COVID-19 Science Update released: January 29, 2021 Edition 74

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Section headings in the COVID-19 Science Update have been changed to align
with the CDC Science Agenda for COVID-19.
Here you can find all previous COVID-19 Science Updates.
PEER-REVIEWED
Mask-wearing and control of SARS-CoV-2 transmission in the USA: a cross-sectional study.external icon Rader et al. Lancet Digital Health (January 19, 2021).
Key findings:
- 84.6% of respondents reported they were very likely to wear a face mask to the grocery store, 40.2% reported they did so to visit friends and family, and 4.7% reported they were not likely at all to wear a mask in either setting.
- A 10% increase in self-reported mask-wearing was associated with a significantly increased odds of community transmission control (OR 3.53, 95% CI 2.03-6.43).
- Communities with both high reported mask-wearing and physical distancing had the highest predicted probability of transmission control.
- Reported mask usage continued to increase after statewide mask mandates were introduced; however, the increases were not significant.
Methods: Self-reported mask-wearing was assessed using online survey responses from 378,207 US individuals between June 3 and July 27, 2020. Multivariate logistic regression models controlling for variables such as physical distancing and population demographics estimated the association between mask-wearing and community transmission control defined as an instantaneous reproductive number (Rt) <1. Limitations: Survey relies on self-reported behavior; survey bias; ecologic study design; model assumptions.
Implications: A higher proportion of mask-wearing and physical distancing was associated with a higher probability of SARS-CoV-2 transmission control; however, mask mandates alone are not sufficient to control the ongoing pandemic and additional health interventions are needed.
Airflows inside passenger cars and implications for airborne disease transmissionexternal icon. Mathai et al. Science Advances (January 1, 2021).
Key findings:
- Air changes per hour (ACH) varied based on which windows were open in the car:
- Highest (250) when all 4 car windows were open.
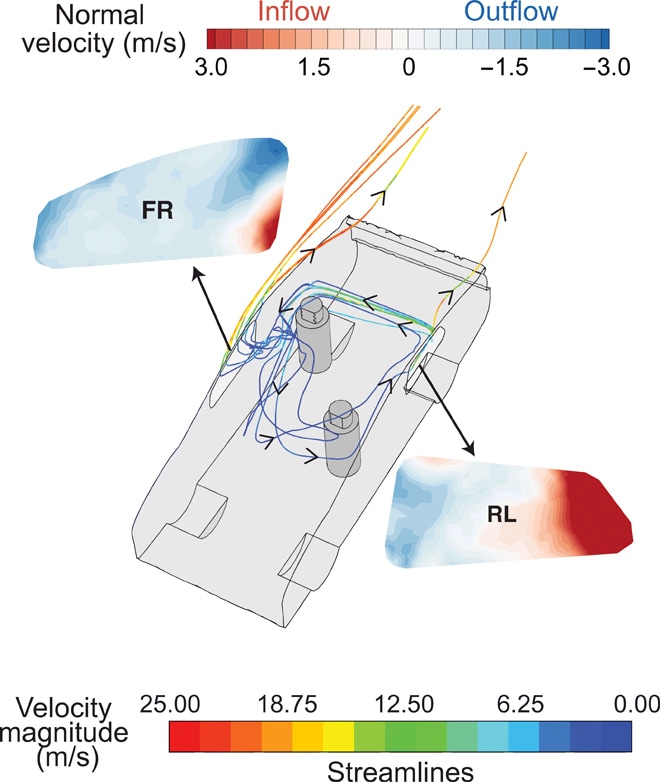
- Intermediate (approximately 150) when 3 windows were open (all 3 except the window next to the driver or the passenger respectively), and when only the 2 windows (front right and left rear) across from the 2 occupants were open (Figure).
- Relatively low (89) when only the front left and right rear windows were open.
- Lowest (62) when all were closed.
- When the rear left and front right windows were open, a strong incoming clean air stream from the rear left window acted as an “air curtain” that could reduce infectious particles reaching the passenger (Figure).
Methods: Calculated flow patterns associated with a range of ventilation options using 6 combinations of fully open and closed windows in a model 4-door car moving at 50 miles per hour, with 2 occupants—a driver in the front left seat and a passenger sitting in the rear right seat. Limitations: Variations such as slower car speeds, larger vehicles, and open moonroofs could be associated with different airflow patterns.
