COVID-19 Science Update released: October 2, 2020 Edition 53

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Here you can find all previous COVID-19 Science Updates.
PEER-REVIEWED
SARS-CoV-2 seroprevalence among healthcare, first response, and public safety personnel, Detroit metropolitan area, Michigan, USA, May – June 2020. Akinbami et al. Emerging Infectious Diseases (September 21, 2020).
Key findings:
- Of 16,397 participants, 6.9% (95% CI 6.5% – 7.3%) were positive for SARS-CoV-2 IgG.
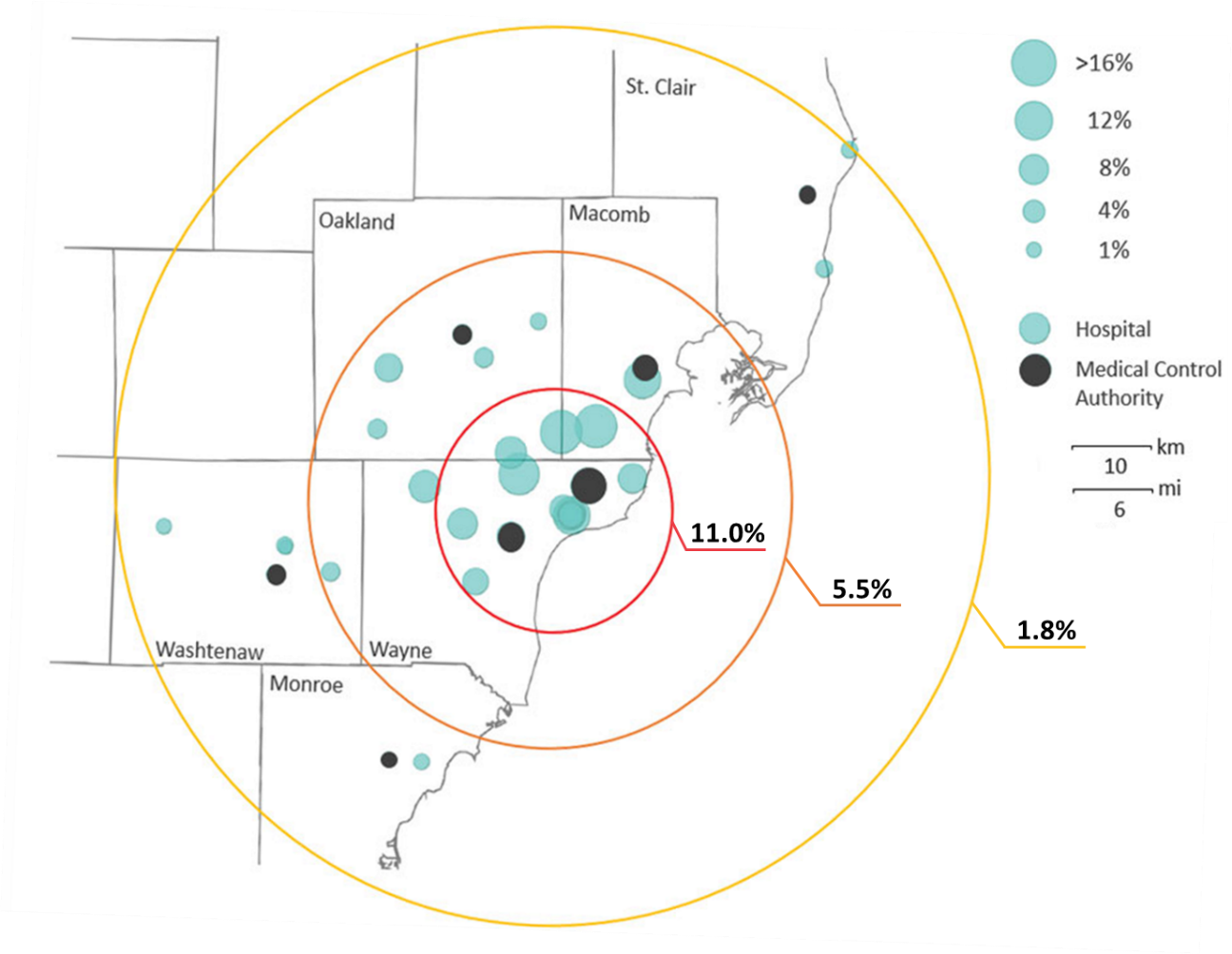
- Seroprevalence was highest (11.0%, 95% CI 10.3% – 11.7%) at facilities within 15 km of Detroit’s center and lowest (1.8%, 95% CI 1.4% – 2.2%) at locations 30 – 55 km away (Figure).
