Pathogen and Environment
Naegleria fowleri is a heat-loving (thermophilic), free-living ameba (single-celled microbe), commonly found around the world in warm fresh water (like lakes, rivers, and hot springs) and soil. Naegleria fowleri is the only species of Naegleria known to infect people. Most of the time, Naegleria fowleri lives in freshwater habitats by feeding on bacteria. However, in rare instances, the ameba can infect humans by entering the nose during water-related activities. Once in the nose, the ameba travels to the brain and causes a severe brain infection called primary amebic meningoencephalitis (PAM), which is usually fatal.
The first report identifying PAM infections was published in 1965 in Australia. The report identified three fatal infections from 1965 and one from 1961. The ameba that caused the infections was identified as a new species, which was later named Naegleria fowleri after one of the authors of the original report, M. Fowler. A 1966 report was the first to identify infections in the United States, which occurred in 1962 in Florida. Later investigations using archived autopsy tissue samples identified PAM infections that had occurred in Virginia as early as 1937.
Naegleria fowleri has three stages in its life cycle: cysts (1), trophozoites (2), and flagellated forms (3). The trophozoites replicate by promitosis (nuclear membrane remains intact) (4). Naegleria fowleri is found in fresh water, soil, thermal discharges of power plants, geothermal wells, and poorly chlorinated recreational and tap water. Trophozoites can turn into temporary non-feeding flagellated forms which usually revert back to the trophozoite stage. Trophozoites infect humans or animals by penetrating the nasal mucosa, usually during swimming or sinus irrigation (5), and migrating to the brain (6) via the olfactory nerves causing PAM. Naegleria fowleri trophozoites are found in cerebrospinal fluid (CSF) and tissue, while flagellated forms are occasionally found in CSF. Cysts are not seen in brain tissue.
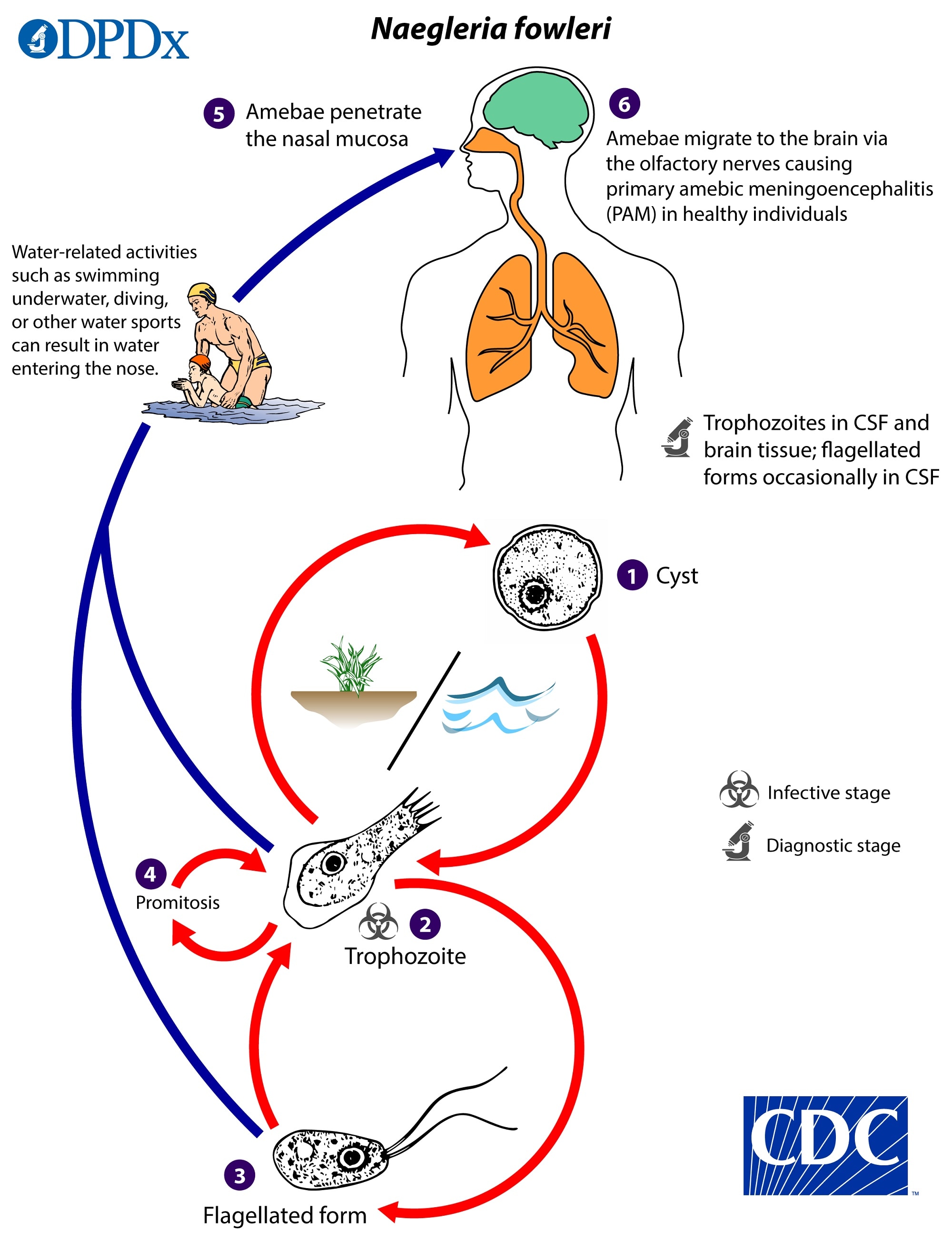
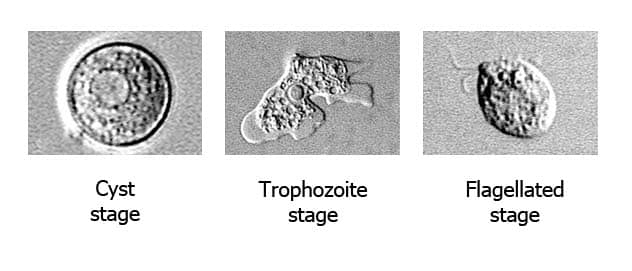
Naegleria fowleri is normally found in the natural environment and is well adapted to surviving in various habitats, particularly warm-water environments. Although the trophozoite stage is relatively sensitive to environmental changes, the cysts are more environmentally hardy.
Drying: Drying appears to make trophozoites nonviable instantaneously and cysts nonviable in less than 5 minutes.
