multi-National Mpox Outbreak, United States, 2022
Mpox Archive Content
You are viewing an archived web page, collected from CDC’s Mpox website. The information on this web page may be out of date.
Report reflects data and terms used as of September 23, 2022 prior to the World Health Organization’s decision to adopt the term “mpox.”
This is a technical report intended for scientific audiences. Additional information, including materials targeted to the general public, are available on the mpox site.
The purpose of this report is to provide timely updates regarding CDC’s ongoing response to the mpox outbreak in the United States, and to share preliminary results of new analyses that can improve understanding of the outbreak and inform further scientific inquiry. This report is intended for a technical audience; each report features a combination of standing topics and the results of special analyses. The first two technical reports, published on July 28, 2022, and September 1, 2022, can be found here.
U.S. Case Data
On May 17, 2022, the United States reported the first monkeypox case in the current outbreak in Massachusetts. As of September 23, there are 24,846 in all 50 states, the District of Columbia, and Puerto Rico (Figure 1). These case counts include those who tested positive for either monkeypox virus or orthopoxvirus as described in the 2022 Monkeypox Response case definition. The epidemiological curve of all monkeypox cases reported to CDC through September 23, 2022, with a 7-day rolling average (to help account for fluctuations in daily reporting), shows cases peaking in August 2022 (Figure 2). Figure 3 shows the cumulative cases per 1 million by days since 1 cumulative case per 1 million for all states. Figure 4 shows daily case counts per 100,000 population for the top 5 reporting jurisdictions. Case data are voluntarily reported to CDC by jurisdictions.
Data on age was available for 84.6% (n=21,008) of the 24,841 monkeypox cases reported to CDC’s Data Collection and Integration for Public Health Event Response (DCIPHER).1 The median age of cases is 34 years (range: <1 year to 89 years). There is one death reported that was attributed to monkeypox, and several others are being investigated. As of September 22, we are aware of 29 total confirmed pediatric cases, defined as those under 18 years of age, who presented clinically compatible disease and tested positive with a monkeypox virus specific test. Currently, all pediatric cases are subject to an in-depth interview and confirmatory molecular testing for monkeypox virus to eliminate the possibility of false positive test results that can occur in populations with low pretest probability of infection (Faisal et al. 2022). As of September 22, investigations were initiated for a total of 78 probable pediatric cases. There were 50 cases aged 13-17 years, 18 of whom were confirmed cases and 32 of whom are under investigation or pending additional test results. Of 28 cases aged 12 years or younger, 15 are under investigation or pending additional test results, and 13 had completed interviews and testing, 11 of whom were confirmed cases. There are four pregnant people and one breastfeeding person reported to have confirmed monkeypox.
Data on sex assigned at birth are available for 58.7% (n=14,593) of cases reported to CDC. Among those with available data, 14,235 (97.5%) report male sex at birth and 358 (2.5%) report female sex at birth. Gender identity data are available for 72.1% (n= 17,906) of cases reported to CDC. Among those for whom gender identity is available, 17,189 (96.0%) are men, 408 (2.3%) are women, 36 (<1%) are transgender men, 117 (<1%) are transgender women, 155 (<1%) are another gender identity, and one (<1%) is multiple gender identities.
Gender or sex data are available for 81.9% (n=20,354) of cases reported to CDC. People whose reported sex differed from their reported gender were classified as transgender. Among people for whom reported gender was not available, but sex was reported, sex was used to categorize people as female or male. While the case report form specifies sex assigned at birth, there are variations in how jurisdictions collect sex, and in some cases, this may represent current gender. Recognizing these limitations, among those with available gender or sex data, 19,554 (96.1%) are men, 453 (2.2%) women, 48 (<1%) transgender men, 143 (<1%) transgender women, and 156 (<1%) another sex or gender.
Race and ethnicity data are available for 67.8% (n=16,847) of cases reported to CDC. Figure 5 shows an epidemiological curve of all monkeypox cases reported to CDC by race and ethnicity. The Hispanic or Latino category includes people of any race, and all other categories exclude those who identify as Hispanic or Latino. Approximately 34.7% (n=5,847) of cases with known race/ethnicity are Black, 29.8% (n=5,027) are White, 29.7% (n=4,996) are Hispanic (of any race), 3.3% (n=549) are Asian, and 1.7% (n=286) are reported as other race. Less than 1% of cases with available race/ethnicity data report being American Indian or Alaska Native (n=62), or Native Hawaiian or other Pacific Islander (n=55), or multiple races (n=25).
Recent sexual history was defined as any sex and/or close intimate contact in the three weeks preceding symptom onset. Among 11,077 people (44.6% of all cases) with data on both recent sexual history and gender, 75.1% (n=8,324) reported male-to-male sexual contact (Figure 6). We note increasing proportions of cases missing data in this variable over time in conjunction with a declining proportion of cases reporting male-to-male sexual contact (discussed in more detail in the Dynamics of U.S. Outbreak section below). Some jurisdictions also report sexual orientation to the CDC. Sexual orientation was reported in previous Technical Reports using data collected during early iterations of case reporting to CDC but is no longer nationally reported and is not included in this report.
Symptom data are available for 42.8% (n=10,629) of cases reported to CDC. Among cases for whom symptom data were available, nearly all reported rash (97.9%) and a majority reported non-specific prodromal symptoms such as fever (75.2%), malaise (72.8%), chills (70.4%), pruritis (67.7%), headache (67.5%), lymphadenopathy (66.9%), myalgia (64.2%), and rectal pain (50.4%). Rash is part of the case definition and usually required for monkeypox testing.
As of September 21, 2022, a total of 104,346 specimens have been tested for non-variola orthopoxvirus or monkeypox virus in the United States, including testing at public health and select commercial laboratories. Among those specimens, some of which may have come from the same patient, 28.6% were positive.
1 DCIPHER counts are from a fixed point in time and can vary slightly from the daily case counts
Untied States Reported Monkeypox Cases per 100,000 Estimated MSM PrEP Indicated + MSM LWHIV*
Data as of September 23, 2022
*MSM = Men who have sex with men
PrEP Indicated = Indicated for HIV PrEP
LWHIV = Living with HIV
Figure 1. Monkeypox Case incidence per 100,000 estimated MSM PrEP Indicated + MSM living with HIV (LWHIV) by state or jurisdiction as of September 23, 2022.
Data from recent days, shown shaded, are incomplete
—7 Day Moving Average
Figure 2. Epidemiological curve of monkeypox cases by report date (defined as the positive laboratory test report date, CDC call center reporting date, or case data entry date into CDC’s emergency response common operating platform, DCIPHER) as of September 23, 2022. Data for some cases may be updated when additional information is provided to CDC via DCIPHER.