Figure:
Note: Adapted from Mathai et al. A typical streamline pattern in the car when the rear left and front right windows are open. The streamline color indicates the flow velocity from 0.00 to 25.00 meters/second (m/s). Insets show the opened front right (FR) and rear left (RL) windows, colored by the normal velocity, from -3.0 to 3.0 m/s. A strong air current enters the cabin from the rear left window due to the car’s motion, travels along the back seat, turns at the closed rear right window, and most of the air exits at the front right window. Licensed under CC BY-NC 4.0.
PEER-REVIEWED
The epidemiological and radiographical characteristics of asymptomatic infections with the novel coronavirus (COVID-19): A systematic review and meta-analysisexternal icon. Chen et al. International Journal of Infectious Diseases (January 12, 2021).
Key findings:
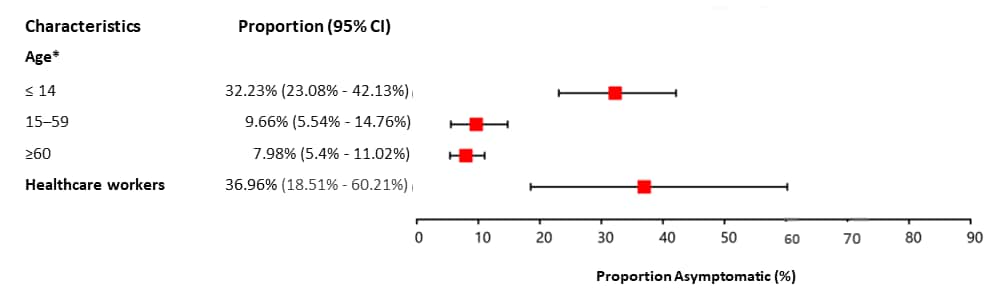
- 13% (95% CI 11%-16%) of all persons with COVID-19 were asymptomatic including:
- 32% (95% CI 23%-42%) of children and 37% (95% CI 19%-60%) of healthcare workers (Figure).
- 48% (95% CI 31%-73%) of all persons with asymptomatic infection had radiographic lung abnormalities the most common being ground-glass opacities (41%, 95% CI 20%-86%), unilateral (31%, 95% CI 22%-43%), and bilateral pneumonia (27%, 95% CI 17%-43%).
Methods: A systematic literature review, of studies published between January 1 and May 13, 2020, and meta-analysis (n = 104 studies with 20,152 cases) were conducted to summarize the epidemiological and radiographical characteristics of asymptomatic SARS-CoV-2 infections. Limitations: Heterogeneity between studies; majority of the studies conducted in one country (China).
Implications: The findings reinforce the importance of self-isolation, mask wearing, and social distancing to prevent SARS-CoV-2 transmission, particularly among children and healthcare workers.
Figure:
Note: Adapted from Chen et al. Proportion of asymptomatic SARS-CoV-2 infection by characteristic. *Statistical differences between subgroups (p<0.05). Licensed under CC BY-NC-ND 4.0.
Reductions in 2020 US life expectancy due to COVID-19 and the disproportionate impact on the Black and Latino populationsexternal icon. Andrasfay et al. Proceedings of the National Academy of Science of the United States of America (February 2, 2021).
Key findings:
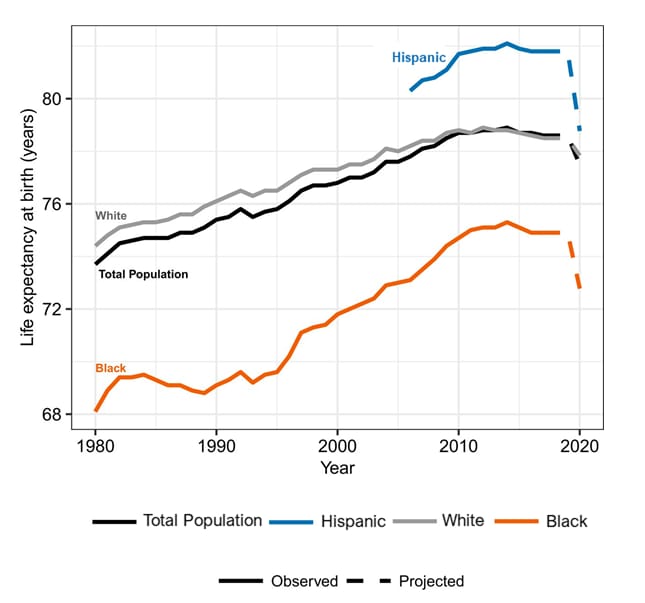
- COVID-19 is projected to reduce US life expectancy in 2020 by 1.13 years.
