COVID-19 Science Update released: May 28, 2021 Edition 91

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Section headings in the COVID-19 Science Update align with the CDC Science Agenda for COVID-19.
Section headings in the COVID-19 Science Update have been changed to align
with the CDC Science Agenda for COVID-19.
Here you can find all previous COVID-19 Science Updates.
PEER-REVIEWED
SARS-CoV-2 seroprevalence in a strictly-Orthodox Jewish community in the UK: A retrospective cohort studyexternal icon. Gaskell et al. Lancet Regional Health Europe (May 18, 2021).
Key findings:
- SARS-CoV-2 seroprevalence in an Orthodox Jewish community in the UK was 64.3% (95% CI 61.6%-67.0%), >5 times higher than the estimated national seroprevalence.
- Seroprevalence was 27.6% in children <5 years old, rising to 73.8% in secondary school students, and remaining high through all ages (Figure).
Methods: A cross-sectional serosurvey of 343 households in the UK between late October and early December 2020 to determine the seroprevalence of SARS-CoV-2, stratified by age and sex, and factors associated with transmission. Limitations: Only 38% of households approached were included; participants who provided serum samples were older than those who did not.
Implications: Interventions are needed to reduce the disproportionate burden of SARS-CoV-2 in some religious and ethnic minority populations, especially those characterized by high density and frequent communal gatherings.
Figure:
Note: from Gaskell et al. Age-specific seroprevalence in female and male participants (n = 1,242). Age cohorts along x-axis and proportion IgG positive for any one of 3 SARS CoV-2 antigens (trimeric spike, receptor binding domain, or nucleocapsid) on the y-axis. Licensed under CC BY.
Patient care and clinical outcomes for patients with COVID-19 infection admitted to African high-care or intensive care units (ACCCOS): A multicentre, prospective, observational cohort study.external icon ACCCOS Investigators. Lancet (May 22, 2021).
Key findings:
- Access to intensive medical care was lower than needed to manage critically ill COVID-19 patients in 10 African countries:
- 45% of patients referred for critical care were not admitted.
