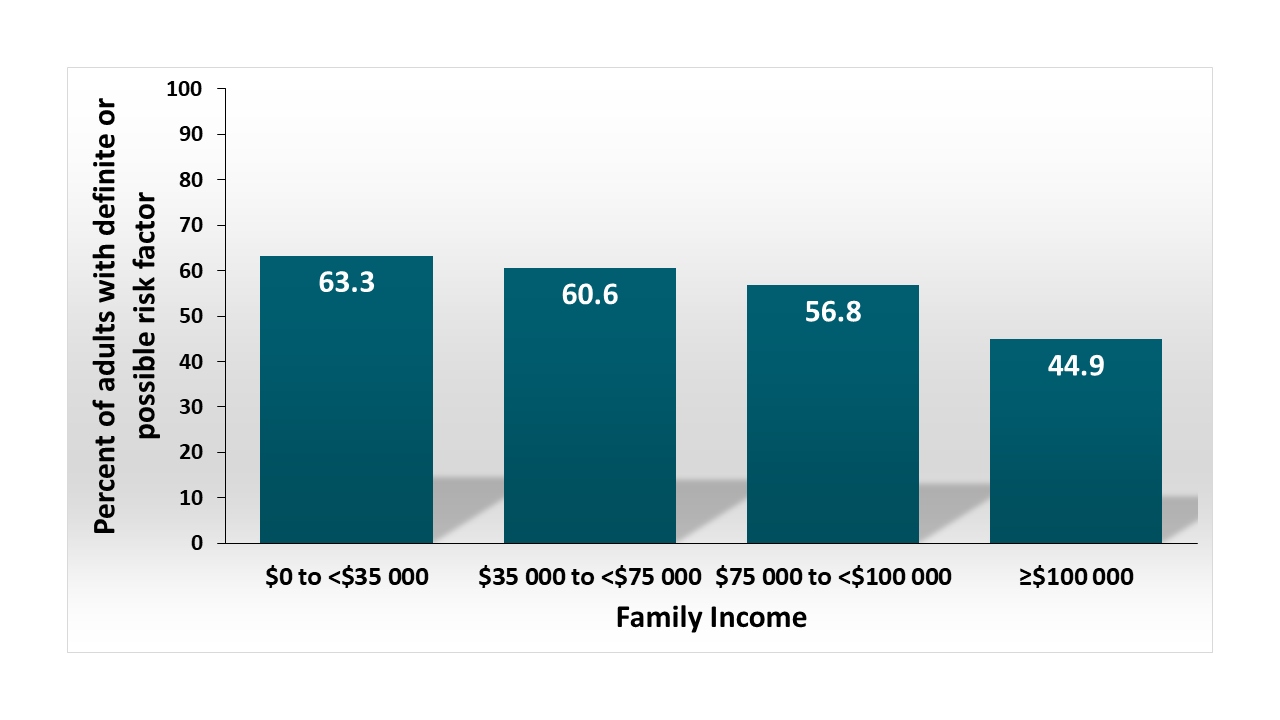COVID-19 Science Update released: September 1, 2020 Edition 44

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Here you can find all previous COVID-19 Science Updates.
SARS-CoV-2 may be transmitted during air travel and the effects of mitigation measures (e.g., seating distance, facemasks, excluding symptomatic passengers from boarding) are not established. Below, we summarize 2 evaluations of in-flight transmission of SARS-CoV-2.
PEER-REVIEWED
Assessment of SARS-CoV-2 transmission on an international flight and among a tourist group.external icon Hoehl et al. JAMA Network Open (August 18, 2020).
Key findings:
- Seven (29.2%) of a 24-member tour group tested positive for SARS-CoV-2, indicating they were infected before boarding the plane.
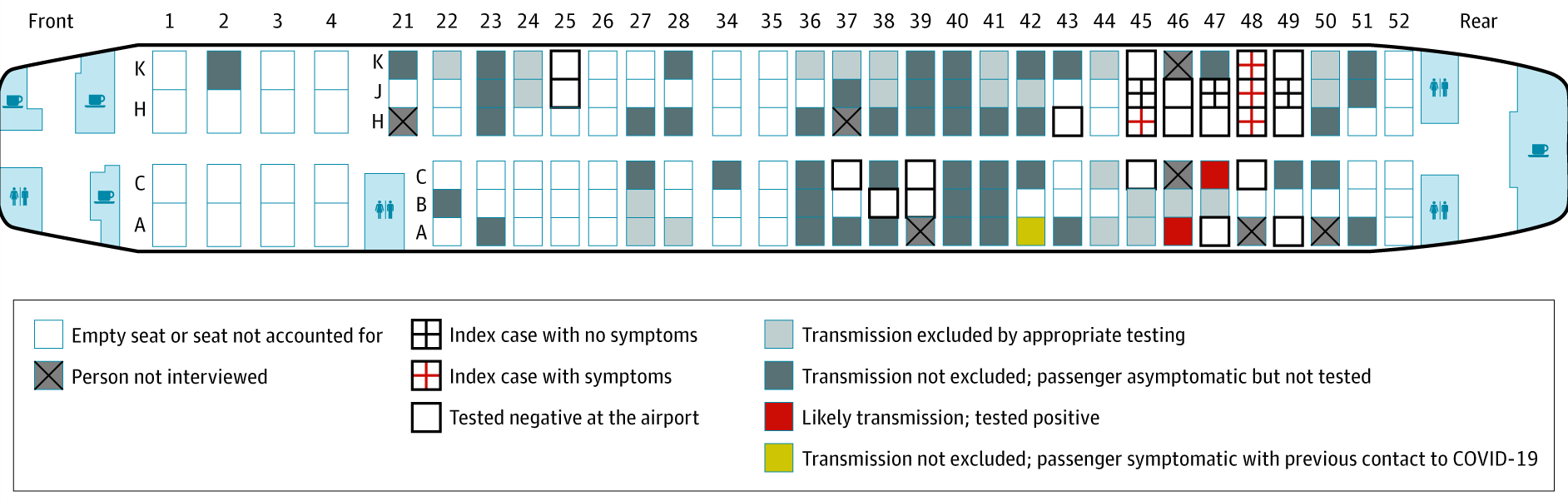
- 13 (16.7%) of 78 other passengers had a serology test done, 2 were positive and both sat within one row of a confirmed index case (Figure).