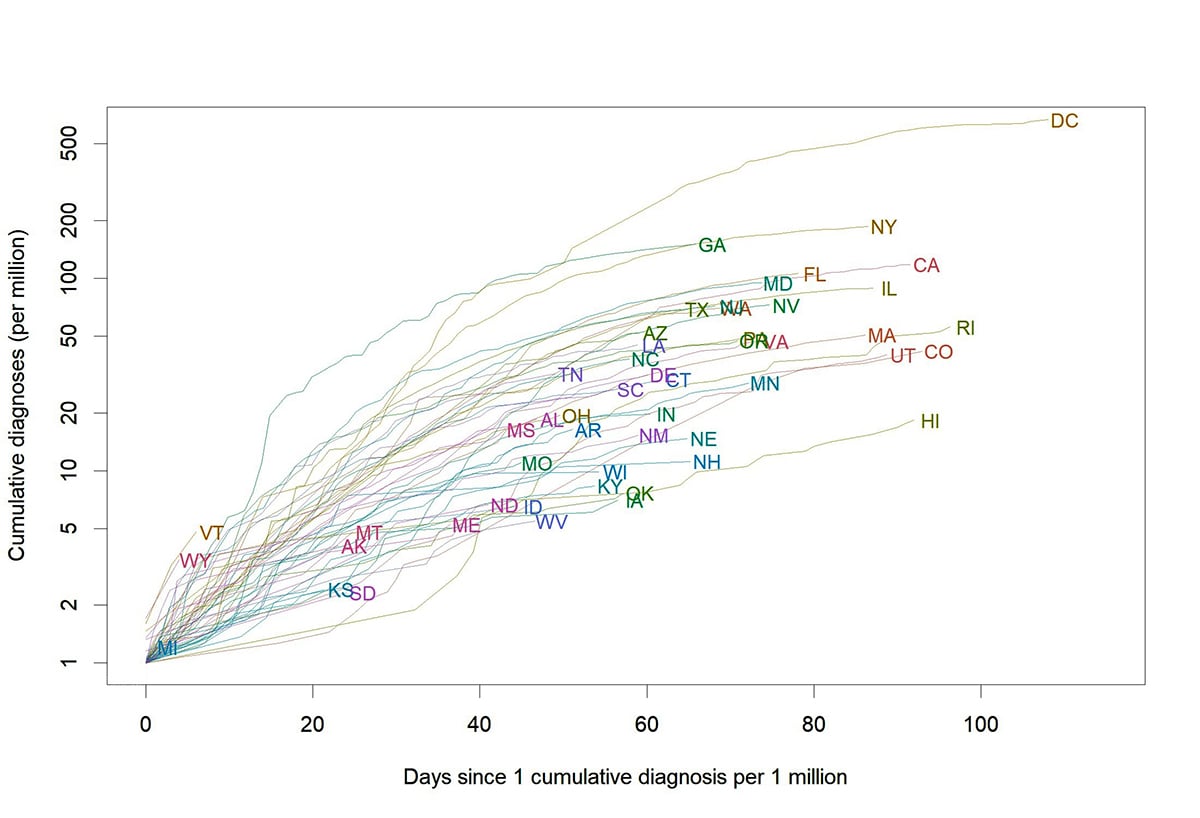
Figure 3. Cumulative monkeypox cases per million by state, by day since 1 cumulative diagnosis per million population using data as of September 23, 2022. Case data are from DCIPHER and population data from the Census.
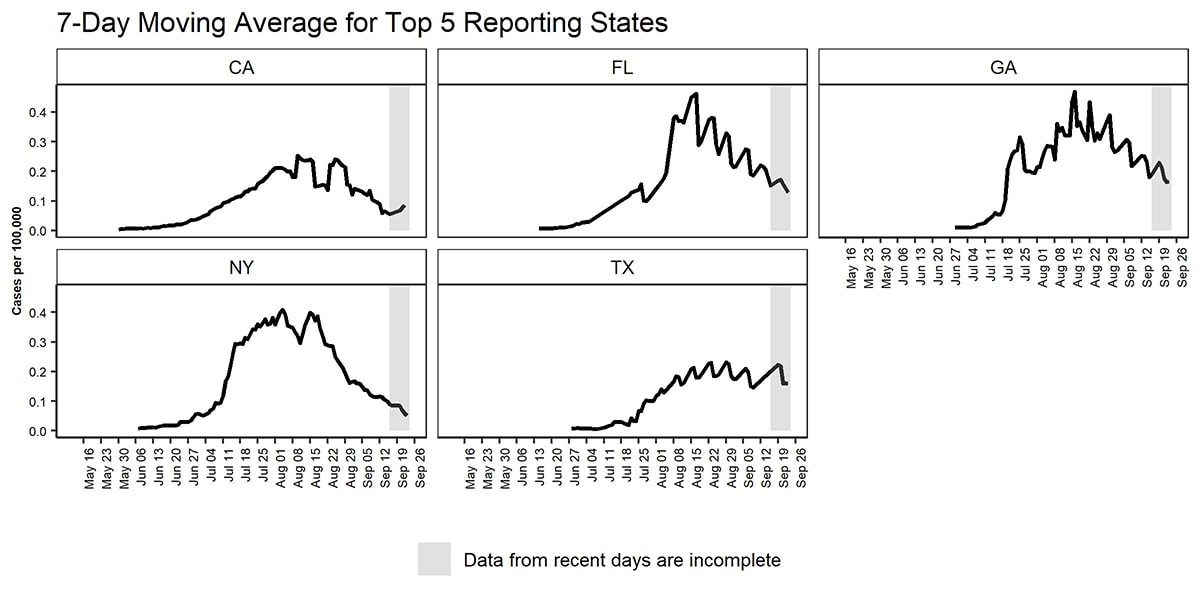
Figure 4. Epidemiological curve of 7-day moving average of monkeypox cases per 100,000 population by report date (defined as the positive laboratory test report date, CDC call center reporting date, or case data entry date into CDC’s emergency response common operating platform, DCIPHER) from top 5 jurisdictions reporting cases as of September 23, 2022 (California (CA), Florida (FL), Georgia (GA), New York (NY), Texas (TX)). Data for some cases may be updated when additional information is provided to CDC via DCIPHER.
