COVID-19 Science Update released: November 3, 2020 Edition 62

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Here you can find all previous COVID-19 Science Updates.
The SARS-CoV-2 major spike protein (S) is cleaved by a host protease to create S1 and S2 functional polypeptides. The S1 polypeptide has a carboxy-terminal sequence that binds to host cell surface neuropilin-1 (NRP1) and neuropilin-2 (NRP2) receptors. NRP receptors facilitate viral entry for several viruses due to their abundance on cells that are exposed to the external environment – cells that paradoxically express low levels of angiotensin-converting enzyme-2 (ACE2), the known SARS-CoV-2 receptor. Here we present two articles that evaluated the potential for NRP1 to act as a co-receptor for SARS-CoV-2.
PEER-REVIEWED
A. Neuropilin-1 is a host factor for SARS-CoV-2 infectionexternal icon. Daly et al. Science (October 20, 2020).
Key findings:
- The S1 fragment of the cleaved SARS-CoV-2 spike protein binds to the cell surface receptor neuropilin-1 (NRP1).
- SARS-CoV-2 utilizes NRP1 for cell entry as evidenced by decreased infectivity of cells in the presence of:
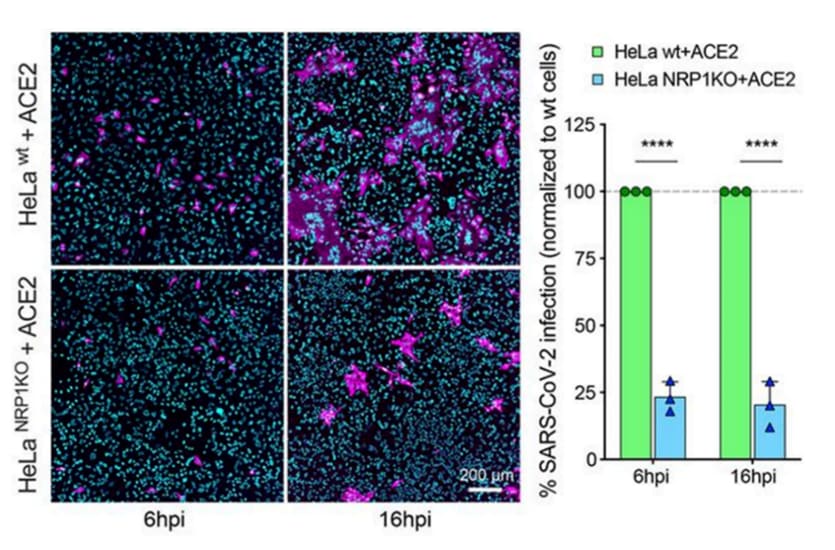
- NRP1 deletion (p <0.0001) (Figure).
- Three different anti-NRP1 monoclonal antibodies (p <0.001).
- Selective NRP1 antagonist, EG00229 (p <0.01).
Methods: Binding of the S1 fragment to NRP1 was assessed and ability of SARS-CoV-2 to use NRP1 to infect cells was measured in angiotensin-converting enzyme-2 (ACE-2)-expressing cell lines by knocking out NRP1 expression, blocking NRP1 with 3 different anti-NRP1 monoclonal antibodies, or using NRP1 small molecule antagonists. Limitations: Upstream research and implications for treatment or prevention of infection unclear.
Figure:
Note: Adapted from Daly et al. Fluorescent image of HeLa cells transfected to express ACE2 (SARS-CoV-2 receptor) with stains for SARS-CoV-2 nucleocapsid protein and nucleic acid, 6 and 16 hours post-infection (hpi). ACE-2 HeLa cells are wild-type (wt, top) or with a NRP1 deletion (NRP1KO, bottom). Bar graph shows % SARS-CoV-2-infected wild-type cells or NRP1-knockout cells. Licensed under CC-BY 4.0.
