Muscular Dystrophy Research and Tracking


The Centers for Disease Control and Prevention (CDC) funds the Muscular Dystrophy Surveillance, Tracking, and Research Network, known as MD STARnet. MD STARnet collects critical information about muscular dystrophy that aims to improve care for those living with the disease.
MD STARnet is the only research program designed to collect health information on everyone with muscular dystrophy living in specific areas of the United States. Most muscular dystrophy research programs collect health information only on people with the disease who are treated by specialists at certain medical clinics. In contrast, MD STARnet collects this information from medical clinics, as well as other sources, including hospitals and birth and death certificates.
By collecting health information on a large number of people from multiple sources, MD STARnet research can be used to help improve the care and quality of life for those living with muscular dystrophy. The more we know about the experiences of people with muscular dystrophy, the more we can learn about the course of the disease and what leads to a better quality of life.
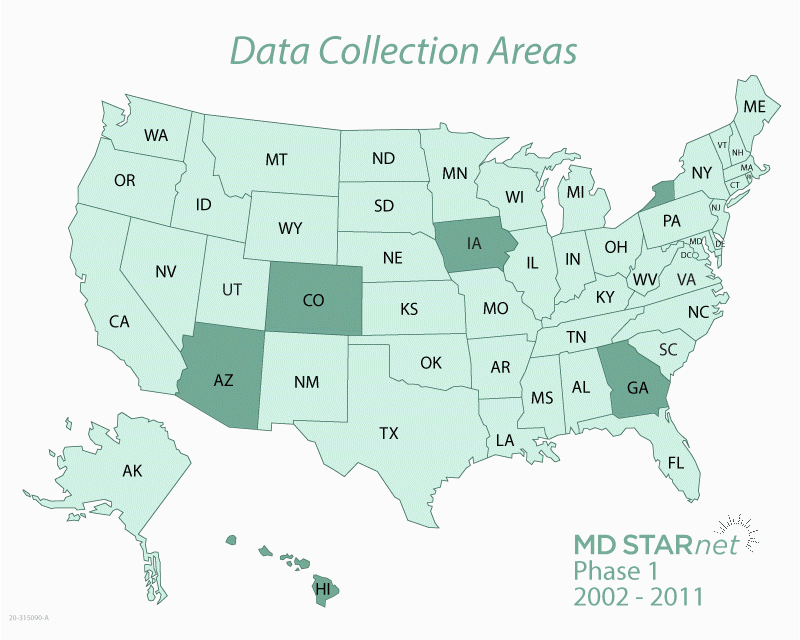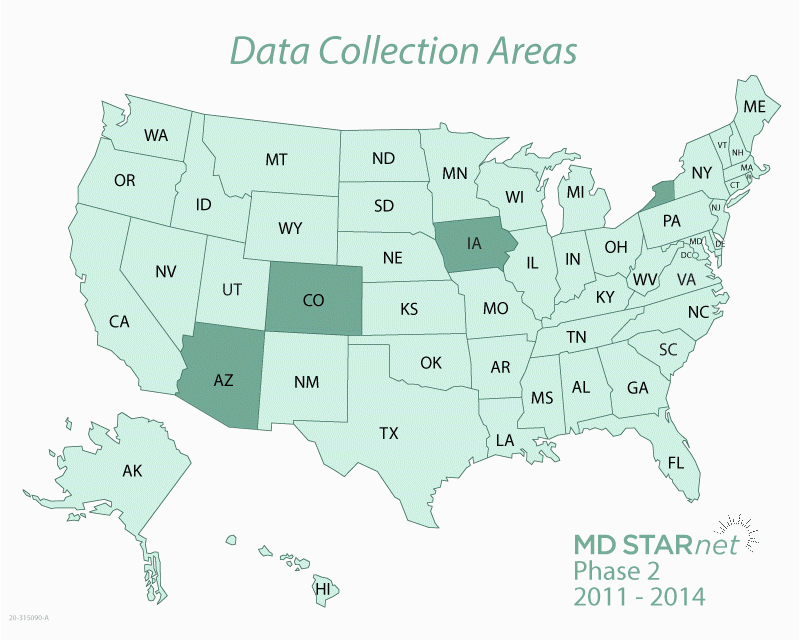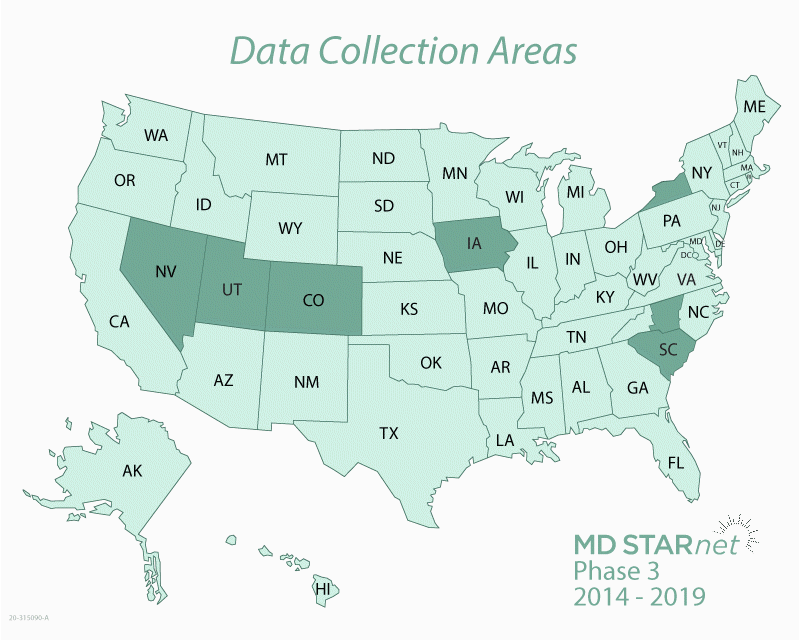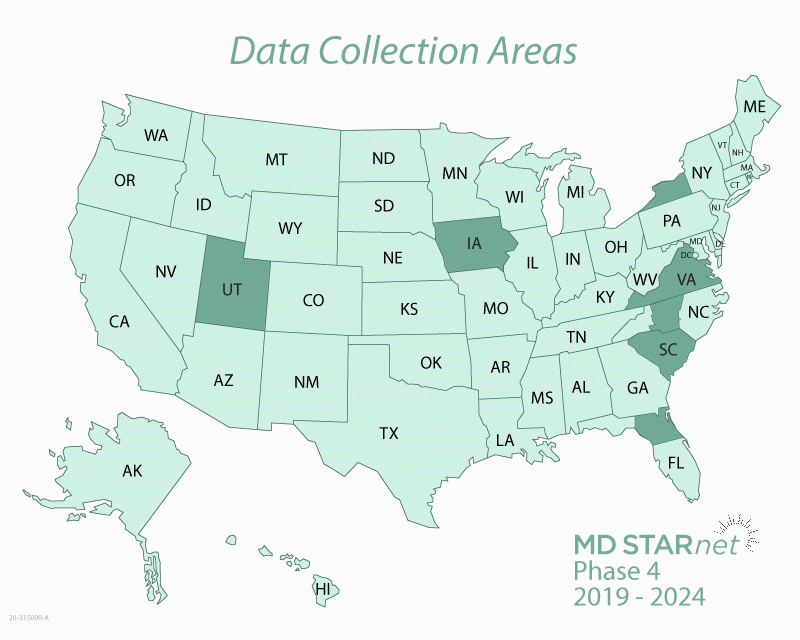
In 2019, CDC awarded funding for Phase 4 of MD STARnet research. The states or regions where health information is being collected for Phase 4 are listed below.
- University of Iowa (Iowa)
- University of Florida (23 counties in Northern Florida: Alachua, Baker, Bradford, Citrus, Clay, Columbia, Dixie, Duval, Flagler, Gilchrist, Hamilton, Hernando, Lafayette, Lake, Levy, Marion, Nassau, Putnam, St. Johns, Sumter, Suwanee, Union, Volusia)