Methods: Because of a pre-flight exposure, a tour group (n = 24) was tested by NP swab RT-PCR upon arrival in Germany following a 4-hour 40-minute flight on March 9, 2020. IgG testing 6 to 9 weeks post-arrival was offered to passengers symptomatic within 14 days of arrival and those seated within 2 rows of a confirmed COVID-19 case. Limitations: Risk factors for transmission during flight were not assessed; flight crew and many passengers were not tested; exposure apart from that on the flight was not determined.
Figure:
Note: Adapted from Hoehl et al. Seating of index cases and other passengers on an international flight, including two passengers with positive serology for SARS-CoV-2. Licensed under CC-BY.
Asymptomatic transmission of SARS-CoV-2 on evacuation flight. Bae et al. Emerging Infectious Diseases (August 21, 2020).
Key findings:
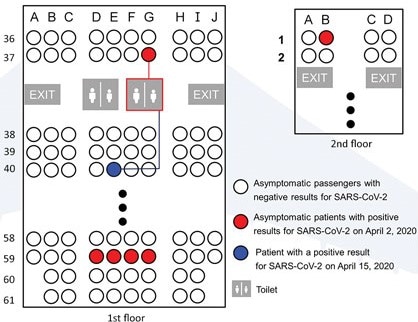
- 6 passengers tested positive for SARS-CoV-2 the day after flight arrival; none had symptoms, including during 14-day quarantine after arrival (Figure).
- One other passenger initially tested negative, developed symptoms eight days after arrival, and tested positive on Day 14.
- She wore an N95 respirator throughout the flight, except when using a toilet that had also been used by an asymptomatic patient seated 3 rows away (Figure 2).
- She quarantined alone for 3 weeks before and 2 weeks after the flight and did not use public transportation to get to the airport.
Methods: Korea Centers for Disease Control and Prevention implemented strict infection control procedures for an 11-hour flight on March 31, 2020, from Milan, Italy, to Incheon, South Korea, including denying boarding for 11 symptomatic passengers. N95 respirators were given to the 299 passengers before boarding. After the flight, passengers were immediately quarantined for 2 weeks, examined by medical staff twice daily for COVID-19 symptoms, and tested for SARS-CoV-2 by RT PCR on quarantine Day 1 (April 2) and Day 14 (April 15). Limitations: Not known if the asymptomatic passenger who likely transmitted this infection wore a mask when using the toilet.
Figure:
Note: Adapted from Bae et al. Seating of passengers on flight. Five asymptomatic patients tested positive on quarantine day 1 (April 2, 2020); one asymptomatic patient tested negative on quarantine day 1 and positive on quarantine day 14 (April 15, 2020). All other passengers tested negative on quarantine days 1 and 14. Open access journal; all content freely available.
Implications for both studies (Hoehl et al. & Bae et al.): SARS-CoV-2 can be transmitted among flight passengers even when index cases are asymptomatic and passengers use masks. Risk could potentially be decreased through physical distancing before boarding and after disembarking at airports, mask use including during toilet use, and hand hygiene. As noted in a recent editorial in The Lancetexternal icon, new rules such as mask requirements and physical distancing, reductions in passengers allowed on flights, increased cleaning of airport spaces, and other new or enhanced prevention measures introduced during the pandemic may become permanent to rebuild travelers’ confidence in air travel.
PEER-REVIEWED
Epidemiology of COVID-19 outbreak on cruise ship quarantined at Yokohama, Japan, February 2020. Expert Taskforce for the COVID-19 Cruise Ship Outbreak. Emerging Infectious Diseases (August 21, 2020).
Key findings:
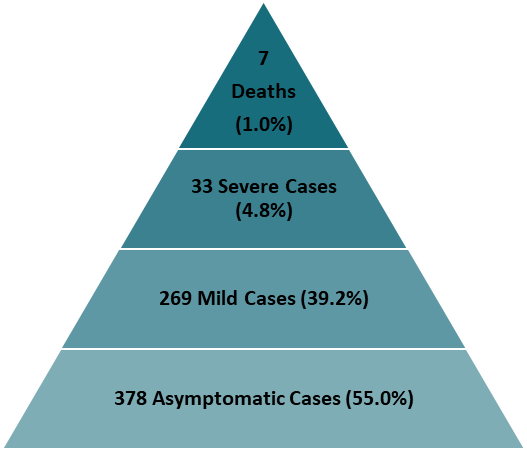
- Among 3,711 people aboard, 687 (18.5%) developed confirmed COVID-19; of these, 378 (55.0%) were asymptomatic (Figure 1).