- Exposure to a household member with confirmed COVID-19 (adjusted odds ratio [aOR] 6.18, 95% CI 4.81 – 7.93) and working within 15 km of Detroit’s center (aOR 5.60, 95% CI 3.98 – 7.89) were strongly associated with seropositivity.
- Consistently using N95 respirators (aOR 0.83, 95% CI 0.72 – 0.95) or surgical facemasks (aOR 0.86, 95% CI 0.75 – 0.98) decreased the likelihood of seropositivity.
Methods: Seroprevalence study in adults (≥18 years of age) working as first responders, healthcare providers, or in public safety settings from May to June 2020. Limitations: Convenience sample with ~80% response rate; comprehensive exposure data were not collected; infected participants may have failed to seroconvert or antibody levels may have decayed in cases of remote infection, or not yet present in cases of recent infection, leading to false negatives.
Implications: The association between seropositivity and working closer to the Detroit city center and exposure to a household member with COVID-19 illustrates the major role of community acquisition of SARS-CoV-2, even in healthcare personnel. Measures to reduce community transmission will be protective for all community members including health care personnel who may be at risk in settings where social distancing and personal protective equipment use may be difficult.
Figure:
Note: From Akinbami et al. Seropositivity of study population by hospital or medical control authority agency location within the Detroit Metropolitan Area. Mean SARS-CoV-2 seroprevalence within 15 km was 11.0%; 15 – 30 km range was 5.5%; 31 – 55 km range was 1.8%. Open access journal; all content freely available.
The effect of vascular risk factor burden on the severity of COVID-19 illness, a retrospective cohort studyexternal icon. Du et al. Respiratory Research (September 21, 2020).
Key findings:
- 90.2% (n = 148) of COVID-19 patients had at least one vascular risk factor.
- There was an increased association between vascular risk factors and severe COVID-19 (adjusted hazard ratio 1.55, 95% CI 1.09 –21, p <0.01).
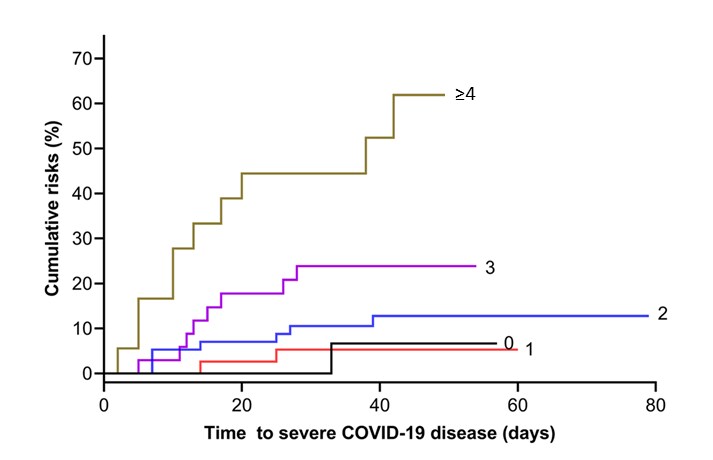
- Increasing burden of vascular risk factors was associated with increased risk of severe COVID-19 illness (log rank p <0.001) (Figure).
Methods: Retrospective cohort of patients with COVID-19 (n = 164) between February 14 and March 14, 2020, admitted to a single center in Wuhan, China. Study evaluated progression to severe COVID-19 illness among those with increasing burden of cardiovascular risk factors, including: hypertension, diabetes, dyslipidemia, atrial fibrillation, current smoking, regular alcohol drinker, physical inactivity, and overweight status. Limitations: Single-center study with small sample size; some risk factors were self-reported; vascular risk factors not evaluated may confound results.
Implications: Vascular risk factor burden is associated with progression to severe COVID-19 illness. Self-isolation or increased personal precautions and interventions to modify vascular risk factors such as exercise, smoking cessation, medication adherence, weight loss, and reducing alcohol intake may benefit this population.
Figure:
Note: Adapted from Du et al. Cumulative probability of severe COVID-19 stratified by 0, 1, 2, 3, and ≥4 vascular risk factors. Licensed under CC 4.0.
Surveillance of COVID-19 school outbreaks, Germany, March to August 2020external icon. Otte im Kampe, et al. Eurosurveillance. (September 24, 2020).
Key findings:
- Schools accounted for 48 (0.5%) of 8,841 COVID-19 outbreaks in Germany and included 61,540 cases.
- 58% of school outbreaks included individuals only in the same grade.
- 21% of school outbreaks occurred in non-students (<21 years of age) only.
- Non-students accounted for 47% of cases from all outbreaks.