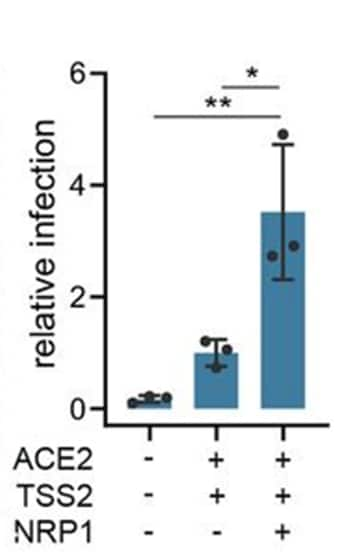
B. Neuropilin-1 facilitates SARS-CoV-2 cell entry and infectivity.external icon Cantuti-Castelvetri et al. Science (October 20, 2020).
Key findings:
- Infectivity of cells expressing angiotensin converting enzyme-2 (ACE2, receptor for SARS-CoV-2), transmembrane protease serine-2 (TSS2, primes the Spike [S] protein), and neuropilin-1 (NRP1) with pseudovirus expressing the SARS-CoV-2 S1 protein was approximately 3-fold higher than in cells expressing either ACE2 or TSS2 alone (p <0.05) (Figure 1).
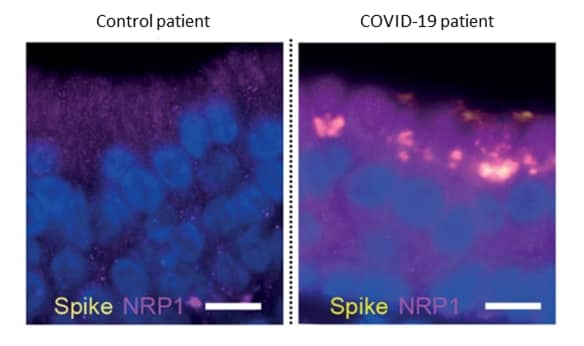
- Analysis of autopsy tissue from COVID-19 patients showed co-localization of the SARS-CoV-2 spike (S) protein and NRP1 in olfactory and respiratory epithelium (Figure 2).
Methods: Cell lines were modified to express ACE2 and TSS2, the two known SARS-CoV-2 host factors, and NRP1 to assess the contribution of NRP1 to infection. Autopsy specimens from multiple airway sites were stained with antibodies against SARS-CoV-2 proteins, ACE2, and NRP1, to visualize co-localization of proteins. Limitations: Upstream research and implications for treatment or prevention of infection unclear.
Figure 1
Note: Adapted from Cantuti-Castelvetri et al. Infectivity of SARS-CoV-2 in cultured cells that express no known host factors (first bar), the host factors ACE2 and TSS2 (middle bar) and ACE2, TSS2, and NRP1 (third bar). *p <0.05, **p <0.01. Licensed under CC-BY 4.0.
Figure 2
Note: Adapted from Cantuti-Castelvetri et al. Co-localization of NRP1 and Spike protein in olfactory epithelial cells in a control patient (left) and a COVID-19 patient (right). Licensed under CC-BY 4.0.
Implications for 2 studies (Daly et al. & Cantuti-Castelvetri et al.) These two articles provide evidence that NRP1 facilitates SARS-CoV-2 entry into cells that express ACE2. These studies elucidate a route by which SARS-CoV-2 infects cells that do not express high levels of the ACE2 receptor and also suggests potential therapeutic options to decrease infection by blocking SARS-CoV-2-NRP1 interactions.
PEER-REVIEWED
Neutralizing antibody responses in COVID-19 convalescent seraexternal icon. Lee et al. Journal of Infectious Diseases (October 26, 2020).