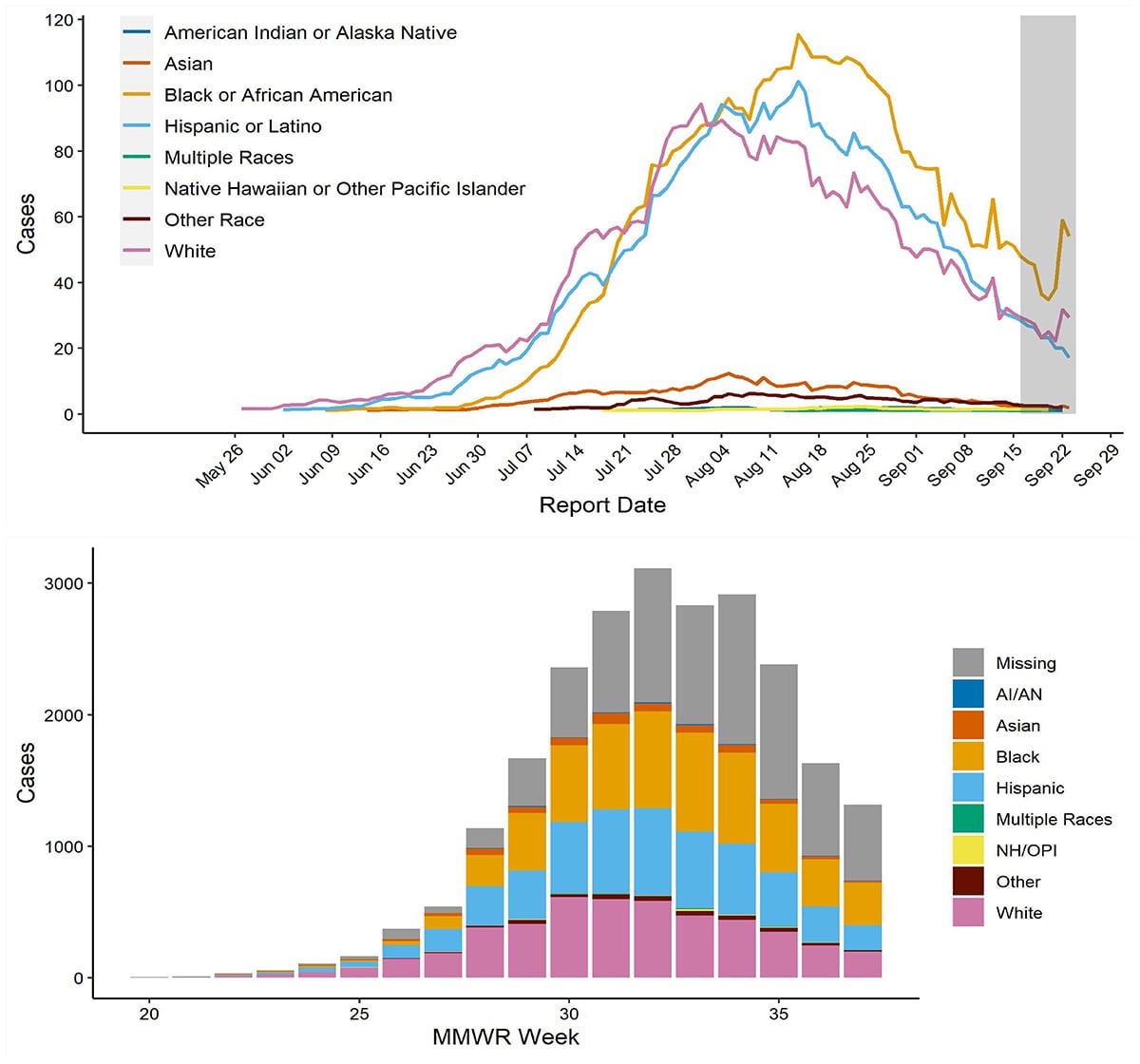
Figure 5. 7-day moving average of monkeypox cases by race/ethnicity and report date (defined as the positive laboratory test report date, CDC call center reporting date, or case data entry date into CDC’s emergency response common operating platform, DCIPHER) and counts of monkeypox cases by race/ethnicity and week as of September 23, 2022. The Hispanic or Latino category includes people of any race, and all other categories exclude those who identify as Hispanic or Latino. Data for some cases may be updated when additional information is provided to CDC via DCIPHER.
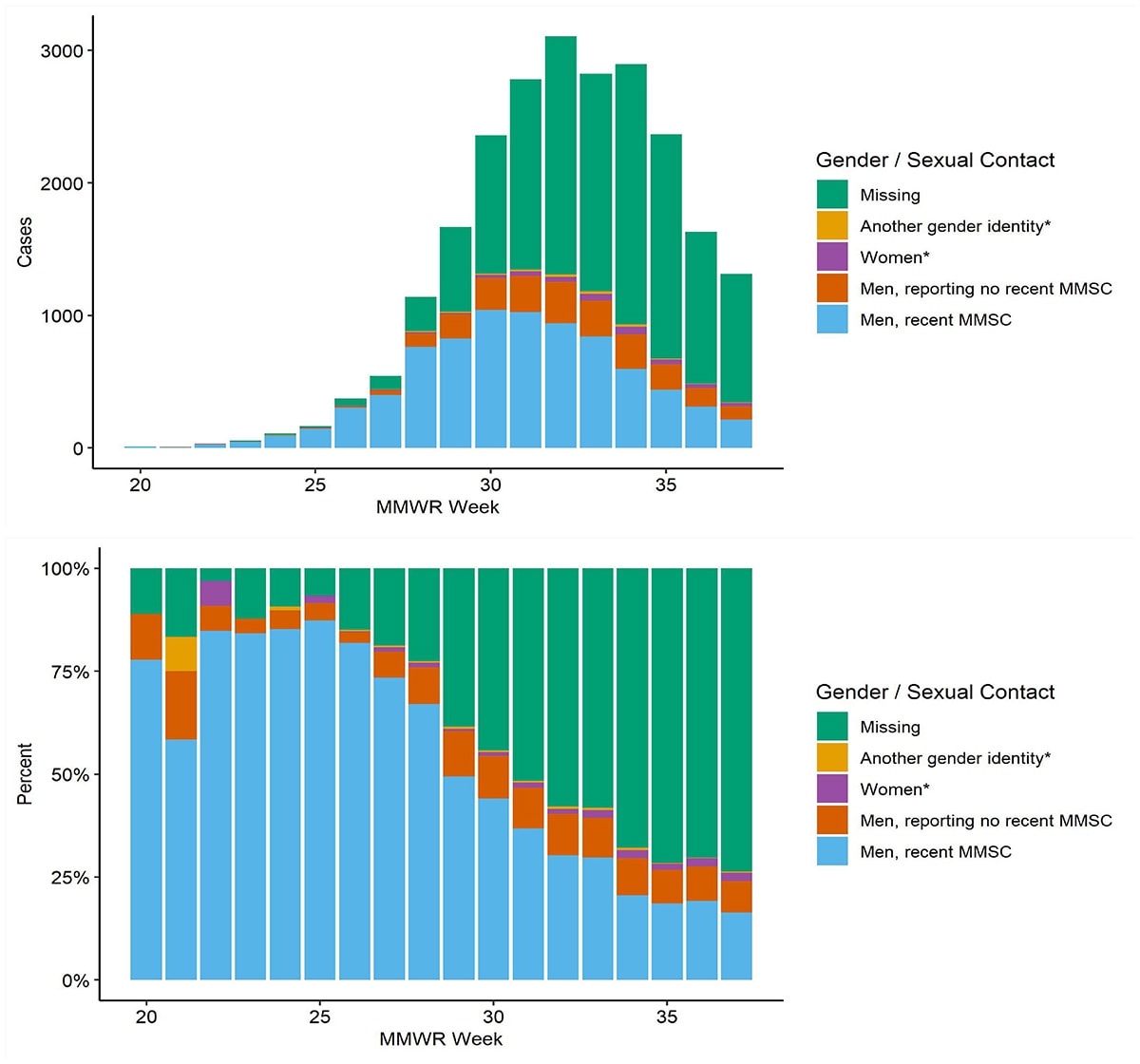
Figure 6. Counts and proportions of adults with known and unknown data on sexual history and gender reporting recent male-to-male sexual contact (MMSC) by the week in which monkeypox case was reported (reporting date defined as the positive laboratory test report date, CDC call center reporting date, or case data entry date into CDC’s emergency response common operating platform, DCIPHER) as of September 23, 2022.