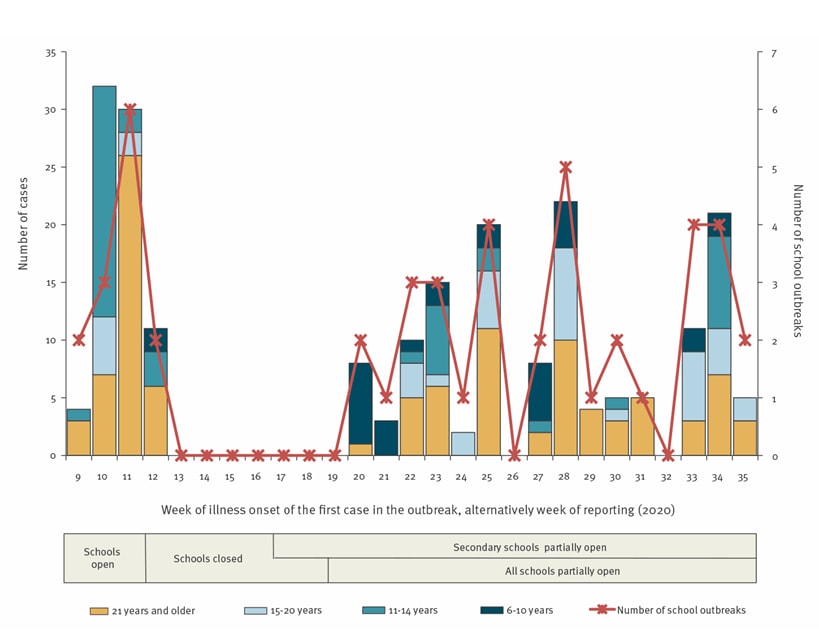
- On average, 2.2 school outbreaks occurred per week with 4 cases per outbreak following school re-openings (Figure).
- 75% of cases with available clinical data reported symptoms; reporting of symptoms increased with age.
Methods: Analysis of German COVID-19 national surveillance system for school outbreaks (≥2 laboratory-confirmed cases in persons from the same school) from January 28 to August 31, 2020. Limitations: Methodology used for investigation not described; outbreaks with large proportion of asymptomatic cases may not have been detected; no clear school denominator for different time periods.
Implications: Few school outbreaks were reported and most were reported within the same grade suggesting preventive measures can prevent spillover to other grades. As younger students were less likely to be symptomatic, symptomatic surveillance should focus on older individuals. These analyses were done as schools were only partially open, which underscores the need for continued surveillance and contact tracing when schools fully reopen.
Figure:
Note: Adapted from Otte im Kampe, et al. School outbreaks of COVID-19. Primary y-axis shows number of cases reported among school outbreaks by week (x-axis) between January 28 and August 31, 2020 by age: cases >21 years old, 15-20, 11-14, and 6-10. Secondary y-axis shows number of school outbreaks reported weekly. Licensed under CC 4.0.
PEER-REVIEWED
The increased risk of COVID-19 morbidity and mortality in residents of nursing homes has been recognized. While nursing homes tend to have patients who require care due to physical or mental conditions, persons in assisted living facilities are also vulnerable based on age, underlying comorbidities and communal living conditions. The following two articles examine factors related to infection and case fatality rates at both nursing homes and assisted living facilities.
A. Racial and ethnic disparities in COVID-19 infections and deaths across U.S. nursing homes.external icon Li et al. Journal of Gerontology. (September 21, 2020).
Key findings:
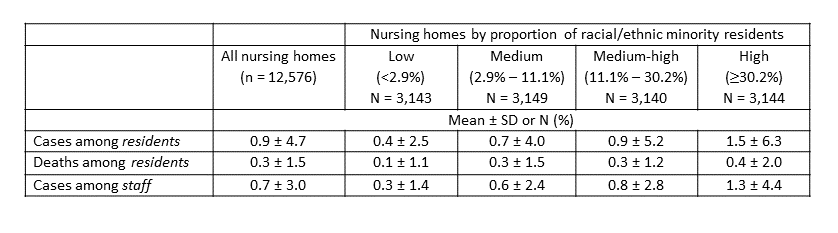
- Nursing homes with >30.2% of racial/ethnic minority residents had an average of 1.5 new resident cases, 1.3 new staff cases, and 0.4 new resident deaths per week per facility compared with those in the bottom quartile, that had an average of 0.4 new resident cases, 0.3 new staff cases, and 0.1 new resident deaths (Table).
- Nursing homes with between 2.9% and 30.2% racial/ethnic minority residents and those with >30.2% minority residents were 25% and 76% more likely to have a new resident case compared with those with the fewest minorities residents (OR 1.25, 95% CI 1.03 – 1.51, p = 0.025 and OR 1.76, 95% CI 1.38 – 2.25, p <0.001, respectively).