Key findings:
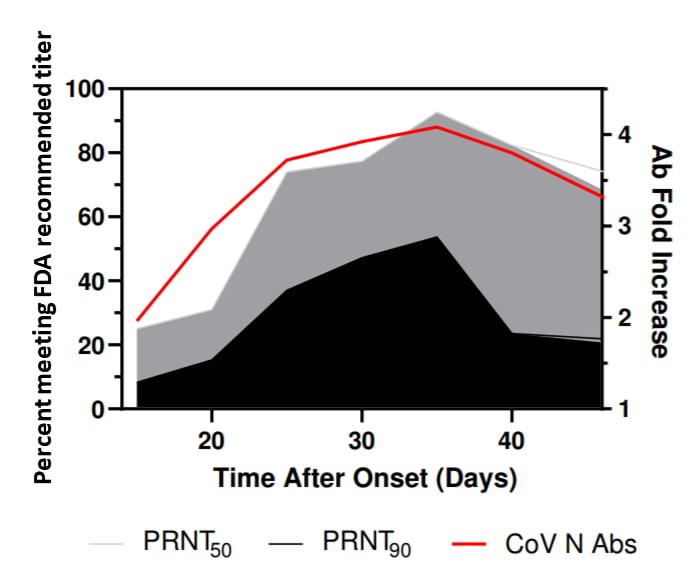
- Neutralizing antibody (nAb) activity increased for several weeks post-symptom onset (Figure).
- 93% of sera had titers ≥1:160 (the initial FDA recommended level for nAb for convalescent plasma donation) at the 50% viral neutralization level, 31–35 days post-symptom onset.
- 54% of sera had titers ≥1:160 at the 90% viral neutralization level, 31–35 days post-symptom onset.
- 24% of sera had titers of ≥1:160 at the 90% viral neutralization level beyond day 35.
- Sera with antibody titers >1:960 generally had higher neutralizing activity, but not all contained nAbs at the initial FDA recommended levels.
Methods: Serum specimens from 3,426 COVID-19 convalescent patients were tested for SARS-CoV-2 antibodies to the receptor binding protein, spike protein, or nucleocapsid. Reactive sera were tested for ability to neutralize SARS-CoV-2 infection using plaque reduction assays. Limitations: Short follow-up period.
Implications: There may be a narrow window to collect convalescent plasma with maximal neutralizing activity for use as potential COVID-19 therapy. Findings also highlight the importance of characterizing serum neutralizing activity.
Figure:
Note: Adapted from Lee et al. Percentage of specimens that can neutralize 50% or neutralize 90% or of viral plaque formation with an antibody titer of ≥1:160 (left Y-axis) and the fold increase of mean antibody levels to the SARS-CoV-2 nucleocapsid (right Y-axis). Lee et al. Neutralizing antibody responses in COVID-19 convalescent sera. Journal of Infectious Diseases. DOI: https://doi.org/10.1093/infdis/jiaa673external icon ; Used by permission of the Infectious Diseases Society of America.
Frequency and duration of SARS-CoV-2 shedding in oral fluid samples assessed by a modified commercial rapid molecular assay.external icon Bordi et al. Viruses (October 20, 2020).
Key findings:
- Parallel testing for SARS-CoV-2 RNA in 337 oral fluid (OF) and nasopharyngeal (NP) specimens found 309 concordant and 28 discordant results (kappa statistic = 0.831, 95% CI 0.771-0.891).
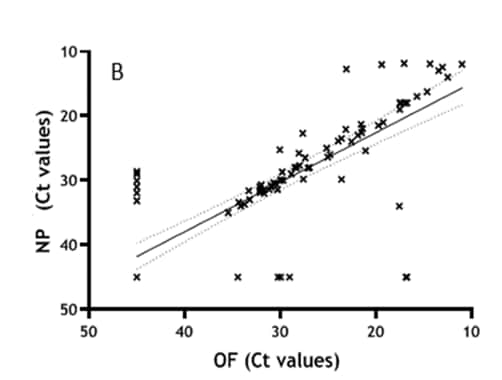
- Ct values for OF and NP specimens were highly correlated (correlation coefficient r = 0.921; p <0.0001) (Figure).