- Gender or sex are available for 81.9% of adults, and sexual history is available for 45.9% of adults. Data for some cases may be updated when additional information is provided to CDC via DCIPHER.
*Due to the small number of cases in women and those with another gender identity, we did not further break these groups down by sexual history
Dynamics of U.S. Outbreak
Current Transmission Level
CDC is closely monitoring the nature of monkeypox transmission dynamics in the United States. Following levels defined by the UK Health Security Agency, the outbreak is likely to follow one of these four transmission scenarios:
1) Imported cases with limited onward transmission
2) Spread within a defined sub-population
3) Spread within multiple sub-populations or larger sub-population
4) Significant, community-wide transmission
We assess the United States is likely experiencing spread within a defined sub-population, currently among gay, bisexual, and other men who have sex with men (MSM); we have moderate confidence2 in this assessment.
We base this assessment on epidemiological data indicating that the vast majority of cases have thus far occurred in adult men and, of those for whom sexual activity is reported, among those reporting sexual contact with other men, both in the United States and globally. We have also observed few cases of transmission to household contacts and non-sexual contacts to date.
Finally, the potential for sustained spread among heterosexual networks is likely low, based on relatively lower partnership formation rates overall, extrapolating from modeling work from the United Kingdom (UK) (Brand et al. 2022; Endo et al. 2022). However, we note there are subpopulations within heterosexual networks that have high partnership formation rates.
We note the reductions in the proportion of cases reporting recent male-to-male sexual contact in the United States (Figure 6). There are at least three possible explanations for these reductions:
The proportion of cases reporting recent male-to-male sexual contact would decrease if there were changes in:
- Ascertainment of cases: If infected individuals with recent male-to-male sexual contact were less likely to be identified over time or infected individuals with other sexual contact were more likely to be identified over time.
- Reporting of sexual contacts: If cases with recent male-to-male sexual contact were less likely to have their sexual contact reported over time.
- The proportion of cases missing gender and sexual history data has increased substantially over time. Data on male-to-male sexual contact are missing for 70% of cases in the last 4 weeks included in this report. Investigations are ongoing to understand reasons behind the increasing missingness trends.
- Transmission routes: If transmission through other routes (e.g., household contacts, heterosexual networks) increased relative to the male-to-male sexual contact route.
The UK has not observed a similar shift, although their enhanced surveillance data (i.e., from additional detailed questionnaires) are only available for 30% of cases. If the dynamics of the outbreaks in the United States and the UK are following similar patterns, this finding would suggest the trends observed in the United States are likely not driven by changes in transmission routes. Given the high proportion of cases missing data on male-to-male sexual contact and that >90% of cases continue to be among males, we assess that changes in reporting of sexual contacts are likely to be an important part of the reason for the trend in the proportion of cases reporting male-to-male sexual contact. We have moderate confidence in this assessment.
2 In this report, we use the following definitions for confidence levels associated with analytic judgments: High confidence judgments are based on high-quality information from multiple sources, although such judgments are not a certainty and still carry a risk of being wrong. Moderate confidence judgments are based on credibly sourced and plausible information, but the information is not of sufficient quality or corroboration to warrant a high level of confidence. Low confidence judgments are based on information that is fragmented or poorly corroborated or upon data sources for which there are significant concerns or problems.
Outbreak Reproduction Number Estimates
Based on analysis through September 19, 2022, the growth rate of the monkeypox outbreak in the United States is slowing. We have high confidence in this assessment.