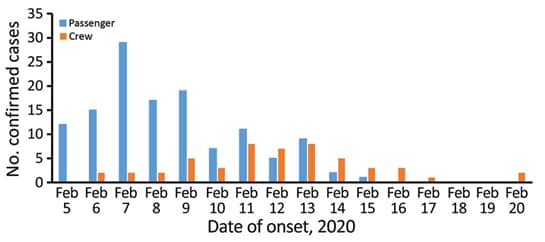
- Passenger incidence peaked February 7, 2020; 35 had onset before quarantine; crew incidence peaked February 11 to 13, 2020 (Figure 2).
- Infection rates were highest among passengers in 4-person cabins (30.0%; n = 18).
Methods: Following identification of a case of COVID-19 in a passenger, a cruise ship was quarantined and testing for SARS-CoV-2 by RT-PCR was conducted between February 5 and 24, 2020 for all 2,666 passengers and 1,045 crew members. Limitations: Disembarkment for health or family reasons, or due to country-assisted repatriation, reduced denominators over time; lab testing was initially limited to symptomatic cases and close contacts and symptoms were ascertained only at the time of testing.
Implications: The high proportion of asymptomatic cases in the face of testing all aboard suggests that substantial under-detection or underreporting of infections would occur without universal testing. These asymptomatic cases would likely challenge good infection control in a closed setting such as this, as well as pandemic control.
Figure 1
Note: Adapted from Expert Taskforce for the COVID-19 Cruise Ship Outbreak. Proportion of fatal, severe, mild, and asymptomatic COVID-19 cases among 544 passengers and 143 crew. Open access journal; all content freely available.
Note: Adapted from Expert Taskforce for the COVID-19 Cruise Ship Outbreak. COVID-19 cases aboard cruise ship February 5 to 20, 2020 by symptom onset date for the cases for which onset date was available for 127 passengers and 51 crew members. Open access journal; all content freely available.
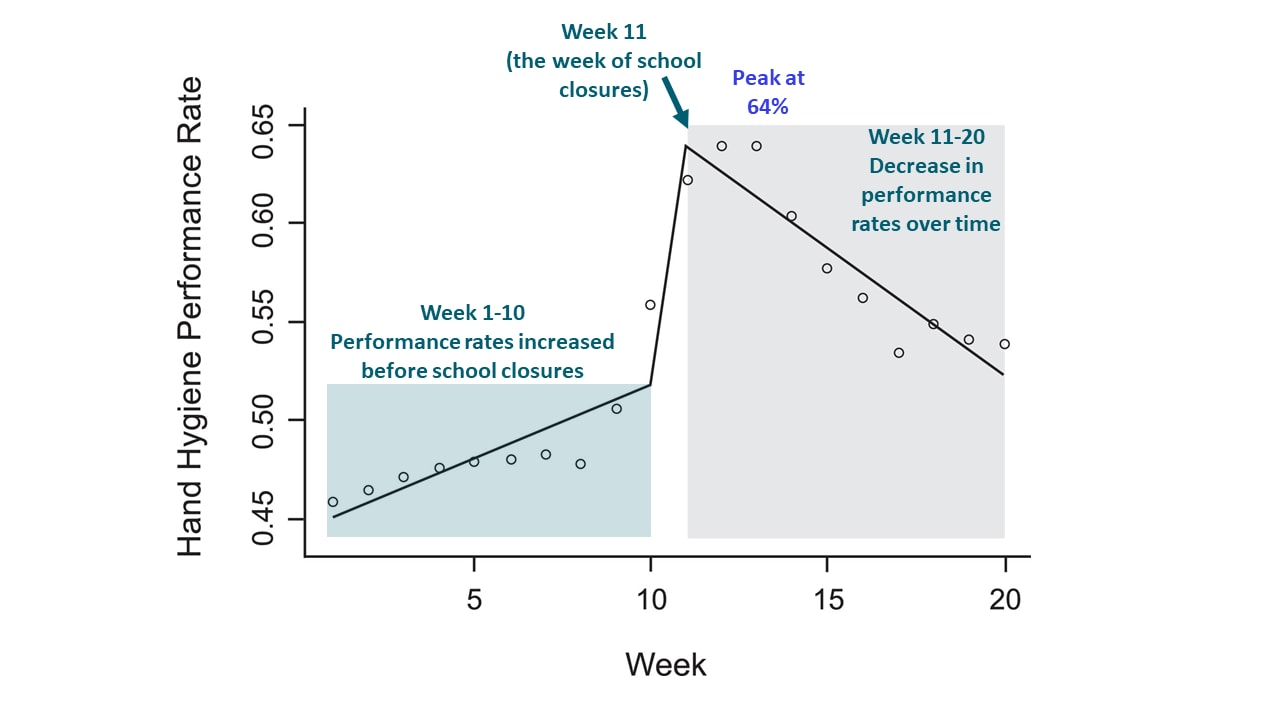
The impact of COVID-19 pandemic on hand hygiene performance in hospitalsexternal icon. Moore et al. American Journal of Infection Control (August 18, 2020).
Key findings:
- Average hospital hand hygiene performance (HHP) rates (Figure):
- Increased from 46% to 56% during 10 weeks before COVID-19-related school closures.