- SARS-CoV-2 RNA was observed in:
- 67% of OF and 72% of NP specimens collected within 30 days of symptom onset (DSO).
- 65% of OF and 76% of NP specimens collected between 30 and 60 DSO.
- 32% of OF and 29% of NP specimens collected between 60 and 100 DSO.
Methods: The Simplexa™ COVID-19 Direct assay was used to test concordance in 337 paired OF and NP specimens from 164 patients hospitalized with suspected COVID-19. A sub-sample of 162 specimens was analyzed for viral RNA in OF and NP specimens, by DSO range. Limitations: Did not evaluate effectiveness of OF or NP samples for specific clinical, surveillance, and epidemiologic uses.
Implications: Several assays find that OF samples are highly correlated with NP samples and may be an alternative to NP samples for diagnosis of SARS-CoV-2 infection. As noted in a recent paper in Clinical Infectious Diseases (Azzi, L. Saliva is the Key Element for SARS-CoV-2 Mass Screeningexternal icon), saliva can be easily and noninvasively self-collected, minimizing transmission risk to clinicians and facilitating more frequent testing of contacts, which may be critical to containing spread of SARS-CoV-2.
Figure:
Note: Adapted from Bordi et al. Results from samples tested for SARS-CoV-2 RNA both in NP and OF displayed in linear regression adjusted for repeated measures, sex and age (r = 0.921; p <0.0001). Licensed under CC-BY 4.0.
PREPRINT (NOT PEER-REVIEWED)
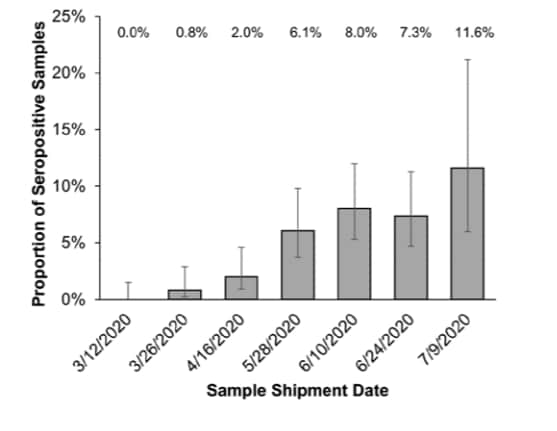
Prevalence of antibodies to SARS-CoV-2 in healthy blood donors in New York.external icon Kamath et al. MedRxiv (October 21, 2020).
Key findings:
- 68 of 1,559 blood donor samples had IgG antibodies against SARS-CoV-2.
- Seropositivity rate increased from 0% (95% CI 0-1.5%) in March 2020 to 11.6% (95% CI 6.0-21.2%) in July 2020.
Methods: Screen for antibodies to SARS-CoV-2 in samples from 1,559 healthy blood donors from the greater New York metropolitan area between March and July 2020. Limitations: Unclear what proportion of donors were asymptomatic cases.
Implications: Although the pandemic significantly affected the greater New York metropolitan area, seropositivity for SARS-CoV-2 antibodies was low indicating that the population is far from reaching a threshold for herd immunity.
Figure:
Note: Adapted from Kamath et al. SARS-CoV-2 seropositivity over time in blood donors. Shipment date is when sample was sent to the laboratory for testing (within 2 weeks of sample collection). 𝙸 bar represents the 95% CI. Used by permission of author.
PEER-REVIEWED
COVID-19 transmission in US child care programs.external icon Gilliam et al. Pediatrics (October 1, 2020).
Key findings:
- 0.7% (n = 427) of child care providers reported testing positive or being hospitalized for COVID-19.
- No association was found between exposure to child care programs and COVID-19 (OR 0.94; 95% CI 0.73-1.21).
- Being a home-based rather than center-based provider was associated with diagnosis of COVID-19 (OR 1.59, 95%CI 1.14-2.23) but this was independent of reported exposure.