We estimate the time-varying reproduction number (Rt), which is the average number of secondary cases infected by a single primary case in a large population, has been below 1 since July 31 (Figure 7). Analyses were conducted using the EpiNow2 package, which adjusts for truncated data and reporting delays, using three parameter distributions: generation interval, incubation period, and delay from symptom onset to report date.3
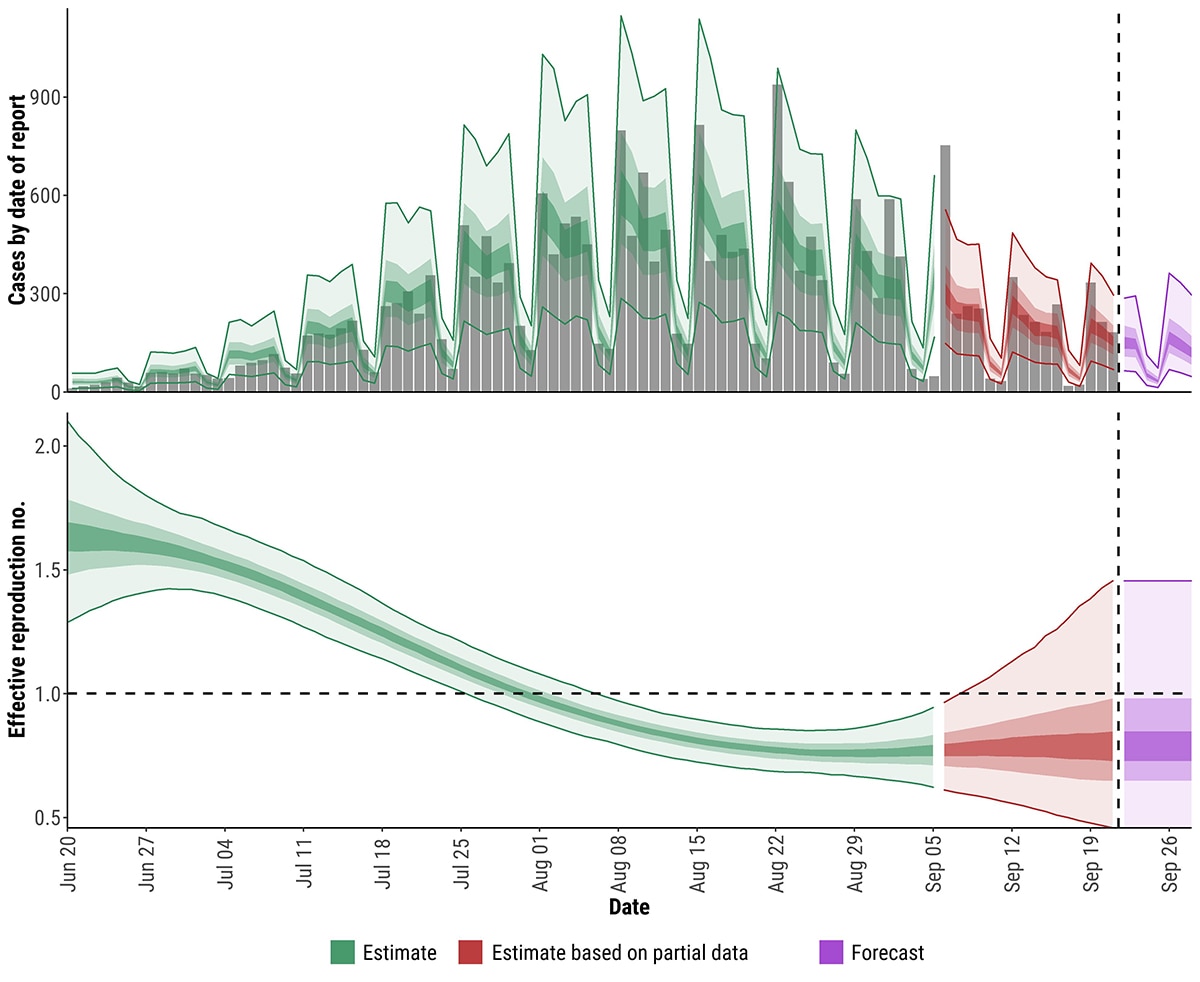
Figure 7. Top panel shows estimates of cases by date of onset, adjusted for reporting delays. Grey bars are reported cases by date of report. The bottom panel shows estimates of the effective reproductive number by date. In both panels shaded regions reflect 90%, 50%, and 20% credible intervals in order from lightest to darkest; green shows estimates, red shows estimates based on partial data, and purple shows forecasts
3 The generation interval and incubation period are internal estimates updated from Charniga K, Masters NB, Slayton RB, Gosdin L, Minhaj FS, Philpott D, et al. Estimating the incubation period of monkeypox virus during the 2022 multi-national outbreak. medRxiv. 2022
The slowing growth of the outbreak is likely due to a combination of many factors, including vaccination, behavior change, and possibly increases in infection-acquired immunity among a segment of the sexual networks at highest risk. An online survey of gay, bisexual, and other MSM conducted in early August found half of respondents reported that they had changed their behavior and reduced sexual partners and encounters due to the monkeypox outbreak (Impact of Monkeypox Outbreak on Select Behaviors | Monkeypox | Poxvirus | CDC). Therefore, continued effective health communication and protective behavior messaging, coupled with strong and equitable vaccination uptake, are necessary to sustain declines in cases. If these efforts are not sustained, there is a possibility that the declining trends could be reversed, and the incidence of new cases could increase again.
Our estimates for rates of outbreak growth suggest the reproductive number began slowing at a time when vaccine coverage was still very low. Specifically, the mean Rt estimate crossed 1 on approximately July 31; as of July 30, 103,554 first doses had been administered, representing less than 10% of the population estimated to be at high risk (i.e., estimated MSM PrEP Indicated + MSM living with HIV (LWHIV)) (Figure 8). If we assume onset of immunity following a first dose takes at least two weeks, the corresponding proportion of the population estimated to be at high risk with any vaccine-induced immunity by late July was approximately 1.3% (i.e., percent vaccinated on July 16, which is two weeks prior to Rt crossing 1). We have also examined coverage in age groups with higher coverage, such as 18–49-year-olds and note it is not substantial enough to alter this interpretation. We therefore assess it is unlikely that vaccination could fully explain the decline in the Rt estimate below one. Analyses in the UK reached similar conclusions.
However, vaccination remains an important tool as the outbreak evolves and vaccination coverage, especially of second doses, increases. As of September 20, 2022, 684,980 doses of JYNNEOS vaccines have been administered (Monkeypox Vaccine Administration in the U.S. | Monkeypox | Poxvirus | CDC). An analysis comparing monkeypox incidence in vaccinated and unvaccinated individuals is underway using surveillance data for individuals in the group recommended to be vaccinated; initial preliminary results posted on September 28 estimates a 14-fold higher monkeypox incidence in unvaccinated individuals compared to those who were vaccinated at least two weeks prior (Rates of Monkeypox Cases by Vaccination Status). Studies to further evaluate vaccine effectiveness are also being planned.