- Mortality following admission to critical care facilities for COVID-19 was 48.2% (95% CI 46.4%-50.0%) compared with 31.5% (95% CI 27.5%-35.5%) globally.
- Delay in admission due to a shortage of resources (OR: 2.14, 95% CI 1.42–3.22) was associated with mortality.
Methods: 30-day follow-up of 3,140 adults hospitalized in 64 intensive- or high-care units for COVID-19 in Egypt, Ethiopia, Ghana, Kenya, Libya, Malawi, Mozambique, Niger, Nigeria, and South Africa from May to December 2020. 57 hospitals provided additional information on hospital characteristics and intervention availability. Limitations: Data unavailable for lower-tier hospitals and referred patients who were not admitted.
Implications: Patient outcomes will likely continue to be compromised until critical care resource scarcity is addressed.
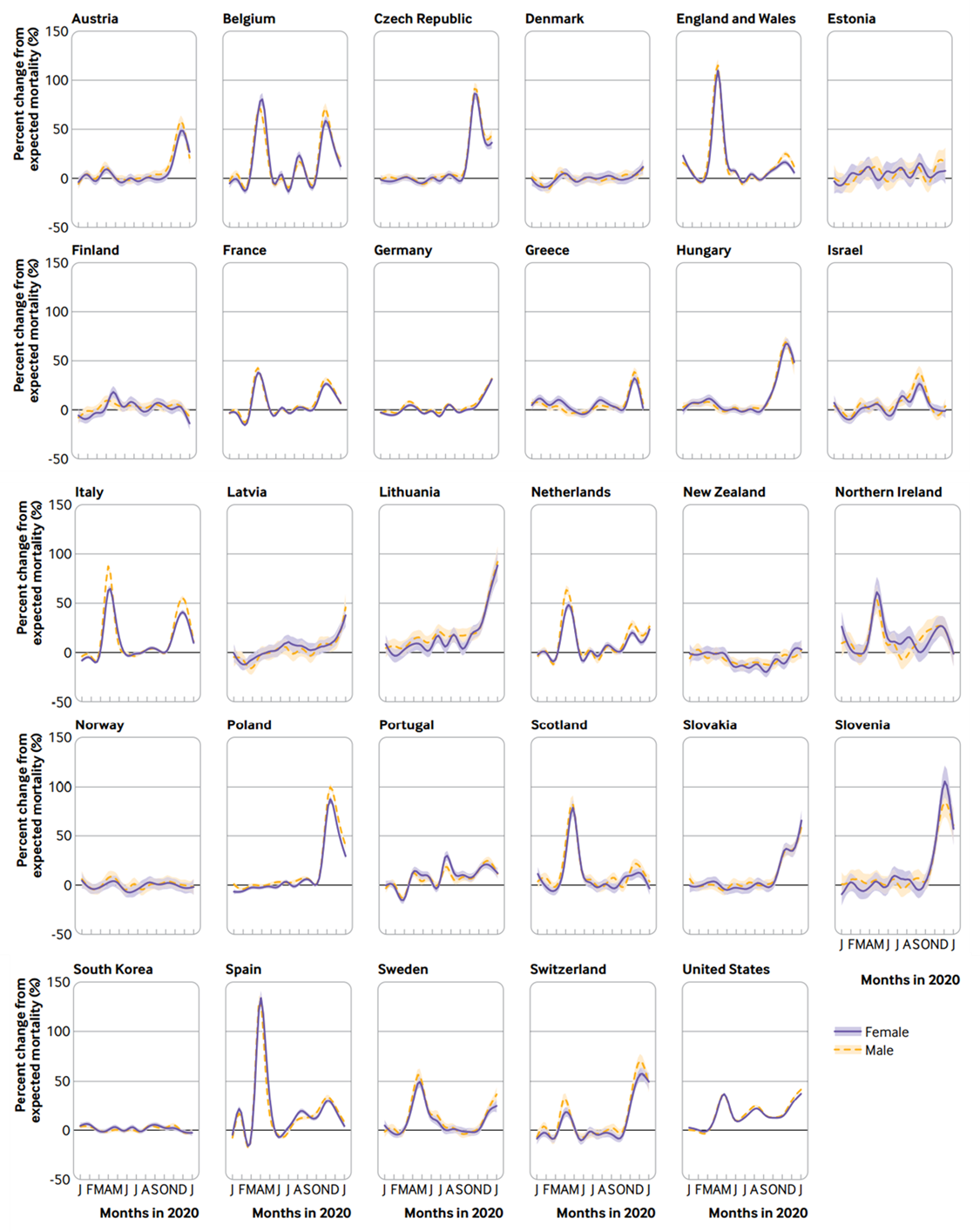
Excess deaths associated with covid-19 pandemic in 2020: Age and sex disaggregated time series analysis in 29 high income countriesexternal icon. Islam et al. BMJ (May 19, 2021).
Key findings:
- 979,000 (95% CI 954,000-1,001,000) excess deaths occurred in 2020 in 29 high-income countries:
- The United States had the highest absolute number of excess deaths (approximately 458,000).
- Most countries reported 1 or more major waves of excess deaths that occurred at different time frames over the pandemic (Figure).
Methods: Time series study of mortality in 29 high income, Organization for Economic Cooperation and Development member countries. Excess deaths were calculated as the difference between the number of deaths from all causes that occurred during 2020 and the expected number of deaths based on historical data from the Human Mortality Database 2016-2020. Limitations: Ethnic and socioeconomic differences in mortality could not be studied; possible effects of policy measures implemented in 2020 could not evaluated.
Implications: Data describing excess deaths could inform healthcare policy decisions; increasing the capacity to collect those data might be of particular value to middle- and lower-income countries where such data are sparse.
Figure:
Note: Adapted from Islam et al. Weekly percentage change from expected mortality, modeled using smooth function of time, in 29 high income countries in 2020, all ages, for females and males. Permission request in progress.