Methods: Survey of 57,335 child care providers between May 22 and June 8, 2020, identified through two national child care organizations and 28 state child care workforce registries. Participants were surveyed on exposure to child care programs in the previous 8 weeks and whether they had tested positive for SARS-CoV-2 or had been hospitalized for COVID-19. Logistic regression was conducted to control for confounders (county COVID-19 mortality rate, hand washing and social distancing, child care facility type). Limitations: Child care program not defined; state policy-level variables may not have been adequately controlled.
Implications: Early in the pandemic, exposure to child care programs did not appear to place child care providers at increased risk of COVID-19. This may be partially attributable to infection mitigation efforts in child care programs and background incidence rates. Infections among child care providers will need to be re-evaluated once facilities more broadly re-open.
Social determinants of COVID-19 mortality at the county level.external icon Fielding-Miller et al. PLoS One (October 14, 2020).
Key findings:
- The number of COVID-19 deaths per 100,000 was substantially higher in urban than non-urban counties, 39.1 per 100,000 (IQR: 15.0–107.3) and 5.6 (IQR: 0–19.7), respectively.
- Factors independently associated with higher numbers of deaths for the US overall included:
- % of farmworkers (5.79 additional deaths per percentage point increase of farmworkers, p = 0.001).
- % of residents living in poverty (4.41 additional deaths per percentage point increase, p <0.001).
- % of residents aged >65 years (4.58 additional deaths per percentage point increase, p = 0.002).
- Population density (0.25 additional deaths per resident per square mile, p <0.001).
- Factors independently associated with deaths per 100,000 persons varied substantially by census region.
Methods: Spatial models to determine county-level associations between COVID-19 mortality and percentage of hired farmworkers, percentage of households without a fluent, adult English-speaker, percentage of uninsured individuals under the age of 65, and percentage of individuals living at or below the federal poverty line, across all 3,024 counties as of July 12, 2020. Limitations: Negative associations in some regions might represent unmeasured confounding or gaps in testing or data collection for poor and uninsured individuals.
Implications: Findings indicate that the epidemic is heterogenous and is affecting geographic regions differently. The differences in geographic regions might, in part, reflect regional demographic variation or varied state and local social distancing and other public health policies.
PREPRINT (NOT PEER-REVIEWED)
What predicts adherence to COVID-19 government guidelines? Longitudinal analyses of 51,000 UK adults.external icon Wright et al. MedRxiv. (October 21, 2020).
Key findings:
- Increased confidence in government to handle the pandemic was related to higher compliance with government COVID-19 guidelines.
- Mental health and wellbeing, worries about future adversities, and social isolation and loneliness were unrelated to compliance.
Methods: Data from a longitudinal study of over 51,000 adults in the UK were used to determine factors associated with compliance with government guidelines during the initial implementation of social distancing measures (April 1 to June 22, 2020). Factors studied included mental health and wellbeing, social experiences, confidence in institutions, knowledge of COVID-19, and time spent on various activities. Limitations: All data were self-reported.
Implications: Findings highlight the role of policy makers in shaping the impact of compliance with mitigation efforts.
PEER-REVIEWED
Registry of arterial and venous thromboembolic complications in patients with COVID-19.external icon Piazza et al. Journal of the American College of Cardiology (November 3, 2020)
Key findings:
- 4.6%, 5.9%, and 8.3% of COVID-19 patients experienced a symptomatic thromboembolic event (VTE), a major arterial or venous VTE, or major adverse cardiovascular event, respectively, within 30 days of diagnosis.
- Events occurred despite high rates of thromboprophylaxis in ICU (90%) and general medical ward (GMW) (85%) patients.
- No events occurred in outpatients.