- Life expectancy reductions for non-Hispanic Black persons (2.10 years) and Hispanic persons (3.05 years) are projected to be 3 to 4 times higher than for non-Hispanic White persons (0.68 years) (Figure).
Methods: Life expectancy was calculated based on an Institute for Health Metrics and Evaluation (IHME) projection of 321,000 COVID-19 deaths through December 31, 2020 using 2019 population estimates. Provisional death counts due to COVID-19 by race, ethnicity, and age were obtained from the National Center for Health Statistics through October 3, 2020. Limitations: Projection assumes distributions of deaths by age and race/ethnicity through October 3, 2020 will be the same through the end of the year.
Implications: The COVID-19 pandemic has reduced life expectancy and impacted progress in reducing the gap in life expectancy between Black and White persons. A rapid return to pre-pandemic life expectancy is unlikely due to the continued presence of SARS-CoV-2 and its detrimental health impacts including prolonged effects in those who recovered from the virus, deaths from other health conditions, and social and economic losses.
Figure:
Note: From Andrasfay et al. Trends in life expectancy at birth by race and ethnicity for Total Population of the US, and for Hispanic, White, and Black persons: 1980–2020. Note that the data for Black and White persons prior to 2006 include Hispanic persons; data for these groups from 2006 onward are for the non-Hispanic Black and non-Hispanic White persons. The projections for 2020 are based on the IHME current projection scenario (October 9, 2020 update). Copyright 2021 National Academy of Sciences.
An external exposome-wide association study of COVID-19 mortality in the United States.external icon Hu et al. Science of the Total Environment (January 7, 2021).
Key findings:
- Two air toxicants, one vacant land measure, and one food environment measure were associated with COVID-19 mortality.
- NO2 increased mortality risk (adjusted mortality rate ratios [aMRR] 1.19, 95% CI 1.13–1.26) and benzidine decreased mortality risk (aMRR 0.92, 95% CI 0.88–0.95).
- Greater occupancy (aMRR 0.89, 95% CI: 0.85–0.92) and greater eligibility for reduced-price student lunches (aMRR 0.90, 95% CI: 0.87–0.93) reduced mortality risk.
Methods: Adjusted associations of 337 long-term environmental exposures with COVID-19 mortality at the county-level across the US were assessed through an exposome-wide association study. The external exposome can be defined as the totality of the external environment to which an individual is exposed across the lifespan. Natural, built, and social environment data were collected before 2020 and included air quality, walkability, food, building vacancies, crime, and industry. Limitations: Potential ecological bias, associations don’t prove causation; assumes pre-pandemic characteristics were stable during pandemic.
Implications: Environmental factors are associated with COVID-19 mortality and warrant further investigation.
PEER-REVIEWED
Household transmission of SARS-CoV-2 and risk factors for susceptibility and infectivity in Wuhan: A retrospective observational studyexternal icon. Fang et al. Lancet Infectious Diseases (January 18, 2021).
Key findings:
- The estimated secondary attack rate for household transmission was 15.6% (95% CI 15.2-16.0).
- Compared with adults ≥60 years old, individuals <20 years old were 66-84% less susceptible to infection and 60% more likely to infect others.
- Asymptomatic individuals were less infectious than symptomatic individuals (OR 0.21, 95% CI 0.14-0.31), and symptomatic individuals were more likely to infect others before symptom onset than after (OR 1.42, 95% CI 1.30-1.55).
- After the implementation of control measures (e.g., case isolation and quarantine of contacts) household reproductive numbers declined by 52% among primary cases and by 63% among secondary cases.