Methods: A cross-sectional analysis of national Center for Medicare and Medicaid Services (CMS) COVID-19 data (collected in CDC’s National Healthcare Safety Network) in 12,576 nursing homes stratified into quartiles by percentage of racial/ethnic minority residents. Limitations: Cross-sectional data only allows for association; data on staff death were too limited for analysis.
Table:
Note: Adapted from Li et al. Numbers of incident laboratory-confirmed COVID-19 cases and deaths reported in US nursing homes during the week of May 25 to May 31, 2020 by proportion of racial/ethnic minority residents. Used by permission of John Wiley & Sons, Inc. © American Geriatrics Society.
B. COVID-19 pandemic in assisted living communities: Results from seven statesexternal icon. Temkin-Greener et al. Journal of the American Geriatrics Society (September 21, 2020).
Key findings:
- COVID-19 death rates in assisted living centers were higher than state-level death rates.
- The proportion of minority residents was associated with the likelihood of COVID-19 cases (incidence rate ratio [IRR] = 1.08; p <0.001).
- The proportion of minority residents was not associated with the likelihood of deaths (IRR = 0.98; p = 0.739) after controlling for co-morbidities and assisted living center characteristics.
Methods: Observational study employing Medicare Beneficiary data and county-level COVID-19 data to examine COVID-19 outcomes, confirmed cases and deaths in assisted living centers from seven states through May 2020. The sample included 3,994 assisted living centers, 2,542 cases and 675 deaths. Limitations: Generalizability may be limited as only 7 states were included; observational data may be subject to unmeasured confounders.
Implications for 2 studies (Li et al. and Temkin-Greener): Nursing homes have structural inequities that contribute to differences in COVID-19 morbidity and mortality by race-ethnicity. While there may be important differences in visitation policies and interaction with the community, these same or similar facility-level structural inequities may extend to assisted living facilities. The impact of COVID-19 on assisted living centers may be similar to that of nursing homes and deserves consideration from policy makers.
PEER-REVIEWED
The prevalence and consequence of coinfection of SARS-CoV-2 with other viral respiratory pathogens is unknown. In pre-pandemic years, pediatric coronavirus bronchiolitis (inflammation and congestion in the small airways of young children and infants caused by a viral infection) was often associated with coinfection with other respiratory viruses; respiratory syncytial virus (RSV) being most commonly identified (Mansbach et alexternal icon). Here we present two studies looking at other respiratory pathogen infections in children with SARS-CoV-2 infection.
A. Characterizing coinfection in children with COVID-19: A dual center retrospective analysisexternal icon. Zhang et al. Infection Control and Hospital Epidemiology (September 23, 2020).
Key findings:
- Among 767 pediatric patients tested for SARS-CoV-2 and other respiratory pathogens, 101 (13.2%) were positive for SARS-CoV-2.
- 12.5% were coinfected with rhinovirus, enterovirus or adenovirus; 2% were coinfected with either influenza A or respiratory syncytial virus (RSV).
Methods: Retrospective review of records at two Chicago medical centers from March 9 through April 30, 2020 of all pediatric patients tested for SARS-CoV-2 by RT-PCR who also were tested for other respiratory pathogens within 7 days of the SARS-CoV-2 test. Limitations: No age range is presented, limiting ability to compare with other studies among children; average age in study is 17.1, but RSV and bronchiolitis are more common in children under 5 years; study was conducted at a time of year when respiratory viral transmission rates were declining.
B. COVID-19 public health measures and respiratory syncytial virus.external icon Britton et al. Lancet Child & Adolescent Health (September 18, 2020).