Methods: Retrospective study of patients diagnosed with RT-PCR-confirmed COVID-19 at one health network between March 13 and April 3, 2020. Primary outcome was the proportion of ICU patients (n = 170), GMW patients (n = 229) and outpatients (n = 715) who experienced a symptomatic VTE (e.g., deep vein thrombosis), major arterial or VTE (e.g., stroke) or major adverse cardiovascular event (e.g., heart failure) within 30 days of diagnosis. Limitations: Microvascular thrombosis was not assessed; unable to assess adherence to antiplatelet or antithrombotic therapy prior to COVID-19.
Implications The high frequency of arterial or venous thromboembolism in hospitalized patients with COVID-19, despite routine thromboprophylaxis, suggests the need for improved risk stratification and enhanced preventive efforts.
PREPRINTS (NOT PEER-REVIEWED)
Viral infections have been associated with several autoimmune diseases. These infections can trigger production of autoantibodies that are directed toward the host’s own body potentially causing tissue damage and transient or chronic symptoms and disease.
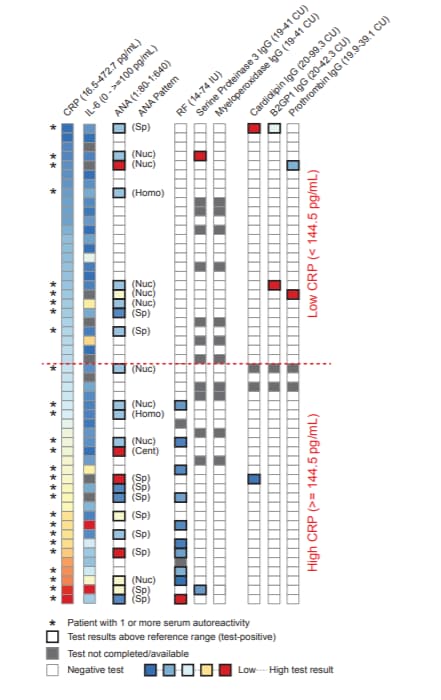
Clinically identifiable autoreactivity is common in severe SARS-CoV-2 infectionexternal icon. Woodruff et al. MedRxiv (October 28, 2020).
Key findings:
- 44% of patients had antinuclear antibody (ANA) ≥1:80 (the diagnostic threshold for the autoimmune disease systemic lupus erythematosus) and 81% of these positive tests had titers ≥1:160.
- 59% of ANA-positive patients had ≥1 other positive autoreactive antibody test such as rheumatoid factor (10/52), phospholipids (3/52), prothrombin (2/52), or antineutrophil cytoplasmic antibodies (2/52) (Figure).
- All patients with >1 positive autoreactive antibody test were positive for either ANA or rheumatoid factor.
- Autoreactivity was correlated with increasing serum levels of c-reactive protein (CRP), a protein found in the bloodstream in response to inflammation.
Methods: Study of 52 severe or critically ill patients with COVID-19 and no known history of autoimmune disease treated at two ICUs in Atlanta, Georgia, between June 1st and July 2nd, 2020. Patients were tested for a variety of autoimmune antibodies. Limitations: Limited follow-up.
Implications: Severe COVID-19 may cause de novo autoreactive antibody production against a variety of self-antigens which could argue for clinical interventions aimed at modifying the immune response.
Figure:
Note: Adapted from Woodruff et al. Heatmap display of clinical results organized by ascending CRP values. Values for individual tests are indicated following the test name (top of figure). Horizontal red dotted line indicates low or high levels of serum CRP. Used by permission of author.