- Peaked at 64% at two weeks after school closures.
- Declined to 54% at 10 weeks after school closures.
Methods: HHP in 84 inpatient units (74 adult, 10 pediatric) of 9 US hospitals between January 5 and May 23, 2020 was measured to examine whether HHP changed among healthcare workers during a pandemic. HHP rate was defined as proportion of times a nearby hand sanitizing dispenser was used when a patient room door was used. Segmented regression model examined changes in weekly HHP rates during pre- and post-COVID-19-related school closure (during week of March 15). The week of school closure was selected as the midpoint for analysis for all hospitals as a proxy for community-level responses to the emerging pandemic. Limitations: Only inpatient units included; measure of HHP included both healthcare workers and visitors, which may represent different groups for intervention.
Implications: Low HHP rates may contribute to transmission, and in this case initial increases in HHP were not sustained. Efforts to improve and maintain hand hygiene compliance, particularly in healthcare settings, are urgently needed.
Figure:
Note: From Moore et al. HHP rates during 10 weeks preceding and 10 weeks following school closure (week of March 15, 2020), in 9 hospitals. Open circles represent observed HHP rate; line represents segmented regression modelled HHP. This article was published in American Journal of Infection Control, Moore et al., The impact of COVID-19 pandemic on hand hygiene performance in hospitals., Copyright Association of Professionals in Infection Control and Epidemiology 2020. This article is currently available at the Elsevier COVID-19 resource center: https://www.elsevier.com/connect/coronavirus-information-centerexternal icon.
Risk for severe COVID-19 illness among teachers and adults living with school-aged childrenexternal icon. Gaffney et al. Annals of Internal Medicine (August 21, 2020).
Key findings:
- Among teachers, 50.6% (estimated 3 million persons) had definite or possible risk factors for severe COVID-19 illness.
- Among adults living with school-aged children, 54.0% (estimated 38 million persons) had definite or possible risk factors.
- Proportion of adults living with children with risk factors was highest in lower income households (Figure).
- Adults living with Black children had 6% (95% CI 1.2-10.9) greater prevalence of risk factors than those living with White children.
Methods: Data from 5,278 persons surveyed in the 2018 National Health Interview Survey (NHIS) were used to determine prevalence of risk factors for severe COVID-19 among teachers and adults living with school-aged children. Risk factors for COVID-19 were defined as definite (cancer, serious heart condition, COPD, obesity, type 2 diabetes, age >64 years) and possible (asthma, cerebrovascular disease, hypertension, dementia, liver disease, pregnancy, smoking, type 1 diabetes). Limitations: NHIS data included only teachers and day care workers, not other school personnel; data did not include some severe COVID-19 risk factors (e.g., chronic kidney disease).
Implications: Decisions about when and how to reopen schools will need to weigh risks against benefits of COVID-19 for children and the adults who teach and care for them. While families would likely benefit educationally and socially from school re-opening, a great many have risks of severe COVID-19.
Figure:
Note: Adapted from Gaffney et al. Proportion of adults with risk factors for severe COVID-19 among household with school-aged children, by family income. From Annals of Internal Medicine, Gaffney et al., Risk for Severe COVID Illness Among Teachers and Adults Living With School-Aged Children, DOI: 10.7326/M20-5413, Copyright 2020 American College of Physicians. All Rights Reserved. Reprinted with the permission of American College of Physicians, Inc.
The issue of reinfection with SARS-CoV-2 is one that has received much attention and scrutiny. These case reports describing episodes of reinfection are based on timing of positive RT-PCR results and analysis of genetic distance of virus recovered from each of the episodes of SAR-CoV-2 infection.
PEER-REVIEWED
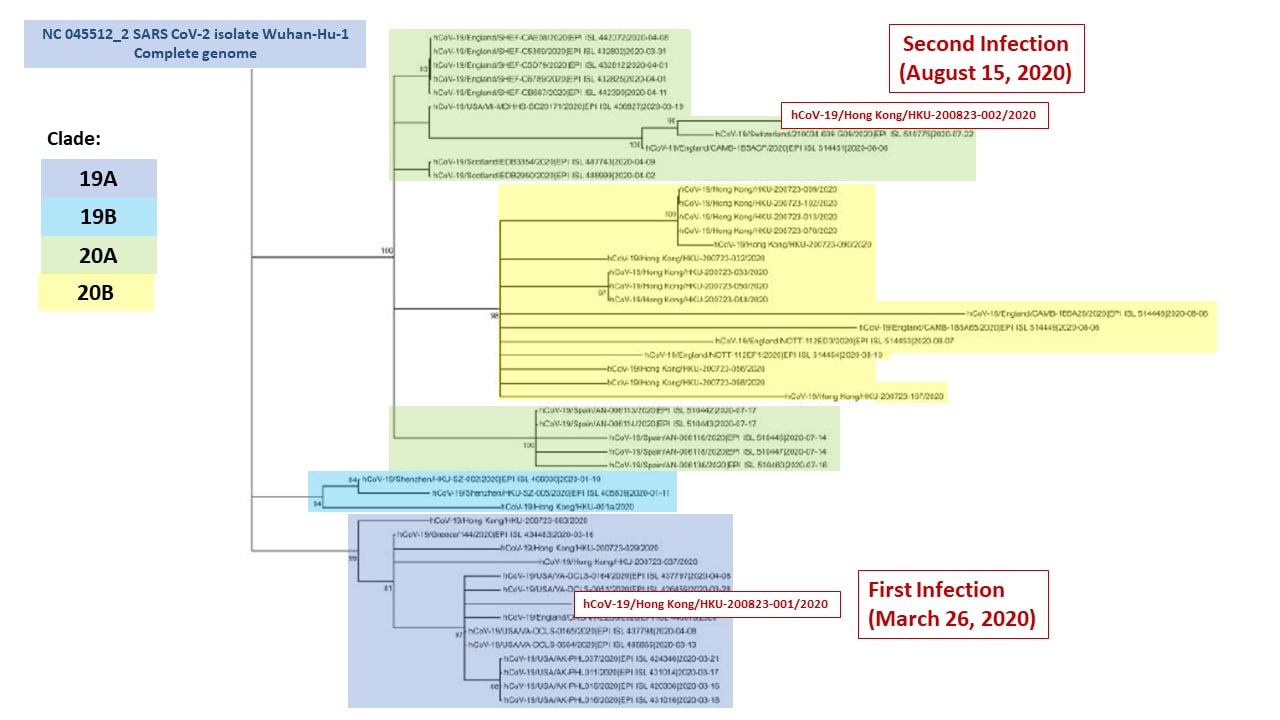
COVID-19 re-infection by a phylogenetically distinct SARS-coronavirus-2 strain confirmed by whole genome sequencingexternal icon. To et al. Clinical Infectious Diseases (August 25, 2020).