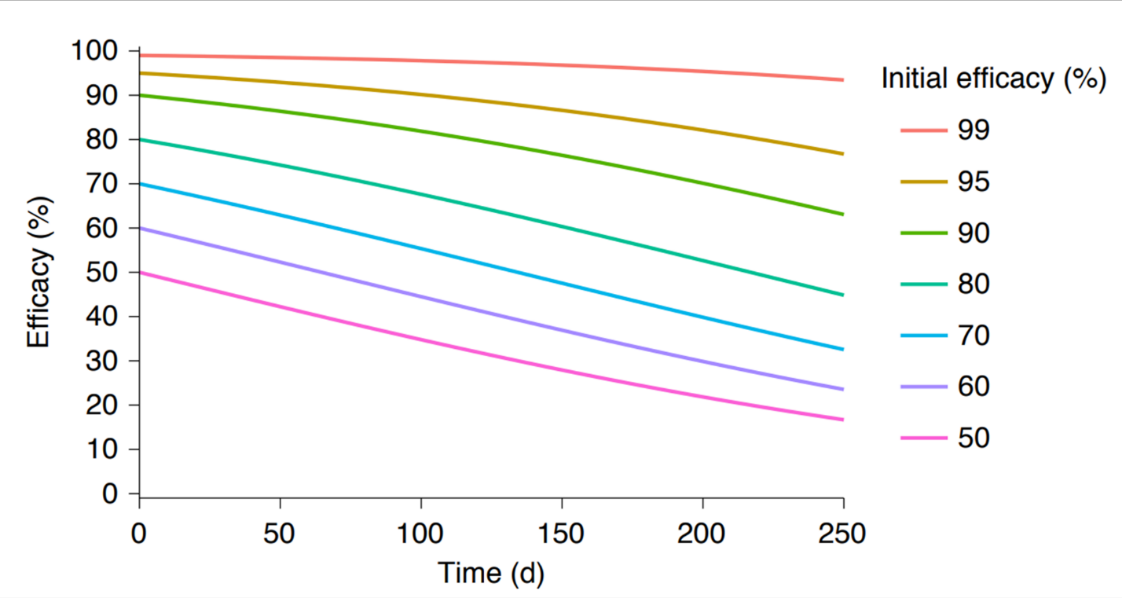
Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infectionexternal icon. Khoury et al. Nature Medicine (May 17, 2021).
Key findings:
- Neutralizing antibody levels correlated with clinical efficacy of 7 different vaccines.
- Long-term protective efficacy is predicted to decline more rapidly for vaccines with initial efficacy <90% compared with vaccines with an initial efficacy >90% (Figure).
- Vaccines with >90% efficacy against wild-type SARS-CoV-2 are predicted to have good efficacy against variants with 10-fold lower neutralization.
Methods: Data from trials of 7 vaccines and from 1 convalescent cohort were used to model correlation of neutralizing antibody level with protection from SARS-CoV-2 infection. Modeled vaccine efficacy over time and against variants with reported lower neutralization. Limitations: Model assumes that waning of neutralization titer over time is the same for different vaccines and that the decay in titer is the same regardless of the initial starting titer.
Implications: Models show neutralization titers are highly predictive of immune protection from vaccination. Immune protection from vaccination might wane as neutralization levels decline, and booster immunization might be required within a year, particularly for vaccines with low initial efficacy.
Figure:
Note: Adapted from Khoury et al. Predicted decline in efficacy for vaccines with different levels of initial efficacy (50%-99%) 250 days (d) after final vaccine dose. Reprinted by permission from Springer Nature Customer Service Centre GmbH: Springer Nature. Nature Medicine, Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection, Khoury et al., COPYRIGHT 2021.
PEER-REVIEWED
COVID-19 outbreaks following full reopening of primary and secondary schools in England: Cross-sectional national surveillance, November 2020external icon. Aiano et al. The Lancet Regional Health Europe (May 18, 2021).
Key findings:
- 969 primary and secondary schools reported COVID-19 outbreaks in the first half of the autumn 2020 term, representing 2% of primary schools and 10% of secondary schools.
- There were 2,314 cases in the 179 schools with outbreaks that provided information on cases:
- Teachers were more likely to be index cases in primary school outbreaks (48/100, 48%) than in secondary school outbreaks (25/79, 32%) (p = 0.027).