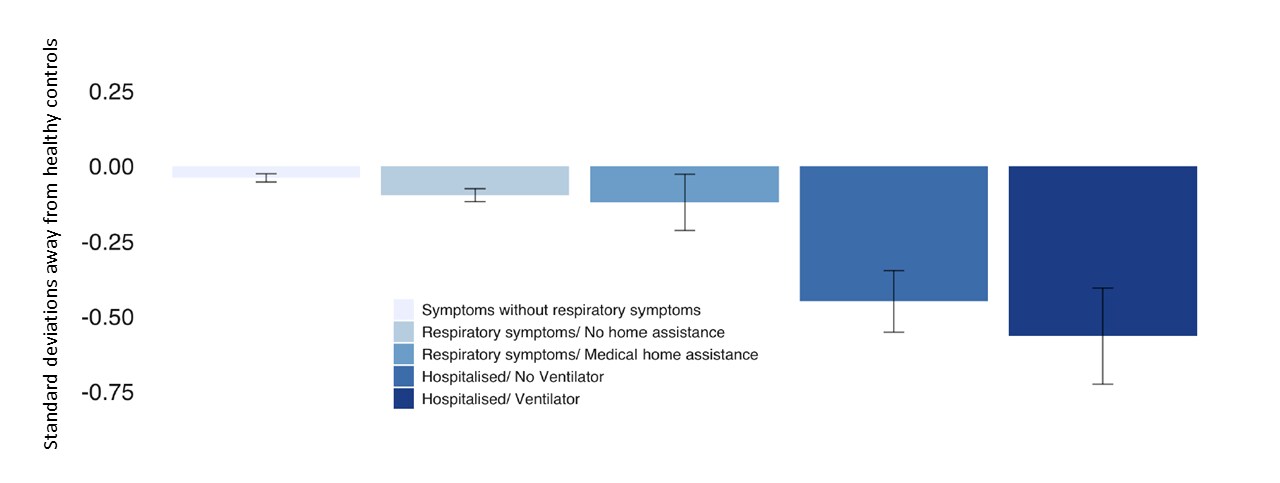
Cognitive deficits in people who have recovered from COVID-19 relative to controls: An N = 84,285 online studyexternal icon. Hampshire et al. mMedRxiv. (October 21, 2020).
Key findings:
- Decreased global cognitive performance was seen among the following (Figure 1):
- In persons hospitalized and on ventilators cognitive performance was -0.57 standard deviations (SDs) below healthy controls (approximates an 8.5-point difference in IQ).
- Persons hospitalized but not on a ventilator, -0.45 SDs.
- Persons assisted at home for respiratory difficulty, -0.12 SDs.
- Persons with respiratory difficulty but received no medical assistance, -0.10 SDs.
- Persons ill without respiratory difficulty, -0.04 SDs.
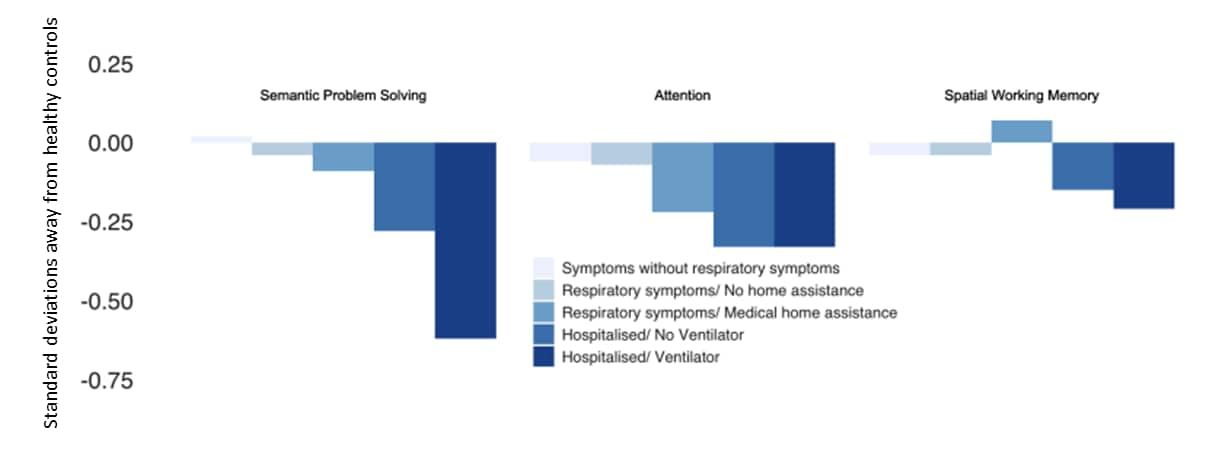
- The most apparent deficits were in semantic problem solving and visual selective attention which were associated with disease severity (Figure 2).