Key findings:
- One person had two episodes of SARS-CoV-2 infection that were detected >4 months apart.
- Symptoms during the first episode lasted for three days; the second episode was asymptomatic.
- Viral nucleic acid sampled during the two episodes was genetically distinct and the two viruses were in different clades (Figure).
- Serology after the first episode was negative but was positive 5 days after hospitalization for the second episode (Figure 1 and 2).
Methods: Case report of a 33-year old male in Hong Kong with symptoms and positive RT-PCR for SARS-CoV-2 on March 26, 2020 and re-infection August 15, 2020. SARS-CoV-2 was sequenced from respiratory specimens from both episodes to compare the virus. Limitations: Single case; multiple specimens were positive for SAR-CoV-2 in second episode but first diagnosis (first episode) was based on a single specimen.
Figure:
Note: Adapted from To et al. Phylogenetic analysis of whole SARS-CoV-2 genomes showing the relationship between the two strains from the patient from Hong Kong. Reproduced by permission of Oxford University Press on behalf of the Infectious Diseases Society of America. Please visit: https://academic.oup.com/cid/advance-article/doi/10.1093/cid/ciaa1275/5897019external icon
PREPRINTS (NOT PEER-REVIEWED)
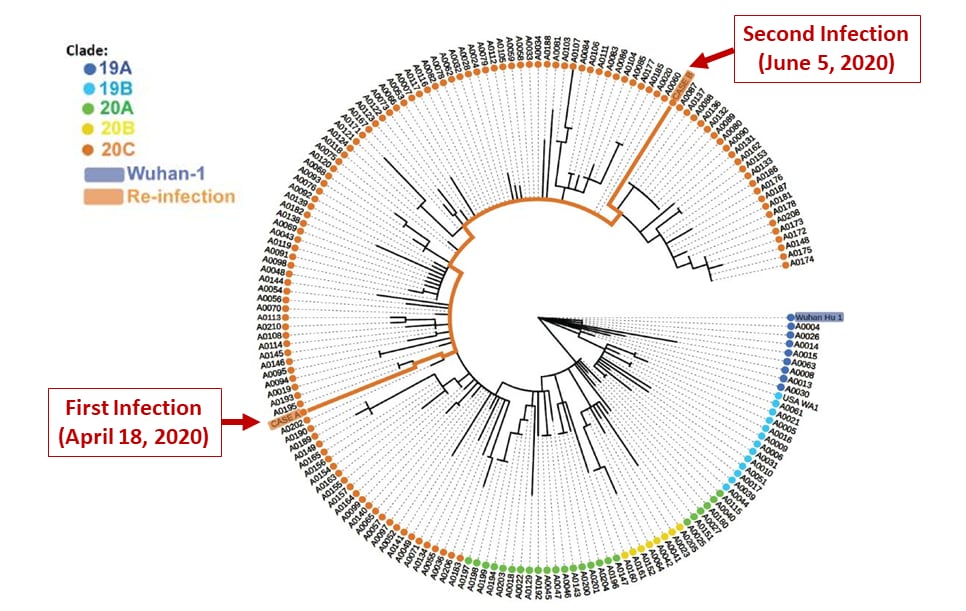
Genomic evidence for a case of reinfection with SARS-CoV-2external icon. Tillett et al. SSRN (August 27, 2020). Published in Lancet Infectious Diseases (October 12, 2020).
Key findings:
- In one person, two episodes of SARS-CoV-2 infection were detected <2 months apart.
- Symptoms during the first episode lasted for >30 days and the second episode required hospitalization for hypoxia.
- The viruses sampled during the two episodes were in the same clade but genetically distinct (Figure).
Methods: Case report of a 25-year old patient in Reno, NV with positive RT-PCR for SARS-CoV-2 on April 18, 2020 and symptoms from March 25 to April 27, 2020. More severe symptoms began on May 28, leading to hospitalization and retesting on June 5. SARS-CoV-2 was sequenced from NP swabs from both episodes. The patient was positive for IgG/IgM on June 6. Limitations: Single case; did not indicate whether serology was performed at the first episode; minor variant analysis not reported.