Methods: Retrospective cohort including 27,101 households of laboratory- or clinically-confirmed COVID-19 cases and laboratory-confirmed asymptomatic SARS-CoV-2 infections in Wuhan, China identified between December 2, 2019, and April 18, 2020. Transmission model to estimate household secondary attack rates, assuming mean incubation and maximum infectious period of 5 and 22 days, respectively. Limitations: Asymptomatic infections may be underestimated; model assumed a long (up to 22 days) infectious period, some results may not be generalizable.
Implications: While children and adolescents were less susceptible to SARS-CoV-2 infection, they were more infectious than older individuals. These findings warrant consideration when planning for school re-opening and developing interventions for reducing household transmission.
PEER-REVIEWED
Evolution of antibody immunity to SARS-CoV-2external icon. Gaebler et al. Nature (January 18, 2021).
Key findings:
- IgM and IgG anti-SARS-CoV-2 spike protein receptor-binding domain (RBD) antibodies and plasma neutralizing activity decreased significantly between 1.3 and 6.2 months after infection.
- Clones of memory B cells exhibited turnover after 6.2 months resulting in antibodies with the ability to evolve, increased potency, and resistance to RBD mutations.
Methods: Humoral memory response was characterized in 87 COVID-19-convalescent persons approximately 1.3 and 6.2 months after SARS-CoV-2 infection. All participants tested RT-PCR negative for SARS-CoV-2 at the 6-month study visit. Limitations: Small sample size.
Implications: Findings suggest that persons can mount an effective immune response that continues to evolve following recovery, reducing likelihood of re-infection by even some variant strains within 6 months after initial infection.
PREPRINTS (NOT PEER-REVIEWED)
Continued need for non-pharmaceutical interventions after COVID-19 vaccination in long-term care facilities. external iconLove et al. medRxiv (January 6, 2021).
Key findings:
- The modeled number of infections, severe infections, hospitalizations, and deaths were lowest in long term care facilities (LTCF) when vaccine deployment was paired with strong adherence to nonpharmaceutical interventions (NPI).
- Prevention of deaths was higher with strategies that prioritize vaccination of residents compared with strategies that prioritize vaccination of health care workers (HCW).
- Impact of HCW vaccination was dependent on NPI adherence more so than resident vaccination.
Methods: Model to simulate disease spread in LTCF based on vaccine influence on disease dynamics and accounting for combinations of vaccination routines and levels of NPI adherence. Limitations: Assumed theoretical vaccine efficacy.
Implications: In addition to vaccination in LTCF, continued emphasis on NPIs is essential. Given pandemic fatigue and the associated decline in NPI adherence, limiting visitors and the interactions between residents, is essential to support vaccination efforts (Kim et al. Looking beyond COVID-19 vaccine phase 3 trials. Nature Medicineexternal icon).
