Hepatitis B Vaccination of Infants, Children, and Adolescents
The Advisory Committee on Immunization Practices (ACIP) recommends:
- administration of hepatitis B vaccine and hepatitis B immune globulin (HBIG) for infants born to HBV-infected women within 12 hours of birth, followed by completion of the vaccine series and postvaccination serologic testing;
- universal hepatitis B vaccination within 24 hours of birth, followed by completion of the vaccine series; and
- vaccination of children and adolescents aged <19 years who have not been vaccinated previously.
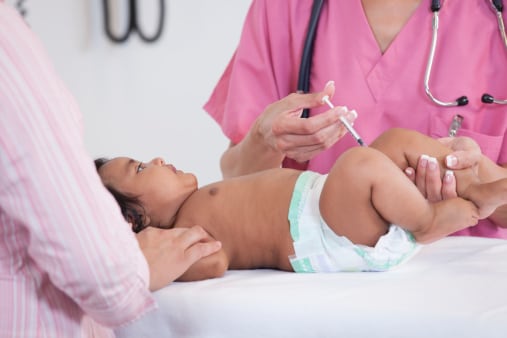
Pediatric Hepatitis B Vaccination Guidance during the Supply Shortage [PDF – 3 pages]
Immunization Schedules & Dosages
- calendar icon
- syringe solid icon
Recommended doses of Hepatitis B vaccines (by age group and vaccine type)
Standing Orders
State Vaccination Requirements
Related Pages
- syringe solid icon
- question solid icon
Hepatitis B Vaccination Questions and Answers for Health Professionals
Page last reviewed: November 8, 2019