Temperature: Naegleria fowleri is a heat-loving (thermophilic) ameba able to grow and survive at higher temperatures, such as those found in hot springs and in the human body, even under fever temperatures. Naegleria fowleri grows best at higher temperatures up to 115°F (46°C). Although the amebae may not be able to grow well, Naegleria fowleri can still survive at higher temperatures for short periods of time. The trophozoites and cysts can survive from minutes to hours at 122–149°F (50–65°C), with the cysts being more resistant at these temperatures. Although trophozoites are killed rapidly by refrigeration, cysts can survive for weeks to months at cold temperatures above freezing, although they appear to be sensitive to freezing. As a result, colder temperatures are likely to cause Naegleria fowleri to encyst in lake and river sediment, where the cyst is offered more protection from freezing water temperatures.
Disinfection: Naegleria fowleri trophozoites and the more resistant cysts are sensitive to disinfectants like chlorine and monochloramine. Chlorine is the most common disinfectant used to treat drinking water and swimming pools. The chlorine sensitivity of Naegleria fowleri is moderate and in the same range as the cysts from Giardia intestinalis, another waterborne pathogen. The inactivation data for Naegleria are limited, but recent CT values (concentration of disinfectant [mg/l] X contact time [in minutes]) have been developed. Under laboratory conditions, chlorine at a concentration of 1 ppm (1 mg/L) added to 104.4°F (38°C) clear (non-turbid) well water at a pH of 8.01 will reduce the number of viable and more resistant Naegleria fowleri cysts by 99.99% (4 logs) in 56 minutes (CT of 56). Cloudy (turbid) water requires longer disinfection times or higher concentrations of disinfectant.
Salinity: Naegleria fowleri does not survive in sea water and has not been detected in sea water.
In general, CDC does not recommend testing rivers and lakes for the presence of Naegleria fowleri in areas where the ameba is known to naturally occur. There is no established relationship between detection or concentration of Naegleria fowleri and risk of infection. Environmental testing may be warranted for investigations in which Naegleria fowleri detection may be useful for establishing geographical distribution or occurrence in new environments, including disinfected water bodies or household water systems. Public health practitioners can contact CDC’s Emergency Operations Center at 770-488-7100 to discuss possible environmental testing with Naegleria fowleri experts if a case of PAM is confirmed.
- Marciano-Cabral F, Cabral G. The immune response to Naegleria fowleri amebae and pathogenesis of infection. FEMS Immunol Med Microbiol. 2007;51:243-59.
- Visvesvara GS. Free-living amebae as opportunistic agents of human disease. J Neuroparasitol. 2010;1.