mRNA vaccine-elicited antibodies to SARS-CoV-2 and circulating variants.external icon Wang et al. bioRxiv (January 19, 2021).
Key findings:
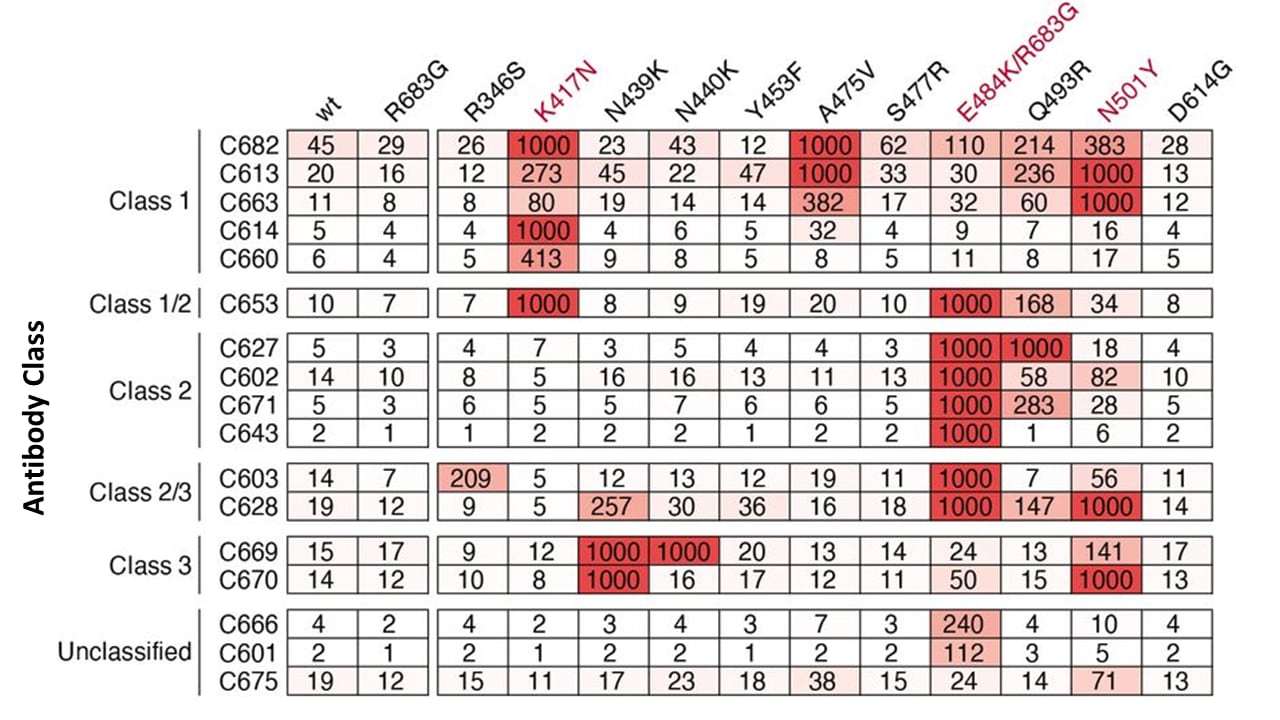
- While selected antibodies generated after receiving mRNA vaccines had some effect, they were overall less effective in neutralizing circulating variants with K417N, N501Y, and E484K mutations, in some cases displaying greater than ten-fold less activity (Figure).
- Presence of specific neutralizing monoclonal antibodies from vaccinated individuals selected for mutations within the receptor binding domain (RBD).
Methods: Sera and monoclonal antibodies from 20 adults without history of SARS-CoV-2 infection and who were vaccinated with either Moderna or Pfizer-BioNTech vaccines were assessed for neutralizing activity against SARS-CoV-2 spike protein and RBD mutants N501Y, K417N, E484K. Limitations: Small sample; not generalizable.
Implications: mRNA vaccines available in the US elicit an antibody response against SARS-CoV-2 variants but might have reduced neutralizing activity against new variants of concern with certain RBD mutations.
Figure:
Note: Adapted from Wang et al. Half-maximal inhibitory concentrations [IC50] for selected antibodies. Columns are wild-type (wt) and variant viruses tested. Rows are the monoclonal antibodies tested. Classes 1 and 2 antibodies bind within the receptor binding domain. Class 3 antibodies bind outside the receptor binding domain but are neutralizing. Increasing redness from white indicates reduced neutralizing capacity. The dark red is IC50 1000 ng/mL. Used by permission of authors.
Detection, Burden, and Impact
- Casanova et al. Epidemiology of coronavirus disease 2019 in US Immigration and customs enforcement detention facilitiesexternal icon. JAMA Network Open (January 19, 2021). Cumulative COVID-19 case rates among persons detained by Immigration and Customs Enforcement (ICE) are higher than in the general population but may be underestimated because cases are likely underreported.
- Jering et al. Clinical characteristics and outcomes of hospitalized women giving birth with and without COVID-19.external icon JAMA Internal Medicine (January 15, 2021). In a large US cohort, pregnant women with COVID-19 had higher rates of preterm birth, preeclampsia, thrombotic events, and death than women without COVID-19.
- Anesi et al. Characteristics, outcomes, and trends of patients with COVID-19–related critical illness at a learning health system in the United Statesexternal icon. Annals of Internal Medicine (January 19, 2021). In a study of 468 patients with COVID-19 requiring admission to the ICU between March and May 2020, 28-day all-cause mortality decreased over time from 43.5% to 19.2% despite stable patient characteristics.
