Program Focus
There are many ways to measure the size of the harm attributed to the use of medications. Recent work at CDC has focused on the short-term, severe harm that results from medicines taken by people outside of hospital settings. It is estimated that more than 1 million individuals are seen in hospital emergency departments for adverse drug events each year in the United States. Over one-quarter of these patients need to be hospitalized for further treatment.
Adverse drug events are an important patient safety problem, but many are preventable. Patient safety and quality initiatives that focus on the medications and patient age groups most commonly involved in adverse drug events have the greatest potential for reducing the number of emergency department visits caused by adverse drug events.
- Overall,
blood thinners (anticoagulants), antibiotics, diabetes drugs, and opioid analgesics
are the most commonly implicated drug classes in ED visits for ADEs. - Among children and adolescents (19 years old or younger),
antibiotics are the leading cause of ED visits for ADEs. - Among adults 20-64 years old,
antibiotics remain a leading cause of ED visits for ADEs. - Among older adults (65 years old or older),
anticoagulants and diabetes drugs are the leading causes of ED visits for ADEs
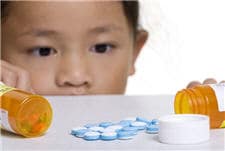
An estimated 60,000 children (18 years old or younger) are seen in emergency departments each year because of unintentional medication poisonings (excluding recreational drug use). Most of these visits (over 80%) were because an unsupervised child found and consumed the medication without adult supervision. Children less than 5 years old are twice as likely as older children to be taken to the emergency department for an adverse drug event, and one out of every 250 two-year-olds visits an emergency department for a medication poisoning annually.

Older adults (65 years or older) are three times as likely as younger patients to come to emergency departments for adverse drug events (over 177,000 emergency visits each year). Older adults are seven times more likely to be hospitalized after an emergency visit, but most of these hospitalizations are due to just a few drugs known to require careful monitoring to prevent problems. Common drugs that can require monitoring are blood thinners (e.g. warfarin), diabetes medications (e.g. insulin ), seizure medications (e.g. phenytoin ) and heart medications (e.g. digoxin).
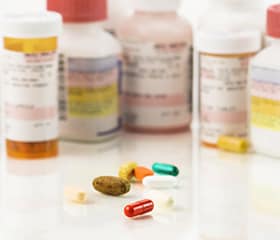
Each year in the United States, about 1 in every 250 Americans goes to a hospital emergency department because of an adverse drug event. Overall, the medications that were identified as the leading causes of emergency department visits for adverse drug events a decade ago, remained the leading causes in 2013-14 – blood thinners (anticoagulants), antibiotics, diabetes drugs, and opioid analgesics. Antibiotics are the leading cause of emergency department visits for adverse drug events in children and adolescents (19 years old or younger), while anticoagulants and diabetes drug are the leading causes for older adults (65 years old or older). Many adverse drug events are preventable. You can reduce the risk of adverse drug events by communicating with your healthcare team about all medicines that you take and by always following instructions when taking your medicines.
Learn more on the Adverse Drug Events from Specific Medicines page.
