Diagnosis
Clinicians: For 24/7 diagnostic assistance, specimen collection guidance, shipping instructions, and treatment recommendations, please contact the CDC Emergency Operations Center at 770-488-7100. More detailed guidance is under Information for Public Health & Medical Professionals.
Photos
Learn more about the appearance of Naegleria fowleri on the Photos page.

Video
Lab Detection of Naegleria fowleri, the Jordan Smelski Foundation.
Subject matter experts, including CDC expert Dr. Jennifer Cope, provide background information on Naegleria fowleri and discuss how to identify the ameba in diagnostic specimens.
Direct Visualization
Cerebrospinal Fluid
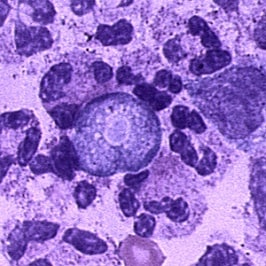
Trophozoite of Naegleria fowleri in CSF, stained with H&E.
The diagnosis of Naegleria fowleri infection can be made by microscopic examination of fresh, unfrozen, unrefrigerated cerebrospinal fluid (CSF). A wet mount of freshly centrifuged CSF sediment might demonstrate actively moving trophozoites. Naegleria fowleri (15-30 µm trophozoite) moves rapidly (~1 µm/s) using eruptive pseudopods and moves sinuously in a generally linear forward direction.
Additionally, Naegleria can be identified in CSF smears or cultures using hematoxylin and eosin (H&E), periodic acid-Schiff (PAS), trichrome, Giemsa, or Wright-Giemsa stains. A Gram stain should be avoided, as the amebae can be destroyed during heat fixation. A stained CSF smear will show ameboid trophozoites with morphology typical of Naegleria (i.e., a nucleus with a large, centrally located and densely staining nucleolus). If amebae are identified in the CSF, the diagnosis of PAM should be subsequently confirmed with polymerase chain reaction (PCR).
Tissue
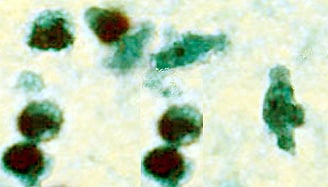
Trophozoite of Naegleria fowleri in CSF, stained with trichrome. Image courtesy of the Texas State Health Department.
The diagnosis can also be made from microscopic examination of hematoxylin and eosin, periodic acid-Schiff, trichrome, Giemsa, or Wright-Giemsa-stained smears of brain biopsy or autopsy specimens, which might demonstrate trophozoites with morphology typical of Naegleria fowleri. The ameboid trophozoites measure 10-35 µm but when rounded are usually 10-15 µm in diameter. The cytoplasm is granular and contains many vacuoles. The single nucleus is large and has a large, dense karyosome. Naegleria fowleri does not form cysts in human tissues.
For microscopy images of Naegleria fowleri, visit the Photos page.
Immunohistochemical Staining
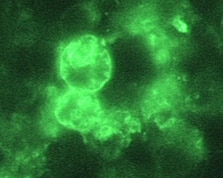
Indirect Immunofluorescence (IIF) assay for Naegleria fowleri, 1000x oil magnification
Immunohistochemical (IHC) staining and indirect immunofluorescent (IIF) staining use an antibody specific for Naegleria fowleri followed by microscopic examination to identify Naegleria fowleri in formalin-fixed tissue, or CSF.
Serology
Serologic testing for Naegleria fowleri using indirect immunofluorescent antibody (IFA) to measure serum antibody titers in patient sera has little diagnostic value, because most patients with PAM die before an immune response is mounted. However, a California PAM survivor did exhibit a serologic response.
Polymerase Chain Reaction (PCR)
Specific molecular tools can amplify DNA from the amebae in CSF or tissue to specifically identify if the amebae are present. DNA has been successfully amplified from CSF and unfixed tissue samples. Typing of isolates can be done, but little is known about the natural populations to put these data in a biological context. An increasing number of PCR-based techniques (conventional and real-time PCR) have been described for detection and identification of free-living amebic infections in clinical specimens, but are only available in selected reference diagnostic laboratories. A real-time PCR was developed at CDC for qualitative assessment of Naegleria fowleri, Acanthamoeba spp., and Balamuthia mandrillaris in clinical samples. This assay uses distinct primers and TaqMan probes for the simultaneous identification of all three free-living amebae. Strains or subtypes of Naegleria fowleri can be looked at, but little is known about the natural populations in the environment, which makes it difficult to interpret the findings. Nevertheless, subtyping can help identify the environmental exposure of Naegleria fowleri in PAM patients. Recently, next-generation sequencing has also been used in the diagnosis of a PAM case.
Culture
Culture procedure may be used to identify free-living amebae in clinical and environmental specimens. It involves inoculating mammalian cell cultures and monitoring for cytopathogenicity or growth on E. coli lawns. For growing on an E. coli lawn, the sample is added to a growth plate covered in bacteria that can serve as a food source for Naegleria fowleri. The initial screening is accomplished by incubating the plate at a higher temperature (108°F/42°C) that kills most free-living amebae, while selecting for thermophilic amebae, such as Naegleria fowleri or other amebae. This initial screen shows up as tracks made by an ameba as it moves across the plate eating the bacteria. If there are no amebae on the plate grown at the higher temperature, then Naegleria fowleri is not present. If thermophilic amebae are present on the plate grown at the higher temperature, then these amebae undergo further specific testing to determine whether Naegleria fowleri is present since other thermophilic free-living amebae could be present. (NOTE: Amebae, including thermophilic amebae other than Naegleria fowleri, can be common in water systems, but none of these other amebae cause PAM.) Naegleria can be specifically identified in cultures from clinical specimens using hematoxylin and eosin (H&E), periodic acid-Schiff (PAS), trichrome, Giemsa, or Wright-Giemsa stains. Stained cultures might demonstrate trophozoites with morphology typical of Naegleria fowleri, as previously described. In culture, trophozoites may measure more than 40 µm. A negative culture result does not rule out the presence of free-living amebae, and other tests should be performed.