- Attack rates in secondary school students (1.20%; 95% CI 1.13%-1.28%) were higher than in primary school students (0.84%; 95% CI 0.75%-0.94%).
- Attack rates in staff members (5.07%; 95% CI 4.75%-5.41%) were almost five times higher than in students (1.09%; 95% CI 1.04%-1.15%) at both primary and secondary schools.
Methods: Cross-sectional investigation of COVID-19 outbreaks occurring in primary and secondary schools in England among 18,943 primary schools, 5,409 secondary schools, and 1,231 special schools that reopened. Schools that reported an outbreak (≥2 laboratory-confirmed cases within 14 days) to Public Health England between August 31 and October 18, 2020 were contacted in November 2020 to complete an online questionnaire. Limitations: 47% of contacted schools did not participate in the online questionnaire.
Implications: Higher attack rates among teaching staff during an outbreak underscores the importance of maintaining low community infection rates for reducing the risk of virus introduction into educational settings.
PEER-REVIEWED
Factors associated with readmission in the US following hospitalization with COVID-19external icon. Verna et al. Clinical Infectious Diseases (May 20, 2021).
Key findings:
- 3.6% of patients were readmitted within 30 days of first hospitalization with COVID-19.
- In-hospital mortality on readmission was 12.3% and associated with need for maximum oxygen support, sepsis, acute kidney injury, and age.
- Risk of re-admission was significantly associated with:
- Older age.
- Comorbidities such as diabetes, chronic kidney disease and cardiovascular disease.
- Being from the Northeast or having Medicare or Medicaid insurance.
Methods: Retrospective cohort study on readmission within 30 days and in-hospital mortality among 29,659 US-based patients ≥18 years old following first hospitalization with COVID-19 between February 15 and June 9, 2020. Limitations: Regional differences might reflect higher hospitalization rates in the Northeast early in the pandemic.
Implications: To reduce readmissions among COVID-19 patients, targeted strategies are needed for older adults and those with high-risk underlying medical conditions.
PEER-REVIEWED
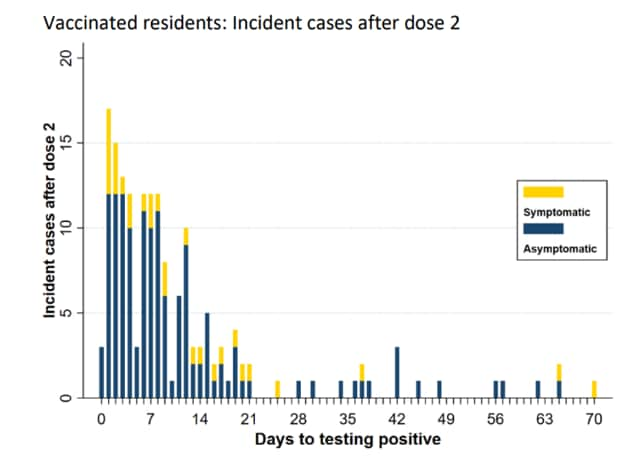
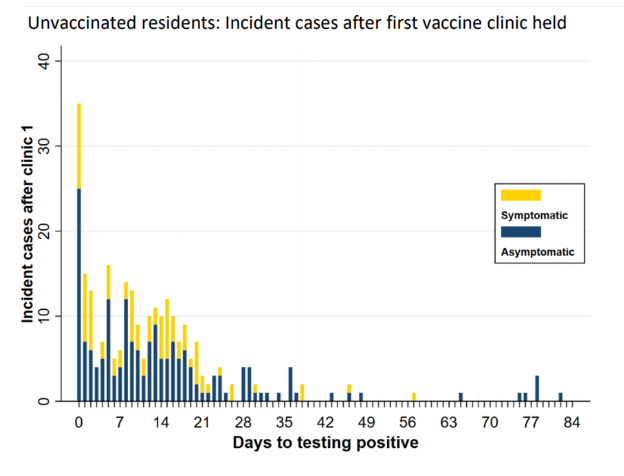
Incident SARS-CoV-2 infection among mRNA-vaccinated and unvaccinated nursing home residents. external iconWhite et al. NEJM (May 19, 2021).