- New York State Department of Health (21 counties in Western New York: Allegany, Cattaraugus, Cayuga, Chautauqua, Chemung, Erie, Genesee, Livingston, Monroe, Niagara, Onondaga, Ontario, Oswego, Schuyler, Seneca, Steuben, Orleans, Tompkins, Wayne, Wyoming, Yates)
- Research Triangle Institute (33 counties in North Carolina’s Piedmont region: Alamance, Anson, Cabarrus, Caswell, Chatham, Davidson, Davie, Durham, Forsyth, Franklin, Gaston, Gilford, Granville, Iredell, Lee, Lincoln, Mecklenburg, Montgomery, Moore, Orange, Person, Randolph, Richmond, Rockingham, Rowan, Stanley, Stokes, Surry, Union, Vance, Wake, Warren, Yadkin)
- South Carolina Department of Public Health & Environmental Control (South Carolina)
- University of Utah (Utah)
- Virginia Commonwealth University (Virginia)
MD STARnet Research Questions
MD STARnet recently expanded to include muscular dystrophies other than DBMD. These include myotonic, limb-girdle, facioscapulohumeral, congenital, distal, oculopharyngeal, and Emery-Dreifuss muscular dystrophies. For more information on these muscular dystrophies, visit our Facts page.
CDC and partners hope to answer the following key questions:
- How many people are affected by these types of muscular dystrophy?
- How does the health of people with muscular dystrophy change over time? Does the treatment and services they receive change over time?
- Are certain muscular dystrophies more common among specific racial and ethnic groups?
- What health services are patients and their families receiving?
- As people with muscular dystrophy become adults, what healthcare services are they receiving, how much education did they receive, and what are their opportunities for employment?
- For people with muscular dystrophies, do race, ethnicity, education, or where a person lives affect access to healthcare services?
- Is the care patients receive in line with the most recent clinical care guidance?