Clinical Specimens for Diagnosis at CDC
If possible, CDC requests that the following specimens be sent for diagnostic testing at CDC:
- Fresh CSF
- If the patient has had a biopsy, we also request the following specimens, if available:
- Fresh brain tissue
- Formalin-fixed and paraffin embedded unstained tissue slides
- Formalin-fixed and paraffin embedded H&E-stained tissue slides
Clinicians and Laboratorians: please contact the CDC Emergency Operations Center (770-488-7100) to discuss specimen submission for suspected cases.
Submission Information
Central Nervous System (CNS) Tissue
Naegleria fowleri is most likely to be detected in biopsy or autopsy tissue collected from the area surrounding the nasal-olfactory bulbs in the brain. However, CDC requests that tissues be collected from other CNS sites in addition to the olfactory bulb to look for other possible locations of ameba entry into the brain, such as around the auditory nerve.
Extra-CNS Tissue
All possible steps should be taken to minimize the possibility of cross-tissue contamination between CNS and extra-CNS tissues. These steps should, at a minimum, include:
- Completing the gross examination and sample collection from all extra-CNS tissues prior to examination of the CNS tissues
- Utilizing separate workspaces and dissecting tools for the extra-CNS and CNS tissues
- Placing recovered samples of extra-CNS and CNS tissues in separate formalin containers
- Processing all tissues, particularly extra-CNS and CNS, separately
- Cutting extra-CNS and CNS tissues separately
- If the same equipment is used to cut the tissue, cut extra-CNS tissues first and include a cleaning step in between different tissues
CDC can accept the following specimens:
- Fresh CSF
- Fresh, unfixed brain tissue
- Fresh, unfixed tissue (other than brain)
- Formalin-fixed, paraffin-embedded, tissue
- Three H&E-stained slides
- Six unstained slides
- Paraffin-embedded tissue block
- Photos of gross brain morphology
- Particularly around olfactory and auditory areas
- Serum
Clinicians and Laboratorians: please contact the CDC Emergency Operations Center (770-488-7100) to discuss specimen submission for suspected cases.
- Visvesvara GS. Amebic meningoencephalitides and keratitis: challenges in diagnosis and treatment.Curr Opin Infect Dis. 2010 Dec;23(6):590-4.
- da Rocha-Azevedo B, Tanowitz HB, Marciano-Cabral F. Diagnosis of infections caused by pathogenic free-living amoebae. Interdiscip Perspect Infect Dis. 2009;2009:251406.
- Seidel JS, Harmatz P, Visvesvara GS, Cohen A, Edwards J, Turner J. Successful treatment of primary amebic meningoencephalitis. N Engl J Med. 1982;306:346-8.
- Visvesvara GS, Moura H, Schuster FL. Pathogenic and opportunistic free-living amoebae: Acanthamoeba, Balamuthia mandrillaris, Naegleria fowleri, and Sappinia diploidea. FEMS Immunol Med Microbiol. 2007;50:1-26.
- Qvarnstrom Y, Visvesvara GS, Sriram R, da Silva AJ. Multiplex real-time PCR assay for simultaneous detection of Acanthamoeba, Balamuthia mandrillaris, and Naegleria fowleri. [PDF – 26 pages] J Clin Microbiol. 2006;44(10):3589-95.
- Robinson BS, Monis PT, Dobson PJ. Rapid, sensitive, and discriminating identification of Naegleria by real-time PCR and melting-curve analysis. Appl Environ Microbiol. 2006;72(9):5857-63.
- Marciano-Cabral F, MacLean R, Mensah A, LaPat-Polasko L. Identification of Naegleria fowleri in domestic water sources by nested PCR. Appl Envirol Microbiol. 2003;69:5864-9.
- Wang Q, Li J, Ji J, Yang L, Chen L, Zhou R, Yang Y, Zheng H, Yuan J, Li L, Bi Y, Gao GF, Ma J, Liu Y. A case of Naegleria fowleri related primary amoebic meningoencephalitis in China diagnosed by next-generation sequencing. BMC Infect Dis. 2018 Jul 28;18(1):349.
- Visvesvara GS. Parasite Culture: Acanthamoebaand Naegleria In: Garcia LS, editor. Clinical Microbiology Procedures Handbook. 3rd ed. Washington, DC: ASM Press; 2010.
- Stockman LJ, Wright CJ, Visvesvara GS, Fields BS, Beach MJ. Prevalence of Acanthamoeba and other free-living amoebae in household water, Ohio, USA–1990-1992. Parasitol Res. 2011;108(3):621-7.
- Seal D, Stapleton F, Dart J. Possible environmental sources of Acanthamoeba in contact lens wearers. Br J Ophthalmol. 1992;76(7):424–7.
- Shoff ME, Rogerson A, Kessler K, Schatz S, Seal DV. Prevalence of Acanthamoeba and other naked amoebae in South Florida domestic water.external icon J Water Health. 2008;6(1):99–104.
- Rohr U, Weber S, Michel R, Selenka F, Wilhelm M. Comparison of free-living amoebae in hot water systems of hospitals with isolates from moist sanitary areas by identifying genera and determining temperature tolerance.