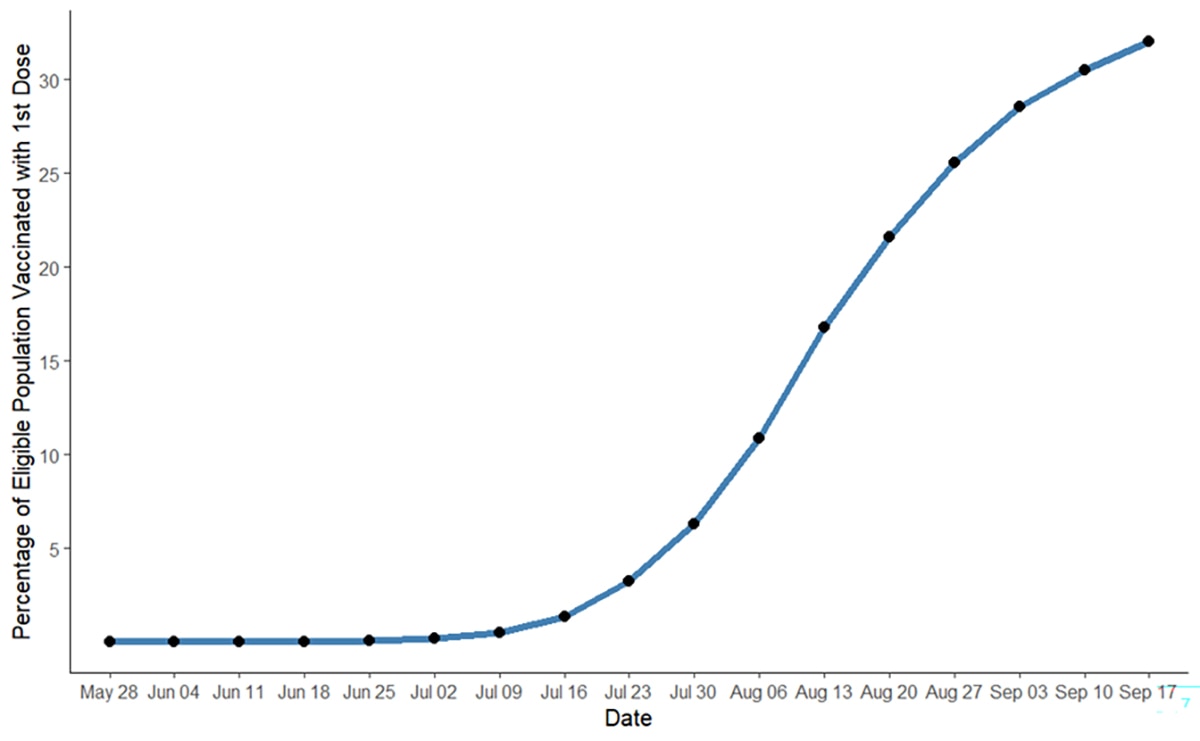
Figure 8. Cumulative percentage of the total population estimated to be at high risk (i.e., estimated MSM PrEP Indicated + MSM living with HIV (LWHIV)) who have received a first dose of vaccine, by week. Note that under the PEP++ strategy, the eligible population for vaccine has been those that had a known or presumed exposure and does not reflect the entire population at high risk.
Potential Future Outbreak Trajectory
Short-Term Scenarios
Possible outbreak growth scenarios for the United States over the next two to four weeks are the following:
1) Daily cases will continue to decline or plateau
2) Very slow growth with daily cases rising slowly
3) Exponential growth with daily cases rising rapidly
Among these scenarios, we assess daily cases in the United States will most likely continue to decline or plateau over the next two to four weeks. We have moderate confidence in this assessment but note the possibility, as described above, that incidence could increase again.
Our rationale for this assessment is based on the estimates of Rt below 1. However, we note cases are not declining in all jurisdictions. Because the causes of cases slowing in the United States and other countries are not well understood and patterns have not been uniform, we cannot predict the timing and precise trajectory of case declines in the United States.
Longer-Term Considerations
There is large uncertainty over the longer-term trajectory for the monkeypox outbreak in the United States, owing to a range of unknowns including the following:
- Detailed characteristics of sexual networks involving gay, bisexual, and other MSM and the impact of behavior change on outbreak trajectory among MSM
- Uptake and effectiveness of vaccines and other interventions on outbreak trajectory among MSM
- Pre-exposure and post-exposure vaccine effectiveness against infection and severe disease
- Potential for sustained transmission among non-MSM populations
- Extent and impact of asymptomatic spread of infection
- Potential impact of viral mutation on transmission dynamics
Our current assessment for the most likely longer-term scenario is that the outbreak will remain concentrated in MSM, with cases slowing over the coming weeks, and falling significantly over the next several months. We have moderate confidence in this assessment. We note that low-level transmission could continue indefinitely, and the cumulative number of cases that could occur among MSM is unknown.
We believe this scenario is most likely because the outbreak to date has remained concentrated among MSM. Monkeypox spread in other subpopulations has so far not been extensive, and there is no country in this outbreak with clear evidence of sustained transmission outside of MSM networks. While the declining proportion of cases reporting recent male-to-male sexual contact in the United States (Figure 6) and among MSM globally could signal potential for spread in other sub-populations, this finding is possibly an artifact due to missing data, as discussed above. We also make this assessment assuming monkeypox infection provides strong immunity to subsequent infection, as is the case for other orthopoxviruses, which will eventually lead to a depletion of susceptible individuals among populations disproportionately affected.
We note that mathematical models have shown considerable variability in predicted levels of cumulative incidence among MSM (Brand et al. 2022; Endo et al. 2022; van Dijck et al. 2022; personal communication and internal modeling results). The duration of the intense transmission phase for this outbreak is also highly uncertain. If spread is concentrated among a high-risk subset of MSM, and vaccination is well focused in this exposure group and strongly protective against infection, a more rapid decline and lower cumulative number of cases could occur (Badham and Stocker 2010). If, however, spread is less concentrated among a high-risk subset of MSM, or vaccination and behavior change only gradually slow spread, a cumulatively high number of MSM infections and a long outbreak tail are possible.
Given our uncertainty over how the monkeypox outbreak may unfold in the United States, we acknowledge several other less likely but possible scenarios that could transpire, some of which could require major shifts in our outbreak response posture.
Elimination: Domestic transmission in the United States is unlikely to be eliminated in the near future. While unlikely, elimination could occur if monkeypox is and remains concentrated in a high-risk subset of MSM, and vaccination efforts are focused on this exposure group and are effective in preventing infection, both factors which would cause faster declines in transmission. However, we view this scenario as unlikely due to the possibility of continued introductions and onward transmission.
Acceleration: The monkeypox outbreak could accelerate in the United States over the next several months and affect an increasingly wider segment of the United States population. This scenario is most likely to transpire if transmission occurs more readily than expected among non-MSM populations including potentially among highly connected heterosexual networks or by non-sexual routes.
Establishment in animal populations: There is a possibility that the monkeypox virus could establish itself in one or more animal populations in the United States, although this scenario would require that a suitable reservoir animal host exists, which is currently unknown. If such a reservoir host exists, this scenario could become more likely as case numbers rise. Several animal species in North America, both wild and domestic, may be susceptible to monkeypox infection and may be able to transmit the virus to other animals or species.
Global Outbreak
As of September 23, 2022, CDC is tracking 65,415 of and 26 deaths from monkeypox in 106 countries, territories, and areas associated with the 2022 multinational outbreak. This count may include confirmed cases not yet reported in World Health Organization (WHO) official counts. Some countries, such as the United States, include both confirmed monkeypox and confirmed orthopoxvirus cases.