Key findings:
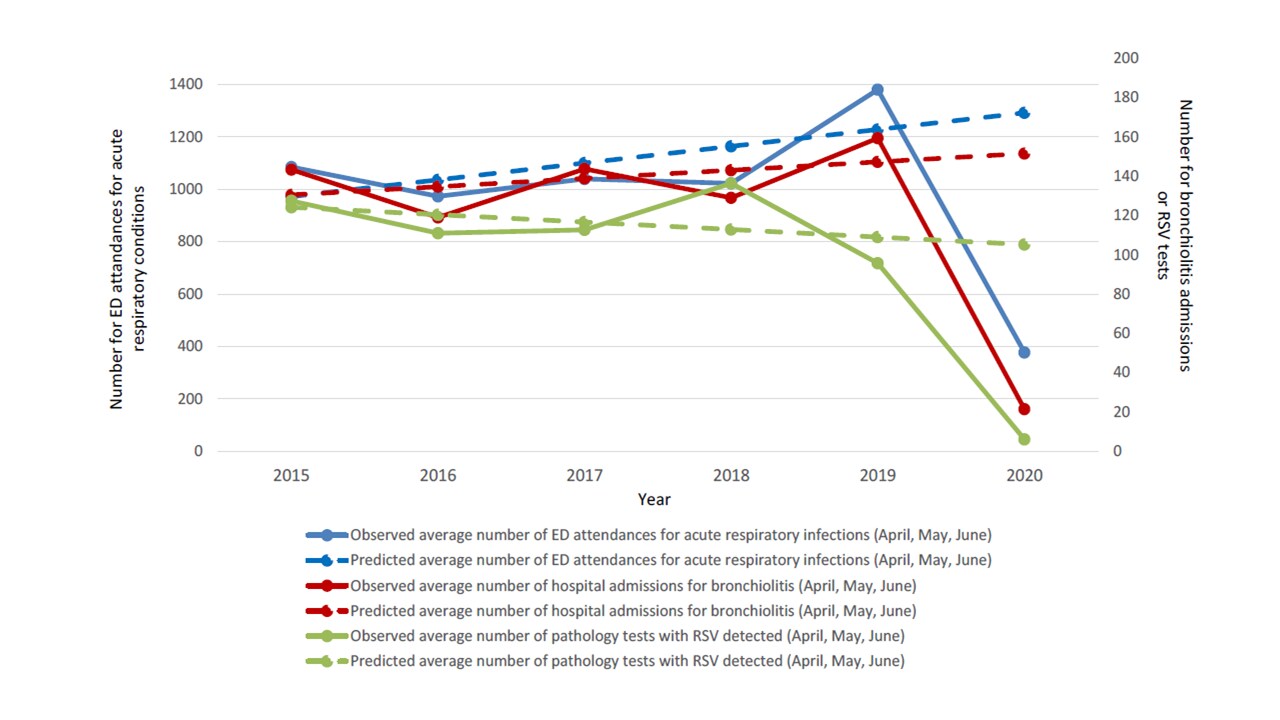
- Over a five-year medical record review, the majority of positive tests for RSV (63.7%); bronchiolitis admissions (99.6%); and respiratory-related pediatric emergency department (ED) visits (63.4%) were in the 0 – 2 year age-group (Figure 1).
- Following implementation of aggressive public health measures to prevent SARS-CoV-2 transmission, there were 94.3% fewer RSV positive tests, 85.9% fewer admissions for bronchiolitis, and 70.9% fewer ED visits in 2020, compared with previous years (Figures 1 &2).
- The number of RSV tests done in 2020 was double that of previous years.
Methods: Retrospective review of records from a hospital network in New South Wales (NSW), Australia, for three events: RSV PCR test (n = 69,646), bronchiolitis hospital admission (n = 6,730), and ED visit for acute respiratory illness (n = 58,491) among children younger than 16 years between January 1 and June 30, 2020. Frequencies of events in peak RSV epidemic months (April – June) in 2020 and same time period in 2015 – 2019 were analyzed. Limitations: Findings may be specific to NSW; actual uptake and effect of mitigation measures such as handwashing, social distancing, and reduced population movement was not measured; study period was brief and cannot determine if results equate to true reduction in RSV.
Figure 1
Note: Adapted from Britton et al. Monthly RSV PCR detections for 2014–2019 (blues) and January – June, 2020 with pandemic response shutdown time period noted.
Figure 2
Note: Adapted from Britton et al. Observed and predicted trends of the frequency of RSV PCR detections, Bronchiolitis admissions, and ED acute respiratory visits 2015 to 2020 for children aged <16 years. This article was published in Lancet Child & Adolescent Health, Vol 4, Britton et al., COVID-19 public health measures and respiratory syncytial virus, Page E42-43, Copyright Elsevier 2020. This article is currently available at the Elsevier COVID-19 resource center: https://www.elsevier.com/connect/coronavirus-information-centerexternal icon.
Implications for 2 studies (Zhang et al. & Britton et al.): Implementation of public health measures to prevent SARS-CoV-2 infection during peak RSV season in NSW, Australia, was strongly associated with a large decrease in the burden of RSV disease among children. Compared to endemic coronaviruses, coinfections with SARS-CoV-2 appear less common in the limited pediatric population data currently available but this may be partially a result of current prevention practices including handwashing and social distancing.
PEER-REVIEWED
Risk factors for hospitalization, mechanical ventilation, or death among 10 131 US veterans with SARS-CoV-2 infectionexternal icon. Ioannou et al. JAMA Network Open (Sept 23, 2020).