- Yoder JS, Eddy BA, Visvesvara GS, Capewell L, Beach MJ. The epidemiology of primary amoebic meningoencephalitis in the USA, 1962-2008. Epidemiol Infect. 2010;138:968-75.
- Gharpure R, Bliton J, Goodman A, Ali IKM, Yoder J, Cope JR. Epidemiology and clinical characteristics of primary amebic meningoencephalitis caused by Naegleria fowleri: a global review. Clin Infect Dis. 2021 Jul 1;73(1):e19-e27.
- Fowler M, Carter RF. Acute pyogenic meningitis probably due to Acanthamoeba sp.: a preliminary report. [PDF – 4 pages] Br Med J. 1965;2:740-2.
- Butt CG. Primary amebic meningoencephalitis. N Engl J Med. 1966;274:1473-6.
- SN Gustavo. Fatal primary amebic meningoencephalitis. A retrospective study in Richmond, Virginia. Am J Clin Pathol. 1970;54:737-42.
- Visvesvara G, Yoder J, Beach MJ. Primary amebic meningoencephalitis Chapter 73. 2012. p. 442-7. In: Netter’s Infectious Diseases, Eds. Yong EC, Stevens DL. Elsevier Saunders. Philadelphia, PA.
- Visvesvara GS, Moura H, Schuster FL. Pathogenic and opportunistic free-living amoebae: Acanthamoeba spp., Balamuthia mandrillaris, Naegleria fowleri, and Sappinia diploidea. FEMS Immunol Med Microbiol. 2007;50(1):1-26.
- Chang SL. Resistance of pathogenic Naegleria to some common physical and chemical agents. [PDF – 8 pages] Appl Environ Microbiol. 1978;35:368-75.
- Griffin JL. Temperature tolerance of pathogenic and nonpathogenic free-living amoebas. Science.1972;178(63):869-70.
- Tiewcharoen S, Junnu V. Factors affecting the viability of pathogenic Naegleria species isolated from Thai patients. J Trop Med Parasitol. 1999;22:15-21.
- De Jonckheere J, van de Voorde H. Differences in destruction of cysts of pathogenic and nonpathogenic Naegleria and Acanthamoeba by chlorine. [PDF – 4 pages] Appl Environ Microbiol. 1976;31:294-7.
- Cursons RT, Brown TJ, Keys EA. Effect of disinfectants on pathogenic free-living amoebae: in axenic conditions. [PDF – 5 pages] Appl Environ Microbiol. 1980;40:62-6.
- P, Gerba C. Inactivation of Naegleria fowleri by chlorine and ultraviolet light. J AWWA. 2012;104:51-2.
- Robinson BS, Christy PE. Disinfection of water for control of amoebae. Water. 1984;11:21-4.
- Ercken D, Verelst L, Declerck P, Duvivier L, Van Damme A, Ollevier F. Effects of peracetic acid and monochloramine on the inactivation of Naegleria lovaniensis. Water Sci Technol. 2003;47(3):167-71.
- Jarroll EL, Bingham AK, Meyer EA. Effect of chlorine on Giardia lamblia cyst viability. [PDF – 5 pages] Appl Environ Microbiol. 1981;41:483-7.
- Rice EW, Hoff JC, Schaefer FW 3rd. Inactivation of Giardia cysts by chlorine. [PDF – 2 pages] Appl Environ Microbiol. 1982;43:250-1.
- Kemble SK, Lynfield R, DeVries AS, Drehner DM, Pomputius WF 3rd, Beach MJ, Visvesvara GS, da Silva AJ, Hill VR, Yoder JS, Xiao L, Smith KE, Danila R. Fatal Naegleria fowleri infection acquired in Minnesota: possible expanded range of a deadly thermophilic organism. [PDF – 5 pages] Clin Infect Dis. 2012;54:805-9.
- Yoder JS, Straif-Bourgeois S, Roy SL, Moore TA, Visvesvara GS, Ratard RC, Hill V, Wilson JD, Linscott AJ, Crager R, Kozak NA, Sriram R, Narayanan J, Mull B, Kahler AM, Schneeberger C, da Silva AJ, Beach MJ. Deaths from Naegleria fowleri associated with sinus irrigation with tap water: a review of the changing epidemiology of primary amebic meningoencephalitis. Clin Infect Dis. 2012;1-7.
- Gharpure R, Gleason M, Salah Z, Blackstock AJ, Hess-Homeier D, Yoder JS, Ali IKM, Collier SA, Cope JR. Geographic Range of Recreational Water–Associated Primary Amebic Meningoencephalitis, United States, 1978–2018 – PMC (nih.gov) Emerg Infect Dis. 2021 Jan;27(1):271-274.