Key points
- Everyone between the ages of 13 and 64 should get tested for HIV at least once.
- People with certain risk factors should get tested more often.
- Most HIV tests are available for free or at a reduced cost.
- Visit gettested.cdc.gov to find HIV testing in your area.
Why get tested
The only way to know your HIV status is to get tested. Knowing your HIV status gives you powerful information to keep you and your partner healthy. If your test result is positive, you can take medicine to treat HIV to help you live a long, healthy life and protect others. If your test result is negative, you can take actions to prevent HIV.
When to get tested
Everyone ages 13 to 64 should get tested for HIV at least once as part of routine health care.
People with certain risk factors should get tested more often. You should get tested at least once a year if:
- You're a man who has had sex with another man.
- You've had anal or vaginal sex with someone who has HIV.
- You've had more than one sex partner since your last HIV test.
- You've shared needles, syringes, or other drug injection equipment (for example, cookers).
- You've exchanged sex for drugs or money.
- You've been diagnosed with or treated for another sexually transmitted infection, hepatitis, or tuberculosis (TB).
- You've had sex with someone who has done anything listed above or you don't know their sexual history.
Did you know?
Gay and bisexual men
Sexually active gay or bisexual men may benefit from more frequent testing (every 3 to 6 months). Talk to your health care provider about your risk factors and what testing options are available to you.
Pregnant people
Pregnant people should get tested for HIV during each pregnancy. Testing pregnant people and treating those who have HIV is a highly effective way to prevent babies being born with HIV.
Types of tests
There are three types of HIV tests: antibody tests, antigen/antibody tests, and nucleic acid tests (NAT). Antibodies are produced by your immune system when you're exposed to viruses like HIV. Antigens are foreign substances that cause your immune system to activate. If you have HIV, an antigen called p24 is produced even before antibodies develop.
HIV tests are typically performed on blood or oral fluid. They may also be performed on urine.
Antibody test
An antibody test looks for antibodies to HIV in your blood or oral fluid. Most rapid tests and the only HIV self-test approved by the U.S. Food and Drug Administration (FDA) are antibody tests. Antibody tests that use blood from a vein can detect HIV sooner than tests done with blood from a finger stick or with oral fluid.
Antigen/antibody test
An antigen/antibody test looks for both HIV antibodies and antigens. Antigen/antibody tests are recommended for testing done in labs and are common in the United States. This lab test involves drawing blood from a vein. There is also a rapid antigen/antibody test available that is done with blood from a finger stick.
Nucleic acid test (NAT)
A NAT looks for the actual virus in the blood. With a NAT, the health care provider will draw blood from your vein and send the sample to a lab for testing. This test can tell if a person has HIV or how much virus is present in the blood (HIV viral load test). A NAT can detect HIV sooner than other types of tests. This test should be considered for people who have had a recent exposure or a possible exposure and have early symptoms of HIV and who have tested negative with an antibody or antigen/antibody test.
Talk to your health care provider about what type of HIV test is right for you.
How to get tested
Order an HIV self-test
Visit gettested.cdc.gov to see if any organizations in your area are offering free or reduced cost self-tests. You can also buy an HIV self-test at a pharmacy or online.
A self-test can be used at home or in a private location. With an HIV self-test, you can get your test results within 20 minutes. You should always interpret HIV self-test results according to the manufacturer's instructions. If the HIV self-test is invalid, then the test did not work. You will need to use another HIV self-test or find testing at a health care provider or testing center.
Go to a testing location
You can ask your health care provider for an HIV test. Many medical clinics, substance abuse programs, community health centers, and hospitals offer them too. Use the locator below to find HIV testing services in your area.
If you get an HIV test in a health care setting or lab, the health care provider will take a sample of blood or oral fluid. With a rapid test (oral fluid or finger stick), you may be able to wait for the results. With a lab test, it may take several days for your results to be available.
If you are tested outside of a health care setting or a lab, you will likely receive a rapid test (oral fluid or finger stick). The counselor providing the test should be able to answer questions and provide referrals for follow-up testing, if needed.
Did you know?
Testing results
Your test results depend on the type of HIV test and where you get tested.
- With a rapid antibody test, usually done with blood from a finger stick or with oral fluid, results are ready in 30 minutes or less. This includes HIV self-tests used at home or in a private location.
- The rapid antigen/antibody test, done with blood from a finger stick, takes 30 minutes or less.
- It may take several days to receive your test results with a NAT or antigen/antibody lab test.
Window period
No HIV test can detect HIV immediately after infection. That's because of the window period—the time between HIV exposure and when a test can detect HIV in your body.