Key findings:
- Those who tested positive for SARS-CoV-2 had higher rates of 30-day hospitalization (30.4% vs 29.3%; adjusted hazard ratio (aHR) 1.13, 95% CI 1.08 – 1.17), mechanical ventilation (6.7% vs 1.7%; aHR 4.15, 95% CI 3.74 – 4.61), and death (10.8% vs 2.4%; aHR 4.44; 95% CI 4.07 – 4.83) compared with patients who tested negative.
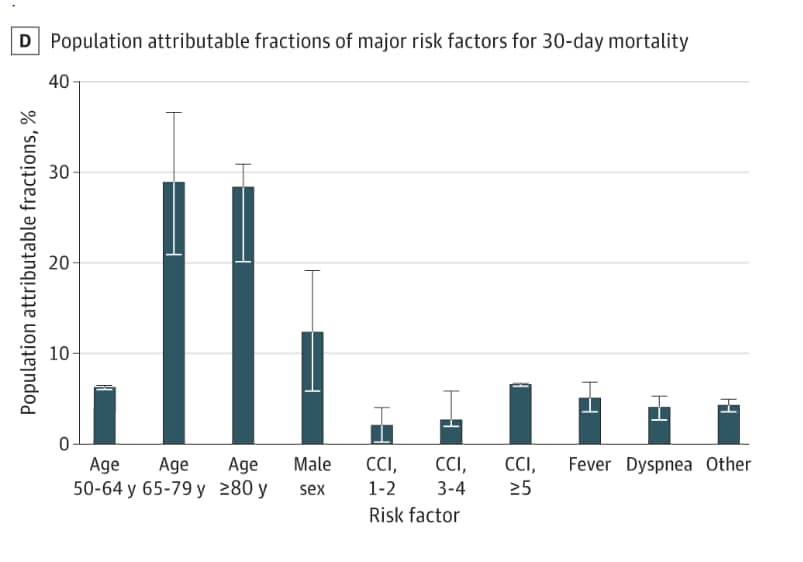
- Among patients who tested positive for SARS-CoV-2, mortality was associated with older age, high regional COVID-19 disease burden, higher Charlson comorbidity index score (CCI, a score that quantifies burden of disease and mortality risk), fever, and dyspnea (Table).
- Most deaths (63.4%) were attributed to older age groups relative to the reference group (18 – 49 years).
- Male sex contributed 12.3% (95% CI 5.8% – 19.1%), comorbidity burden contributed 6.5% (95% CI 6.3% – 6.6%) for CCI score of 5 or greater (Figure).
- Notable characteristics not significantly associated with mortality included obesity, Black race, Hispanic ethnicity, chronic obstructive pulmonary disease, hypertension, and smoking.
Methods: National cohort study of 88,747 veterans tested for SARS-CoV-2; 10,131 tested positive by RT-PCR from NP swabs between February 28 – May 14, 2020 and followed up through June 22, 2020. Outcomes were captured by ICD-10 codes in electronic health records. Limitations: ICD-10 codes might over- or underestimate outcomes; data describes primarily male veterans (91% male).
Implications: This national study of US veterans found that most deaths from SARS-CoV-2 occurred in older men who had comorbidities; deaths were not associated with obesity, hypertension, COPD, smoking, and race/ethnicity. This information is useful to identify veterans at risk for adverse outcomes of SARS-CoV-2 infection.
Figure:
Note: Adapted from Ioannou et al. Population attributable fraction of deaths due to various factors. Whisker bars represent the 95% CI. Licensed under CC-BY.
Table:
Note: Adapted from Ioannou et al. Sociodemographic medical factors association with mortality among 10,131 US veterans who tested positive for SARS-CoV-2. Licensed under CC-BY.
PEER-REVIEWED
Whole-genome sequencing to track SARS-CoV-2 transmission in nosocomial outbreaks.external icon Lucey et al. Clinical Infectious Diseases (September 19, 2020).
Key findings:
- 52 patients had hospital-acquired (HA) SARS-CoV-2 infection.
- Phylogenetic analysis identified six independent groups related to four outbreaks occurring in patients sharing a room, patients in private rooms, or patients who shared healthcare workers (HCWs).
- Study began before mask wearing was implemented; after mask implementation, HA cases decreased (Figure).
Methods: Study involving lab-confirmed SARS-CoV-2-infected patients and HCWs in a tertiary referral center in Ireland between March 7 and May 10, 2020. HA SARS-CoV-2 infections were defined as testing positive at least 7 days after admission. HCWs were tested by the hospital occupational health clinic based on symptoms. Whole genome sequencing and phylogenetic analysis were performed on SARS-CoV-2 RNA isolated from HCWs and patients to identify potential transmission linkages. Limitations: Only one location; may not be generalizable; only symptomatic HCWs were tested.