Key findings:
- Incidence of SARS-CoV-2 infection decreased among all residents within 42 days of first vaccination clinic (Figure).
- Among residents receiving 1 vaccine dose, SARS-CoV-2 incidence fell from 4.5% up to 14 days after vaccination to 1.4% at >14 days post-vaccination.
- Among residents receiving both vaccine doses, incidence fell from 1.0% up to 14 days after the second dose to 0.3% at >14 days post vaccination.
- Among unvaccinated residents, incidence fell from 4.3% up to 14 days after the vaccination clinic to 0.3% at >42 days after the vaccination clinic.
Methods: Electronic health record data were used to determine the incidence of SARS-CoV-2 infection through March 31, 2021 among vaccinated and unvaccinated residents of 280 nursing homes in 21 states. As of February 15, 2021, 18,242 residents received at least one dose of mRNA vaccine and 13,048 received the second dose. 3,990 residents were unvaccinated. Residents were tested every 3 to 7 days if there were confirmed cases in the facility or if they had any new symptoms or potential exposure. Limitations: Local area infection rates might affect observed incidence; no report of the vaccination status of the nursing home staff.
Implications: SARS-CoV-2 vaccination might provide protection against infection for vaccinated residents that extends to unvaccinated nursing home residents as well.
Figures:
A:
B:
Note: Adapted from White et al. Incident SARS-CoV-2 cases by days since first vaccine clinic for nursing home residents who were vaccinated with 2 doses (A) and unvaccinated residents (B) showing the number of symptomatic or asymptomatic incident cases. From the New England Journal of Medicine, White et al., Incident SARS-CoV-2 infection among mRNA-vaccinated and unvaccinated nursing home residents. May 19, 2021, online ahead of print. Copyright © 2021 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
The effectiveness of the two-dose BNT162b2 vaccine: Analysis of real-world data.external icon Chodick et al. Clinical Infectious Diseases (May 17, 2021).