Figure:
Note: Adapted from Tillett et al. Phylogenetic placement within isolates and global clades of the viral sequences obtained from first and second episodes of illness, Nevada patient. Used by permission from authors and SSRN.
Implications for the two case reports (To et al. & Tillett et al.): There is growing evidence for instances of reinfection with SARS CoV-2. Consensus guidance for defining reinfection and more data are needed before drawing inferences for vaccination or epidemiological control measures.
PEER-REVIEWED
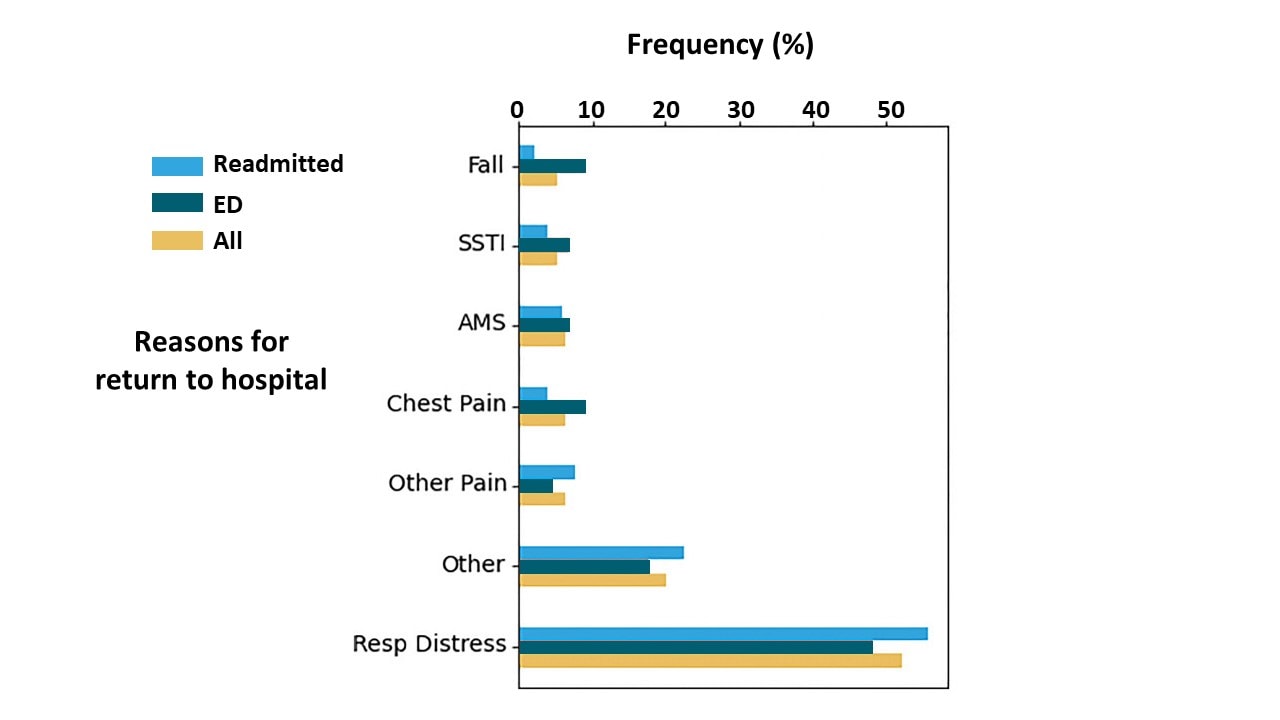
Characterization of patients who return to hospital following discharge from hospitalization for COVID-19external icon. Somani et al. Journal of General Internal Medicine (August 19, 2020).
Key findings:
- 103 (3.6%) of 2,864 patients discharged after COVID-19 treatment returned for emergency care 4.5 days (median) after discharge. Of those returning to care:
- Respiratory distress most common cause of repeat visit (50%) (Figure).
- 56 (54.4%) required readmission.
- 6 (10.8%) were readmitted to the ICU.
- 3 (5.4%) died.
- Returning patients, compared with those who did not return, had:
- Higher proportion of chronic obstructive pulmonary disease (6.8% vs 2.9%).
- Higher proportion of hypertension (36.0% vs 22.1%).
- Shorter median length of stay during initial hospitalization (4.5 [95% CI 2.9-9.1] vs 6.7 [95% CI 3.5-11.5] days; p = 0.006).
- Less frequent admission on initial hospitalization (6 of 103 [5.8%] vs 524 of 2,761 [19%]; p = 0.001).
Methods: Retrospective cohort study of patients with COVID-19 hospitalized in one of five New York City hospitals from February 27 to April 12, 2020. Assessment of those returning to the Emergency department or requiring readmission within 14 days of discharge. Limitations: Small sample size; missing data; readmission to hospitals outside studied hospital system may not have been captured.
Implications: Return rates after discharge for COVID-19 were relatively low but such patients may be quite ill. Discharge planning should specifically define conditions that should prompt early return to the ED.
Figure:
Note: Adapted from Somani et al. Reasons for return among all 103 COVID-19 patients who returned for emergency care, 47 discharged from the emergency department (ED) and 56 readmitted (inpatient) to the hospital. SSTI-skin/soft-tissue infections, AMS-altered mental status; Resp Distress-respiratory distress. Available via Nature Public Health Emergency Collection through PubMed Central.