Implications: This study shows that in nosocomial infections, the majority of transmissions occurred through close contact and highlights the importance of consistent mask usage in both clinical and non-clinical areas.
Figure:
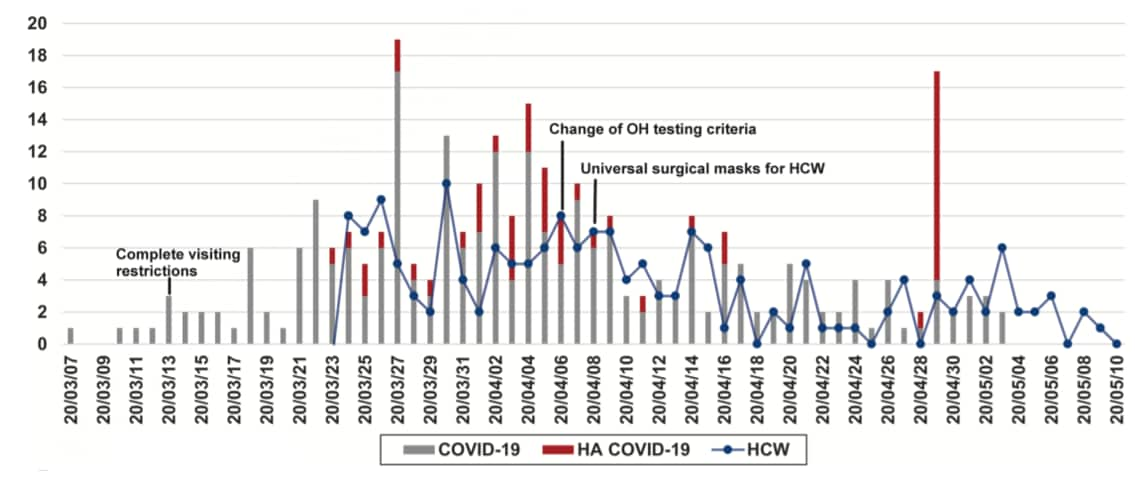
Note: From Lucey et al., Number of persons with community-acquired SARS-CoV-2, Hospital-acquired SARS-CoV-2 and healthcare worker COVID-19 cases. Dates are in YY/MM/DD. On April 6th, 2020 the Occupational Health clinic (OH) altered its criteria for SARS-CoV-2 testing of HCW. Reproduced by permission of Oxford University Press on behalf of the Infectious Diseases Society of America. Please visit: https://academic.oup.com/cid/advance-article/doi/10.1093/cid/ciaa1270/5898577external icon.
PREPRINTS (NOT PEER-REVIEWED)
Molecular architecture of early dissemination and massive second wave of the SARS-CoV-2 virus in a major metropolitan area.external icon Long et al. (September 25, 2020). Publishedexternal icon in mBio (October 30, 2020).
Key findings:
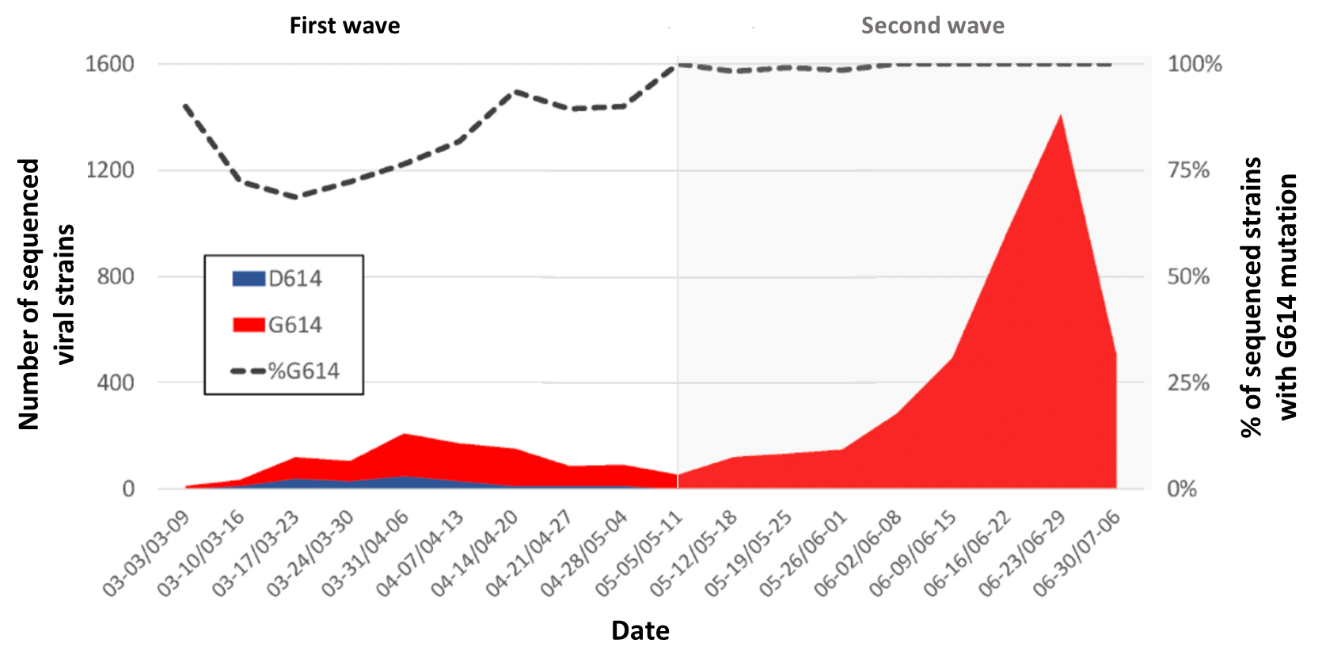
- Almost all of second wave virus strains were from 4 virus clades and 99.9% contained the G614 mutation in the spike protein compared to 82% from the first wave (Figure 1).