- Moreno-Perez et al. Post-acute COVID-19 syndrome. incidence and risk factors: A Mediterranean cohort study.external icon Journal of Infection (January 11, 2021). Among a prospective cohort of 277 patients who recovered from COVID-19 between February and April 2020, 141 (50.9%; 95% CI 45.0-56.7) had post-acute COVID-19 syndrome defined as persistence of at least one clinically relevant symptom, or abnormalities in spirometry or chest radiology on an evaluation 10-14 weeks after disease onset.
Transmission of SARS-CoV-2
- Wibmer et al. SARS-CoV-2 501Y.V2 escapes neutralization by South African COVID-19 donor plasmaexternal icon. bioRxiv (January 19, 2021). The new lineage of SARS-CoV-2, 501Y.V2, that emerged in South Africa in December, contains nine spike mutations and is resistant to neutralizing antibodies found in convalescent serum from patients who recovered from wild-type infection, posing a significant risk of reinfection.
- Muik et al. Neutralization of SARS-CoV-2 lineage B.1.1.7 pseudovirus by BNT162b2 vaccine-elicited human seraexternal icon. bioRxiv (January 19, 2021). The B.1.1.7 lineage of SARS-CoV-2 that was detected in the United Kingdom in September 2020, is unlikely to escape protection by the BioNTech-Pfizer mRNA vaccine.
Natural History of SARS-CoV-2 Infection
- Gottlieb et al. Effect of bamlanivimab as monotherapy or in combination with etesevimab on viral load in patients with mild to moderate COVID-19external icon. JAMA (January 21, 2021). In this randomized phase 2/3 trial at 49 centers across the US, combination therapy with bamlanivimab and etesevimab, but not bamlanivimab alone, resulted in a significant reduction in SARS-CoV-2 viral load at day 11 among non-hospitalized patients with mild to moderate COVID illness.
- Weinreich et al. REGN-COV2, a neutralizing antibody cocktail, in outpatients with COVID-19external icon. NEJM (January 21, 2021). Randomized controlled trial of REGN-COV2, a cocktail of two monoclonal antibodies, was safe and showed reduction in viral load, especially in persons who had a high viral load at baseline or who had not yet mounted an immune response (see Editorial by Cohen et al. Monoclonal antibodies to disrupt progression of early COVID-19 infectionexternal icon. NEJM).
- Veiga et al. Effect of tocilizumab on clinical outcomes at 15 days in patients with severe or critical coronavirus disease 2019: randomised controlled trialexternal icon. BMJ (January 20, 2021). In a randomized clinical trial of 129 patients with severe COVID-19 enrolled in Brazil between May and July 2020, tocilizumab, an anti–interleukin-6 receptor monoclonal antibody, was not superior to standard care and might increase mortality; the trial was stopped early due to an increased number of deaths at 15 days in the tocilizumab group.
- Perez et al. Reported adverse drug reactions associated with the use of hydroxychloroquine and chloroquine during the COVID-19 pandemicexternal icon. Annals of Internal Medicine (January 26, 2021). Using the FDA Adverse Event Reporting Systemexternal icon database, the authors determined that adverse drug reactions (ADRs) with hydroxychloroquine and chloroquine doubled in 2020 compared with 2018 and 2019; reported ADRs seemed to coincide with positive endorsements in the media.
Prevention, Mitigation, and Intervention Strategies
- Shimabukuro et al. Allergic reactions including anaphylaxis after receipt of the first dose of Pfizer-BioNTech COVID-19 vaccine. JAMA (January 21, 2021). Based on case reports submitted to the Vaccine Adverse Event Reporting Systemexternal icon during December 14-23, 2020, anaphylaxis after the first dose of the Pfizer-BioNTech COVID-19 vaccine occurred at an estimated rate of 11.1 cases per million doses administered. The reports of anaphylaxis may lead to vaccine hesitancy but it is important to note that a history of severe allergies is not a contraindication to vaccination unless the allergy is to the vaccine or one of its components (Glover et al. Vaccinating against COVID-19 in people who report allergiesexternal icon. BMJ).
Protection in Healthcare and Non-Healthcare Work Settings
- Quilty et al. Quarantine and testing strategies in contact tracing for SARS-CoV-2: A modelling studyexternal icon. The Lancet Public Health (January 20, 2021). Modelling showed that testing can allow for reduced quarantine duration without substantial increases in transmission risk. Results support current CDC guidance.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.