Effect of remdesivir vs standard care on clinical status at 11 days in patients with moderate COVID-19. A randomized clinical trialexternal icon. Spinner et al. JAMA (August 21, 2020).
Key findings:
- Patients in treatment group receiving 5-day course of remdesivir were more likely to improve than those receiving standard care (OR 1.65, 95% CI 1.09-2.48).
- No difference in clinical status between 10-day remdesivir and standard care groups.
Methods: Randomized, open-label, multi-center trial of hospitalized patients with confirmed SARS-CoV-2 infection and moderate COVID-19 pneumonia assigned to three arms: 10-day course of remdesivir (n = 193), 5-day course of remdesivir (n = 191), standard care (n = 200). Primary end point was clinical status on Day 11. Relative odds of better clinical status among treatment groups were estimated. Limitations: Potential biases in patient care and reporting of data; 76% of the 5-day remdesivir group and 38% of the 10-day remdesivir group finished treatment; median length of treatment in the 10-day group was 6 days; no virologic outcome assessed; discharge decisions may be driven by factors other than clinical improvement; outcome scale not fully validated.
Implications: Compared with standard care, hospitalized COVID-19 patients with moderate disease experienced better clinical status after a 5-day course of remdesivir, whereas those receiving a 10-day course did not experience any difference in clinical status. Clinical importance of this finding is uncertain. McCreary and Angusexternal icon point out that lack of clinical improvement for 10-day course of remdesivir could be due to study design or that remdesivir is less efficacious than hoped.
PEER-REVIEWED
COVID-19 primarily presents as lung illness, yet virus may be present in other sites. Histopathology from post-mortem examination can improve understanding of pathophysiology and inform clinical treatments.
A. Histopathological findings and viral tropism in UK patients with severe fatal COVID-19: A post-mortem studyexternal icon. Hanley al. Lancet Microbe (August 20, 2020).
Key findings:
- Multi-system injury in ten patients with severe disease:
- 10/10 diffuse alveolar damage in lungs.
- 9/10 chronic bronchiolitis.
- 9/9 acute tubular injury in kidneys.
- 8/9 microthrombi in vessels.
- 7/8 CD8 T cell depletion in hilar lymph nodes.
- 5/5 microglial activation and mild T-cell infiltrate in brain.
- Unexpected findings included acute pancreatitis (2/9), adrenal micro-infarction (3/9), pericarditis (2/9).
- Virus was detected outside of the respiratory tract in 4/5 patients.
Methods: Case series of post-mortem examinations of 10 decedents with severe COVID-19, March 1 to April 30, 2020, London, UK. Patients were aged 22 to 97 years, 70% were men. Limitations: Few individuals examined.
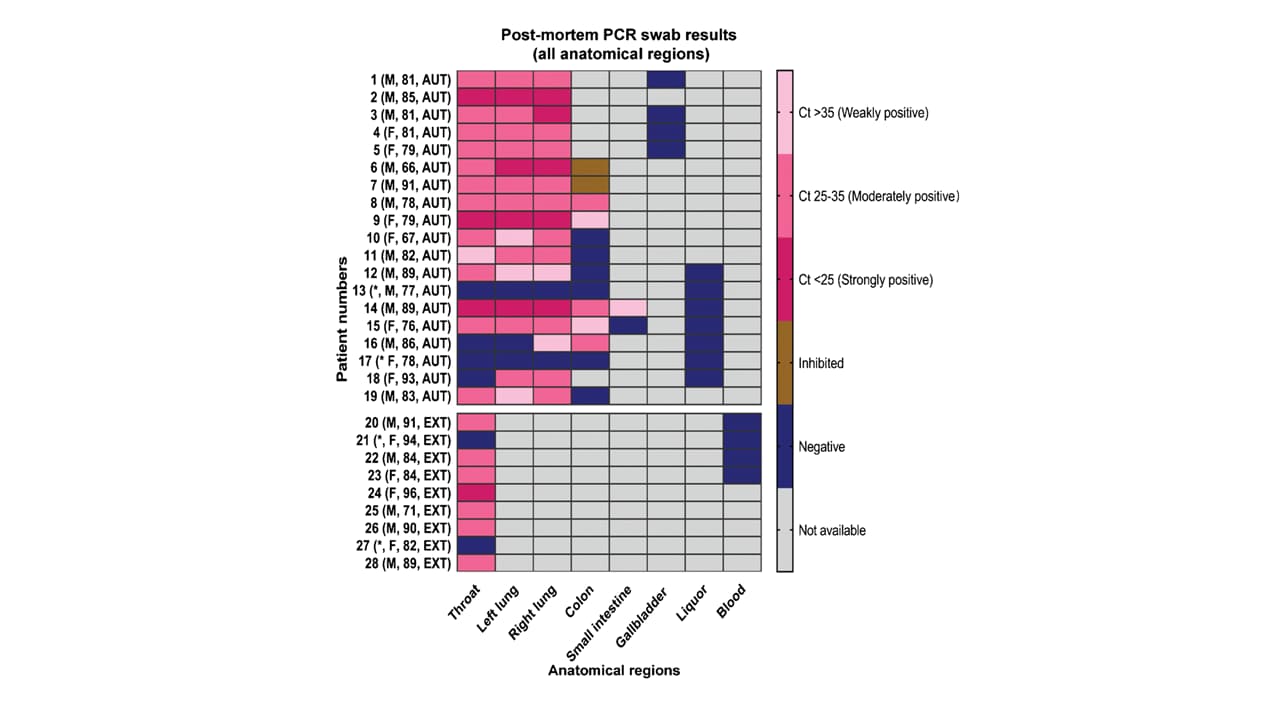
B. Post-mortem viral dynamics and tropism in COVID-19 patients in correlation with organ damageexternal icon. Skok et al. Virchows Archiv (August 20, 2020).