- Patients infected with the G614 variant strain had more viral RNA detected by RT-PCR as compared to the reference strain, D614 (Figure 2).
- There is no association between disease severity, length of stay, mortality, ethnicity and virus clades.
Methods: 5,085 full SARS-CoV-2 genomes were isolated from patients registered at Houston Methodist Hospitals and associated facilities between March and July, 2020. Genomic analysis was performed to identify phylogenetic differences. Limitations: Virus from ~10% of all infections included and might not be representative of the Houston Metropolitan Area; did not account for potential differences in timing of testing in relation to symptom onset that could have shifted over the period of study and be associated with detected viral RNA load.
Implications: The large and diverse set of genomic information for SARS-CoV-2 virus for the Houston Metro Area may provide insights into differences among virus variants, new infection spikes, viral transmission within the population, as well as the ability to analyze potential genomic changes and their relation to disease severity. The results support laboratory studiesexternal icon that found that persons with infection with the G614 variant strain had higher viral burden, reflected by lower Ct values from respiratory specimens.
Figure 1
Note: Adapted from Long et al., Number of D614 and G614 SARS-CoV-2 variants in the Houston Metro Area from March to July 2020. The percentage G614 strains from total sequenced strains is represented by the dashed line and the secondary Y-axis. The second wave of COVID19 cases are in the gray shaded area. Licensed under CC-BY-NC-ND 4.0.
Figure 2
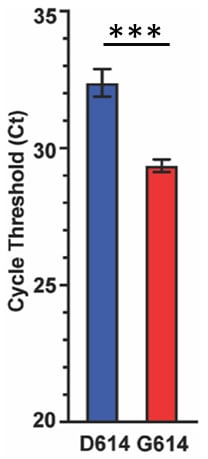
Note: Adapted from Long et al. Cycle thresholds for D614 and G614 in Houston. Lower Ct means greater viral RNA and more viral burden. Error bars are the standard error of the mean. Bars are statistically different, ***p-value <0.001. Licensed under CC-BY-NC-ND 4.0.
- Vogel, L. Have we misjudged the role of children in spreading COVID-19?external icon Canadian Medical Association Journal. Discusses new evidence that children can play a larger role in spreading SARS-COV-2 than previously considered.
- Bonow et al. Cardiology and COVID-19.external icon Review of the direct and indirect cardiac complications of COVID-19.
- Cevik et al. SARS-CoV-2 transmission dynamics should inform policy.external icon Clinical Infectious Diseases. Review of large-scale SARS-CoV-2 studies to provide guidance in developing policies to reduce the spread of COVID-19.
- Peiris et al. What can we expect from first-generation COVID-19 vaccines?external icon Review of the challenges around first-generation COVID-19 vaccines, including generating sterilizing immunity, duration of protection, and vaccine hesitancy.
- Kupferschmidt K WHO unveils global plan to fairly distribute COVID-19 vaccine, but challenges await.external icon Science. Challenges the WHO faces in distributing future COVID-19 vaccines outside of high-income countries.
- Han et al. Lessons learnt from easing COVID-19 restrictions: an analysis of countries and regions in Asia Pacific and Europe.external icon Health policy paper comparing several countries’ approaches to lift COVID-19 restrictions with recommendations.
- Wang et al. COVID-19 confirmed patients with negative antibodies results. external iconBMC infectious diseases and To et al. Serum antibody profile of a patient with COVID-19 reinfection. external iconClinical Infectious Diseases. Two articles describing patients where seroconversion was either partial or failed to occur.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.