Key findings:
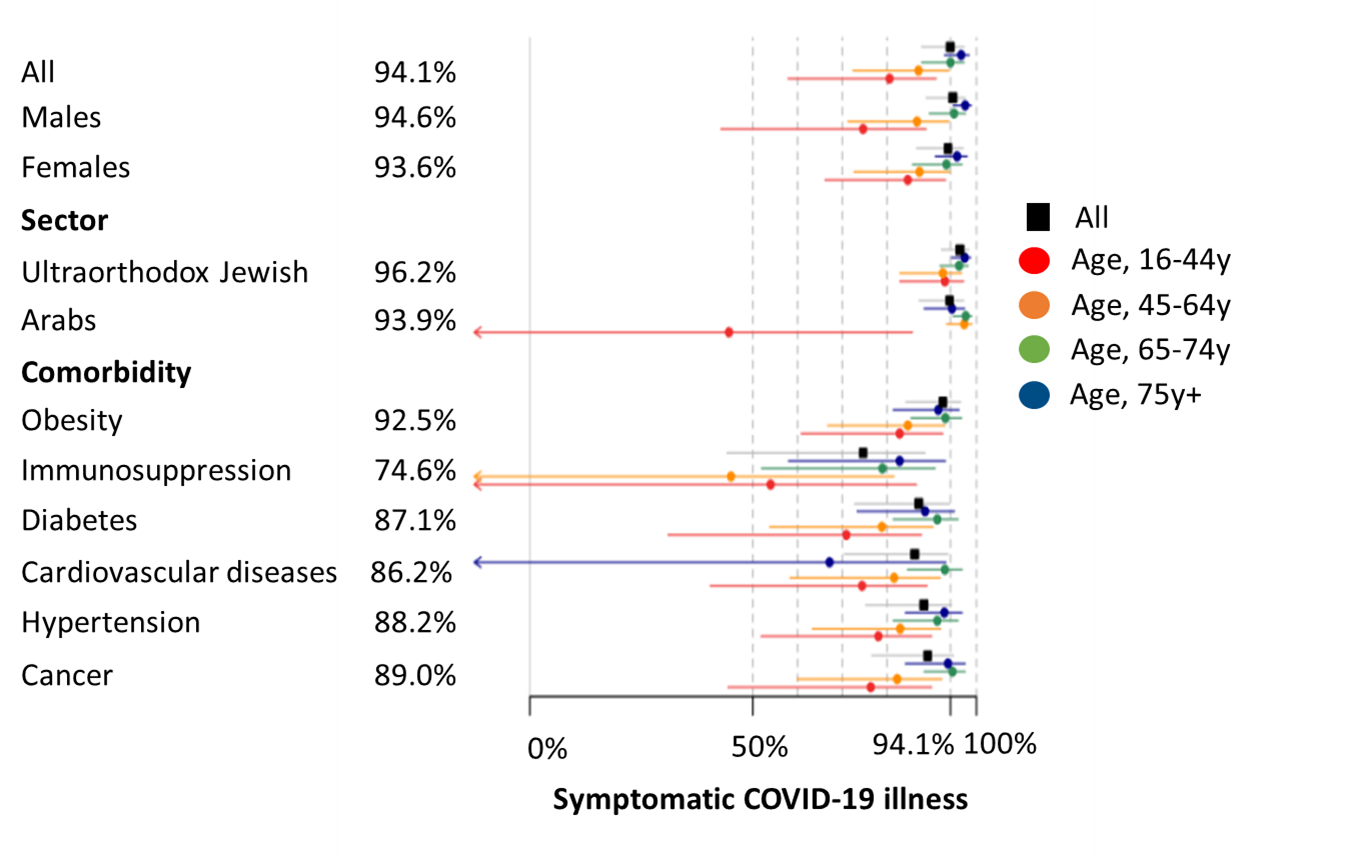
- Pfizer/BioNTech BNT162b2 vaccine was 90% (95% CI 79%-95%) effective in preventing SARS-CoV-2 infection and 94% (95% CI 88%-97%) effective in preventing symptomatic COVID-19 illness (Figure).
- Effectiveness was lower in people who were older, immunosuppressed, or had diabetes, cardiovascular disease, cancer or hypertension (Figure).
Methods: Retrospective cohort study evaluating Pfizer/BioNTech BNT162b2 vaccine effectiveness against SARS-CoV-2 infection, hospitalization and death among 1,178,597 individuals aged 16 years and older vaccinated between December 19, 2020 and February 20, 2021 in Israel. SARS-CoV-2 incidence (RT-PCR confirmed) 7-27 days after second dose was compared to a reference period (1-7 days after first dose). Limitations: Although tests were freely available, not all individuals received testing, potentially allowing some infections to be undetected.
Implications: Findings affirm the effectiveness of Pfizer/BioNTech BNT162b2 vaccine against infection and COVID-19 disease but indicate less protection for individuals who are older, immunocompromised, and with certain comorbidities.
Figure:
Note: Adapted from Chodick et al. Point estimates (squares and circles) and 95% CIs (lines) for vaccine effectiveness by age, sex, population sector, and comorbidities. Chodick et al., The effectiveness of the two-dose BNT162b2 vaccine: Analysis of real-world data. Clinical Infectious Diseases, published online May 17, 2021. By permission of the Infectious Diseases Society of America.
PREPRINTS (NOT PEER-REVIEWED)
Durability of mRNA-1273-induced antibodies against SARS-CoV-2 variants.external icon Pegu et al. bioRxiv (May 16, 2021). Published in Scienceexternal icon (August 13, 2021).
Key findings:
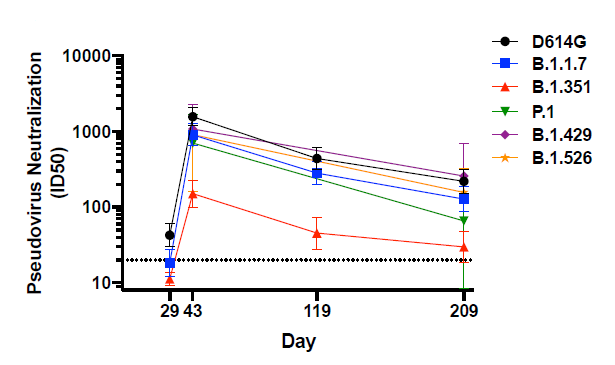
- Most people fully vaccinated with Moderna mRNA-1273 maintained neutralizing (Figure) and binding antibodies against 5 SARS-CoV-2 variants for at least 6 months.