The epidemic curve by WHO region (Figure 9) shows an evolving outbreak, which predominated in Europe initially, but shifted to the Americas during the summer.
Figure 10 displays weekly new cases per million population over time among the 12 countries with the highest cumulative cases reported, The United States, Canada, and European countries are experiencing clear declines in incidence. The other countries’ incidences have either plateaued (Brazil and Peru) or increased (Colombia and Mexico).
The 2022 Monkeypox and Orthopoxvirus Global Outbreak Map shows the geographical distribution of global monkeypox cases.
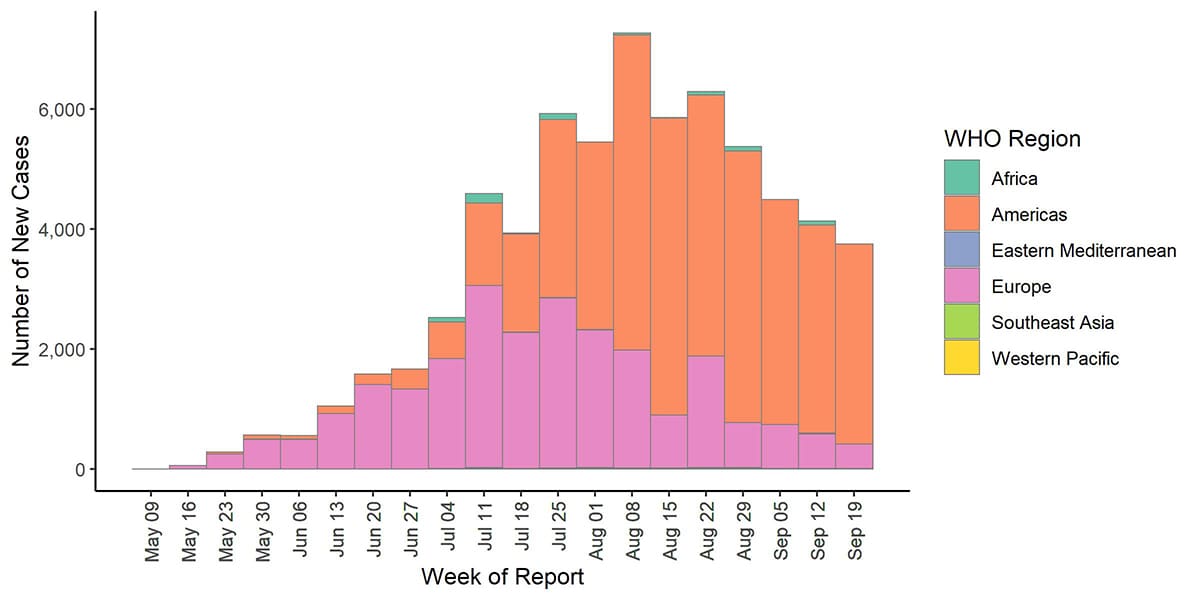
Figure 9. Weekly new monkeypox cases by WHO region globally as of September 23, 2022. CDC, WHO, European CDC, US CDC, and Ministries of Health
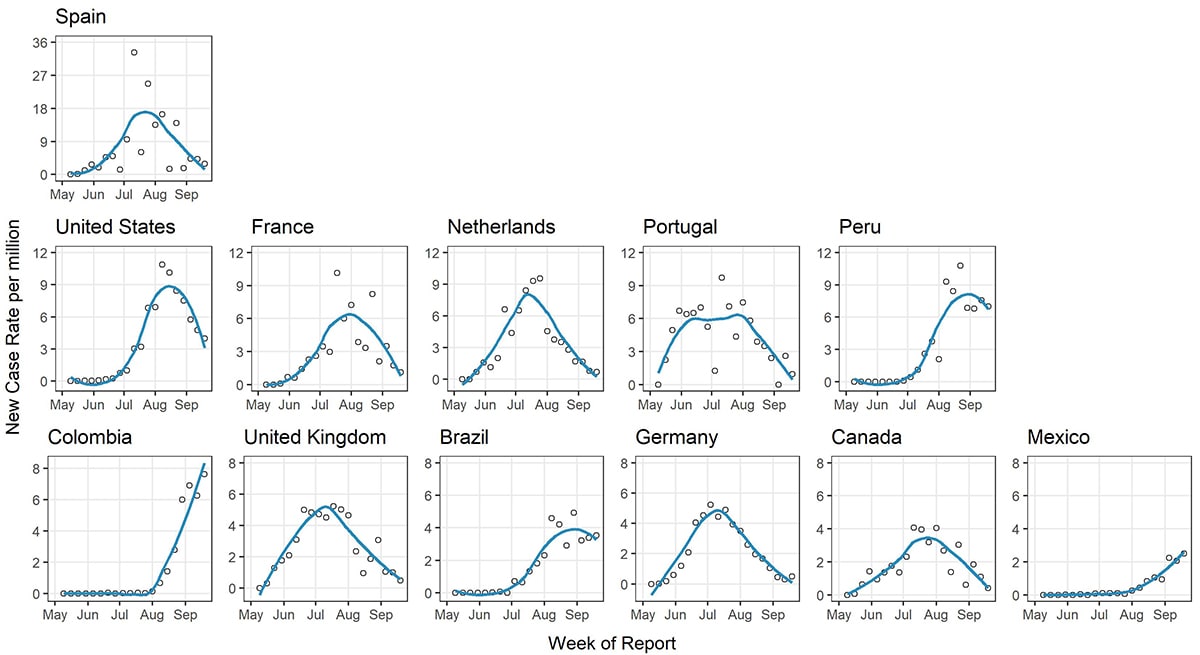
Figure 10. New weekly case counts per million population for the top 12 countries by cumulative case count as of September 23, 2022. The graphs use 3 separate y-axis scales, one for each row of graphs. Note the most recent weeks’ data might represent case undercounts, as some data might not yet bet reported. CDC, WHO, European CDC, US CDC, and Ministries of Health.
Special Projects
Monkeypox Vaccine Equity Pilot Program
On September 15, 2022, CDC announced a new opportunity for jurisdictions to submit proposals for collaborative vaccination projects intended to expand access to disproportionately affected populations experiencing vaccination disparities. Further information can be found here.
Ongoing Investigations
CDC has initiated several investigations to answer key unknowns related to monkeypox, including seroprevalence studies, clinical studies, animal studies, and lab studies. Some examples are highlighted below.
Serosurvey
Investigation: Three-week prospective study with certain public health clinics in San Francisco that predominantly care for MSM. The study involves collection of serum samples and answers to specific questions about exposures and behaviors among a racially and ethnically diverse population with a broad range of socio-economic status. The goal is to determine the prevalence of monkeypox.