Methods: An online survey of cognition, wellbeing, and behavior of 84,285 participants who completed a questionnaire regarding impacts of the pandemic among persons with suspected (n = 12,689) and confirmed (n = 361) SARS-CoV-2 infection were analyzed to determine associations between COVID-19 and cognitive deficits. Testing addressed semantic problem solving, spatial working memory, selective attention and emotional processing. Limitations: Most cases unconfirmed; symptom severity was self-reported; cross-sectional design.
Implications: Cognitive deficits were found in persons with history of COVID-19 and there may be a need for cognitive assessment of persons recovered from COVID-19.
Figure 1
Note: Adapted from Hampshire et al. Global cognitive performance by COVID-19 symptom severity. Licensed under CC-BY 4.0.
Note: Adapted from Hampshire et al. Cognitive domain performance by symptom severity. Licensed under CC-BY 4.0.
Testing
- Rafiei & Mello. The missing piece — SARS-CoV-2 testing and school reopeningexternal icon. Authors argue that SARS-CoV-2 testing is necessary to safely re-open schools and discuss challenges in implementing school testing programs and ways in which state and federal governments can assist.
- Rubin, R. The challenges of expanding rapid tests to curb COVID-19external icon. Authors discuss the merits of ramping up rapid SARS-CoV-2 testing and challenges including variable test accuracy, manufacturing logistics and public perceptions that testing can be used in lieu of other preventive measures such as mask wearing.
Mental health and well being
- Fong & Iarocci. Child and family outcomes following pandemics: A systematic review and recommendations on COVID-19 policiesexternal icon. Journal of Pediatric Psychology. A systematic review of mental health outcomes and needs of children and families during past pandemics.
- Ickert et al. Maintaining resident social connections during COVID-19: Considerations for long-term careexternal icon. Gerentology and Geriatric Medicine. Considerations for facilitating social connections of long-term care residents and their families while restrictions on visitation are in place.
Miscellaneous
- Vignesh et al. Is herd immunity against SARS-CoV-2 a silver lining?external icon Frontiers in Immunology. Authors discuss a variety of elements that factor into whether populations reach herd immunity thresholds and argue that the most practical way to achieve this is through vaccination.
- Blecker et al. Hospitalizations for chronic disease and acute conditions in the time of COVID-19external icon. JAMA Internal Medicine. Study found a substantial decrease in the number of non-COVID-19 related hospitalizations between March 22 to April 11, 2020.
- Lan et al. Effects of universal masking on Massachusetts healthcare workers’ COVID-19 incidenceexternal icon. Occupational Medicine. Study found universal masking, implemented in healthcare systems statewide, was associated with decreasing incidence trend of COVID-19 among healthcare workers while incidence rose in the surrounding community.
- Figueiredo-Campos et al. Seroprevalence of anti‐SARS‐CoV‐2 antibodies in COVID‐19 patients and healthy volunteers up to six months post disease onsetexternal icon. European Journal of Immunology. Study of 2,998 COVID-19 patients demonstrated that IgG antibodies to SARS-CoV-2 receptor-binding domain protein lasted up to 6 months post-symptom onset and that titers were proportional to neutralizing activity.
- Ong et a Environmental contamination in a COVID-19 intensive care unit (ICU) – what is the riskexternal icon? Infection Control and Hospital Epidemiology. Study found that environmental contamination with SARS-CoV-2 in a hospital intensive care unit was lower than that of a general medical floor and that use of mechanical ventilation or high-flow nasal oxygen was not associated with greater surface contamination.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.