- By 6 months post-vaccination, ≥85% of persons still had neutralizing antibodies against all variants except for B.1.351, for which only 54% of persons still had neutralizing antibodies.
Methods: Vaccine-induced antibody responses to 5 SARS-CoV-2 variants (B.1.1.7, B.1.351, P.1, B.1.429, and B.1.526) were assessed over 7 months in 24 subjects who received 2 vaccine doses. Limitations: Small sample size; correlates of vaccine-induced protection are unknown.
Implications: The Moderna mRNA-1273 vaccine induces relatively long-lived functional antibodies, able to neutralize 5 circulating SARS-CoV-2 variants; however, it induces a less robust, shorter-lived response to B.1.351.
Figure:
Note: Adapted from Pegu et al. Neutralizing antibody titers for 5 SARS-CoV-2 variants (B.1.1.7, B.1.351, P.1, B.1.429, B.1.526 and D614G [predominant circulating virus as of summer 2020], n = 24) sampled at 4 timepoints after vaccination. Pseudovirus neutralization, expressed as 50% inhibitory dilution (ID50). Dotted line, limit of detection (>20). U.S. Government work not subject to copyright.
Fatal breakthrough infection after anti-BCMA CAR-T therapy highlights suboptimal immune response to SARS-CoV-2 vaccination in myeloma patientsexternal icon. Aleman et al. medRxiv (May 15, 2021).
Key findings:
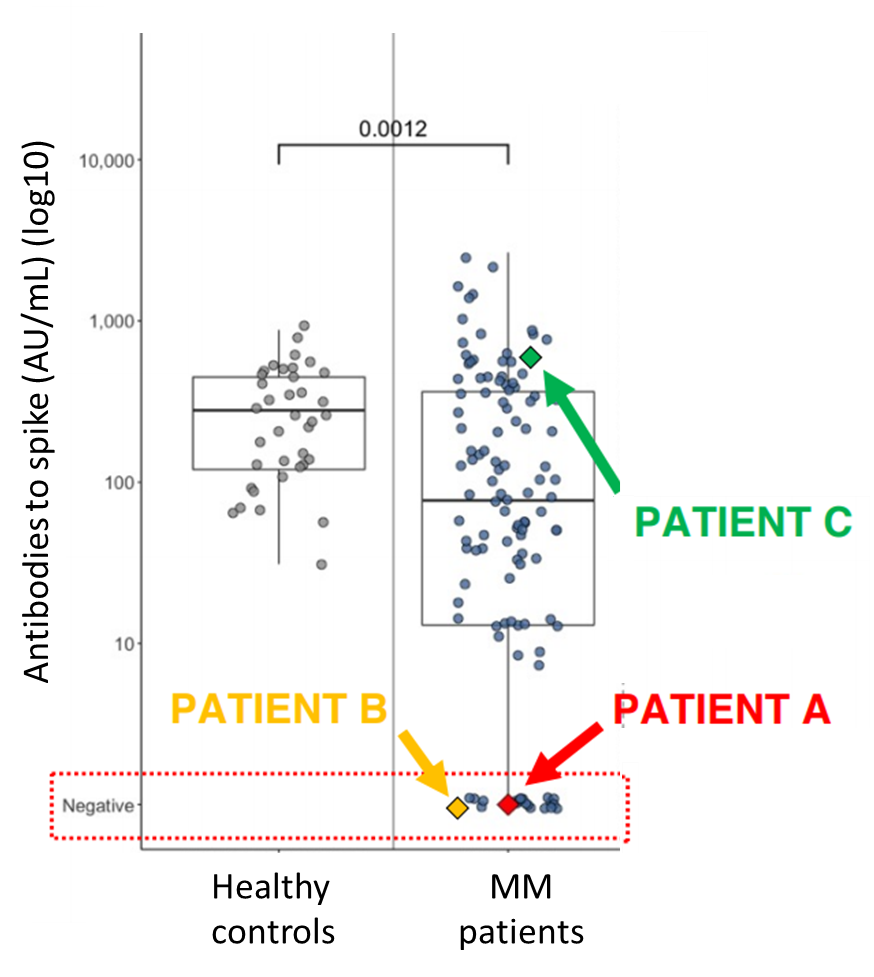
- A multiple myeloma (MM) patient developed fatal SARS-CoV-2 infection 2 weeks after receiving the second dose of the Pfizer/BioNTech BNT162b2 vaccine.