Collaborating Institutions: CDC, San Francisco Department of Public Health
Status: Serum collection and serologic testing is complete. The laboratory tested 233 serum samples and found 8 had significant IgG positive results, with 3 of those 8 IgM positive. Four of the IgG positive results (2 IgM positive) have been linked with survey responses, and 4 additional IgG positive results (1 IgM positive) are currently undergoing linkage with survey responses for interpretation. One individual was likely IgG positive due to childhood smallpox vaccination. A second had no history of rash like disease; this individual was IgM negative but also less than age 50 and had no reported smallpox vaccination history. The source of this antibody response is uncertain. Two IgM positive results were reported in individuals who had a history of rash like disease but were not tested by PCR for monkeypox virus or orthopoxvirus; these were likely previously undiagnosed monkeypox virus infections.
Presence of monkeypox DNA in throat, rectal swabs, and blood and immune response against the JYNNEOS vaccine among persons at elevated risk of monkeypox exposure but without lesions at the time of collection/vaccination
Investigation: To determine if monkeypox DNA can be detected in throat, rectal swabs, and/or blood prior to lesion/rash presence among persons at elevated risk of monkeypox exposure, i.e., persons presenting for post-exposure prophylaxis (PEP) or PEP++ (people with certain risk factors and recent experiences that might make them more likely to have been recently exposed to monkeypox). To assess the immune response against JYNNEOS from the US population after at least one dose of vaccine.
Collaborating Institutions: CDC, DC Health, District of Columbia Department of Forensic Sciences Public Health Laboratory Division
Status: This study is ongoing. All data are preliminary and may change as additional data are obtained. Initial specimens arrived at CDC from the Day 0 visit and samples from the 2nd visit are expected to arrive over the next several weeks. A total of 529 throat swabs and 166 rectal swabs from the Day 0 visit have been tested by PCR. One throat swab and 2 rectal swabs tested positive via PCR. These results are under further review, and culture results of these samples will follow. These results are also currently undergoing linkage with Day 0 survey responses for interpretation. Blood samples will also be tested via PCR once all of the swab testing has been completed. A total of 327 samples from the Day 0 timepoint have been tested by IgG ELISA, and approximately 18% have been IgG positive. Results analysis and linkage with Day 0 survey responses, including correlation of IgG results with prior vaccination, are in progress. IgM analysis of select samples will follow.
Assessing monkeypox infections in animals associated with human cases
Investigation: While little is known about monkeypox susceptibility in companion and other domestic animals, the virus has a broad host range and can infect a wide variety of mammal species, including non-human primates, anteaters, hedgehogs, and prairie dogs. CDC is working with partners in states and other jurisdictions to collect samples from animals that are present in the home while their owners are isolating due to monkeypox. A standard questionnaire is administered to examine risk factors for human-to-animal transmission and to assess if behavioral modifications following monkeypox diagnosis are associated with reduced likelihood of animal infection in the household setting. The findings will help create evidence-based guidance for public health and animal health professionals.
Collaborating Institutions: CDC, DC Health, Virginia Department of Health, Minnesota Department of Health, Tennessee Department of Health
Status: Sample collection is ongoing, to date 20 animals have been sampled, including 17 dogs, 2 cats, and 1 rabbit.
Priority Research Topics
The following is a list of topics that CDC is interested in studying in more depth if there are sufficient resources to do so. For some of these topics, CDC is planning or has begun on some aspect of the topic, including for particular geographic areas or time periods, however the broader topics remain areas for future investigation.
Gaps and Potential Areas of Focus:
- Medical Countermeasure Effectiveness
- Effectiveness of vaccine to prevent or ameliorate disease when administered as pre- or post-exposure prophylaxis
- Decentralized trials, remote case monitoring, and improved access to interventions
- Comparing performance and safety of 1 vs. 2 dose vaccine regimens
- Novel methods for evaluating medical countermeasures during an outbreak
- Epidemiology, pathogenesis, and clinical characteristics
- Modeling transmission dynamics
- Clinical presentation (particularly distribution and intensity of the rash), risk factors, and patient characteristics associated with severe outcomes
- Natural history of disease, disease pathogenesis and relationship to clinical presentation and transmission route
- One Health – transmission to animals, including pets, domestic animals, and wildlife
- Social and behavioral factors associated with transmission
- Diagnostic Tools and Surveillance
- Validation and strategic deployment of pre-existing and novel diagnostics
- Viral detection across specimen types
- Levels of undetected transmission, especially in populations and regions with less access to monkeypox testing
- Serosurveys and improved serologic assays
- Community and wastewater surveillance
- Sequencing to identify changes in circulating viruses
- Sensitivity analysis to test for anti-viral resistant strains
- Communications
- Understand knowledge, attitudes, and practices of key communities and affected populations.
- Monitor mis/disinformation and implement actions to address
- Health Equity and Stigma Reduction during Public Health Emergency
- Equitably balancing resources for outbreak response (e.g., access and distribution of vaccines and therapeutics)
- Implement and evaluate the equitable distribution of testing, treatment, prevention measures and other resources; and evaluate measures to improve access for underserved groups
- Improve collection of racial and ethnic data in prevention and treatment services
- Social and Behavioral Science
- Gather qualitative and quantitative data directly from affected communities to guide implementation of testing, vaccination, and other prevention strategies.
Limitations of the Report
All data are preliminary and may change as additional data are obtained.
Due to the limited availability of detailed data from case reports and contact tracing, we continue to have knowledge and data gaps related to monkeypox transmission dynamics, case ascertainment, clinical characteristics, and other key features of this outbreak. CDC is working with state, local, tribal, and territorial partners, as well as clinical and laboratory partners, on obtaining improved case and contact tracing data to better understand this rapidly changing outbreak. CDC is also working with other federal partners and academic partners to improve our ability to respond to this outbreak.
Use of trade names and commercial sources is for identification only and does not imply endorsement by HHS or CDC.
References to non-CDC sites on the Internet are provided as a service to readers and do not constitute or imply endorsement of these organizations or their programs by CDC or the U.S. Department of Health and Human Services. CDC is not responsible for the content of pages found at these sites.
Acknowledgements
Authors of and contributors to this report are members of CDC’s 2022 Multi-National Monkeypox Outbreak Response. This report was co-led by the Center for Forecasting and Outbreak Analytics and the Epidemiologic Task Force.
References to non-CDC sites are provided as a service and do not constitute or imply endorsement of these organizations or their programs by CDC or the U.S. Department of Health and Human Services. CDC is not responsible for the content of pages found at these sites. URL addresses listed were current as of the date of publication.