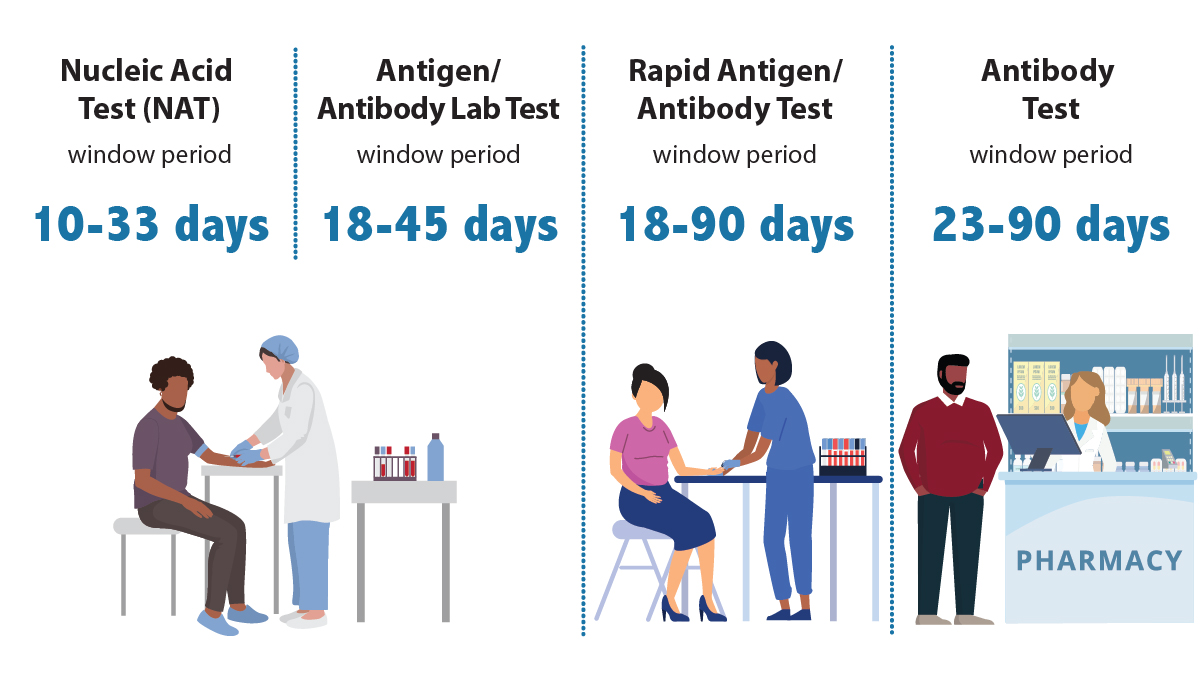
- Antibody tests can usually detect HIV 23 to 90 days after exposure. Most rapid tests and self-tests are antibody tests.
- A rapid antigen/antibody test done with blood from a finger stick can usually detect HIV 18 to 90 days after exposure.
- An antigen/antibody lab test using blood from a vein can usually detect HIV 18 to 45 days after exposure.
- A NAT can usually detect HIV 10 to 33 days after exposure.
If you get an HIV test after a potential HIV exposure and the result is negative, get tested again after the window period for the test you took.
After you get your results
If your test result is positive
If you use any type of antibody test and have a positive result, you will need a follow-up test to confirm your results.
- If you test in a community program or take an HIV self-test and it's positive, you should go to a health care provider for follow-up testing.
- If you test in a health care setting or a lab and it's positive, the lab will conduct the follow-up testing, usually on the same blood sample as the first test.
- If the follow-up test is also positive, it means you have HIV.
Receiving an HIV diagnosis can be life changing. Take time to process the news. Then, find HIV care and start treatment as soon as possible. HIV treatment can make the amount of HIV in your body so low that a test can't detect it (undetectable viral load). Getting and keeping an undetectable viral load is the best way to stay healthy and protect others.
You should share your HIV status with your sex or needle-sharing partners. Communicating your HIV status allows each person to take steps to stay healthy.
Testing positive for HIV does not mean you have AIDS (acquired immunodeficiency syndrome), the most advanced stage of HIV disease. However, HIV can lead to AIDS if a person with HIV does not get treatment or take care of their health.
If your test result is negative
A negative result doesn't necessarily mean that you don't have HIV. That's because of the window period.
- If you get an HIV test after a potential HIV exposure and the result is negative, get tested again after the window period for the type of test you took.
- If you test again after the window period, have no possible HIV exposure during that time, and the result is negative, you do not have HIV.
- If you're sexually active or use needles to inject drugs, continue to take actions to prevent HIV.
- If you have certain risk factors, you should continue getting tested at least once a year.
Remember, your HIV test result reveals only your HIV status. If your test result is negative, that doesn't mean your partner's test result is negative. Consider getting tested with your partner so you can both know your HIV status and take steps to keep yourselves healthy.