Key findings:
- The degree of organ damage seen did not correlate with levels of viral load estimated from Ct values.
- Throat swabs positive for SARS-CoV-2 up to 128 hours (>5 days) after death.
- Multiple organs positive for SARS-CoV-2; highest levels of virus seen in specimens from the respiratory system (Figure).
Methods: Post-mortem specimens for testing by RT-PCR were obtained from decedents with symptomatic, confirmed COVID-19 in Graz, Austria, March 24 to May 13, 2020. Time from symptom onset to death ranged from 4 to 36 days. The number of swabs obtained per person ranged from 1-10 before and after death. Limitations: Various organs tested for each autopsy; no viral culture; small numbers.
Figure:
Note: From Skok et al. RT-PCR results of collected samples as a heatmap. The parentheses denotes patient age and sex: M-male; F-female; AUT-autopsy; EXT-external examination; *Swab negative post-mortem on all tissues. In 2 colon swabs, the RT-PCR reaction was inhibited which means that the test was unable to produce valid results. Licensed under CC 4.0.
C. SARS-CoV-2 in cardiac tissue of a child with COVID-19-related multisystem inflammatory syndromeexternal icon. Dolhnikoff et al. Lancet Child & Adolescent Health (August 20, 2020).
Key findings:
- Virus was present in cardiac tissues by RT-PCR and electron microscopy at autopsy in a pediatric patient with multisystem inflammatory syndrome in children (MIS-C) and cardiac failure.
- DNA sequencing of the patient showed no primary immunodeficiency that could explain the disease outcome.
Methods: Case report of 11-year old female with MIS-C who developed cardiac failure and died, Sao Paulo, Brazil. Post-mortem electron microscopy, histology, RT-PCR for SARS-CoV-2 and patient’s DNA sequencing was performed. Limitations: Single case.
Implications for 3 studies (Hanley et al., Skok et al., & Dolhnikoff et al.): Although the lungs are the primary site of infection and injury, virus found outside of the lungs may be common in severe COVID-19 and be seen in all age groups. Injury as a result of tissue invasion or inflammatory response is likely.
COVID-19 and Flu Season
- Rubin R. What happens when COVID-19 collides with flu season?external icon JAMA. The importance of rapid diagnosis to distinguish between flu and COVID-19 for providing appropriate treatments and warning that coinfection could increase illness severity.
- Grohskopf et al. Addressing influenza vaccination disparities during the COVID-19 pandemicexternal icon. JAMA. lu vaccination rates in the US will need to dramatically increase in order to reduce the potential for severe co-infections with flu and SARS CoV-2, particularly among non-Hispanic Black, Hispanic, and American Indian/Alaskan Native adults.
Evidence Base for Treatments
- McCreary et al. Efficacy of Remdesivir in COVID-19. external iconJAMA Network Open. Limitations of the study design and analytic approach of Spinner et al.external icon on benefits of remdesivir for COVID-19 patients.
- Ledford H. Evidence lags behind excitement over blood plasma as a coronavirus treatment.external icon Nature. Concerns about FDA’s recent Emergency Use Approval include the need for rigorous research to confirm extent to which convalescent plasma improves clinical outcomes for COVID-19 patients, and if it does, at what dosage and stage of infection.
- Chaisemartin et al. BCG vaccination in infancy does not protect against COVID-19. Evidence from a natural experiment in Sweden.external icon Clinical Infectious Diseases. Study from Sweden showing that adults who received Bacille Calmette-Guérin (BCG) tuberculosis vaccine in infancy are not protected from COVID-19.
Other Topics
- Agarwal et al. Insurance coverage after job loss — The importance of the ACA during the COVID-associated recession.external icon NEJM. Unlike in prior economic downturns, very little association between job losses and health coverage has occurred in 2020, due to expanded options for health insurance unrelated to employment.
- McCarty et al. How do presenting symptoms and outcomes differ by race/ethnicity among hospitalized patients with COVID-19 infection? Experience in Massachusetts.external icon JAMA Network Open. In a retrospective analysis of adult patients in 9 hospitals with lab-confirmed COVID-19, no differences based on race/ethnicity were found in hospitalization outcomes, including in-hospital mortality.
- Bilinski et al. Modeling contact tracing strategies for COVID-19 in the context of relaxed physical distancing measures.external icon JAMA Network Open. Effectiveness of contact tracing to maintain low transmission rates for SARS-CoV-2 depends on the percent of symptomatic cases and asymptomatic contacts identified, and adherence to isolation and quarantine among those traced.
- Editorial. Air travel in the time of COVID-19external icon. Lancet. Measures to minimize the chances of SARS CoV-2 infection and to rebuild travelers’ confidence in air travel may become normalized, akin to the increased security measures introduced after September 11, 2001.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.