- Patient did not develop SARS-CoV-2-specific antibodies, or B or T cell responses post-vaccination.
- 15.9% of fully vaccinated MM patients did not develop SARS-CoV-2 antibodies (Figure).
- Patients receiving treatment targeting the BCMA protein on cancer cells had a higher risk for failing to develop antibodies (OR 32.0; 95% CI 4.2-360.1) following vaccination compared with controls.
- The case patient–Patient A–with fatal breakthrough infection was vaccinated 3 months after anti-BCMA CAR-T therapy (Figure).
- An MM patient on a different therapy–Patient Balso developed low antibody responses (Figure).
- In contrast to Patient A, Patient C, who was vaccinated 18 months after BCMA CAR-T therapy, developed robust antibody responses (Figure).
Methods: Case report of SARS-CoV-2 infection and death following second dose of the Pfizer/BioNTech vaccine in an MM patient, ≥3 months after B-cell maturation antigen (BCMA)-targeted chimeric antigen receptor (CAR)-T cell therapy. Antibody responses were evaluated in an additional 139 MM patients >10 days after second mRNA vaccine dose and compared to age-matched controls. Limitations: Possible overrepresentation of patients on BCMA-targeted treatments.
Implications: Given the relatively high percentage of vaccine non-responders, and potential for severe outcomes from COVID-19, there might be a need to monitor vaccine response in this immunosuppressed population. Additionally, timing of vaccination in relation to MM treatment might need to be evaluated.
Figure:
Note: Adapted from Aleman et al. Anti-spike IgG titers among healthy controls (n = 37) and multiple myeloma patients with no previous history of COVID-19 (n = 118), >10 days after second dose of mRNA vaccine. Case patient A (with fatal breakthrough infection), patient B on other MM therapy, and patient C vaccinated 18 months after anti-BCMA CAR-T therapy denoted by diamonds. Permission request in progress.
Detection, Burden, and Impact
- Fernández-de-Las-Peñas et al. Published in iScience as Partial resistance of SARS-CoV-2 Delta variants to vaccine-elicited antibodies and convalescent sera (November 19, 2021).external icon Journal of Infection (May 11, 2021). Of 1,142 patients diagnosed with COVID-19 in Spain from March 10 to May 31, 2020, 81.4% had ≥1 post (mean 7 months) COVID-19 symptom. Fatigue (60.8%), hair loss (26.3%), shortness of breath (23.5%), and memory loss (19.0%) were the most common symptoms.
- Contreras et al. Published in iScience as Partial resistance of SARS-CoV-2 Delta variants to vaccine-elicited antibodies and convalescent sera (November 19, 2021).external icon Emerging Infectious Diseases (May 12, 2021). Nearly 60% of 698 nonhealthcare worksite COVID-19 outbreaks occurred in 3 sectors: manufacturing (26.4%), retail trade (19.6%), and transportation and warehousing (10.5%). Outbreaks were larger and longer in facilities with more on-site staff.
- Augustin et al. Published in iScience as Partial resistance of SARS-CoV-2 Delta variants to vaccine-elicited antibodies and convalescent sera (November 19, 2021).external icon The Lancet Regional Health Europe (May 17, 2021). Between April 6 and December 2, 2020, symptoms such as shortness of breath or loss of taste and smell were observed in 27.8% (123/442) of non-hospitalized persons in Germany with confirmed SARS-CoV-2 infection 4 months after diagnosis and in 34.8% (123/353) 7 months after diagnosis.
Natural History of SARS-CoV-2 Infection
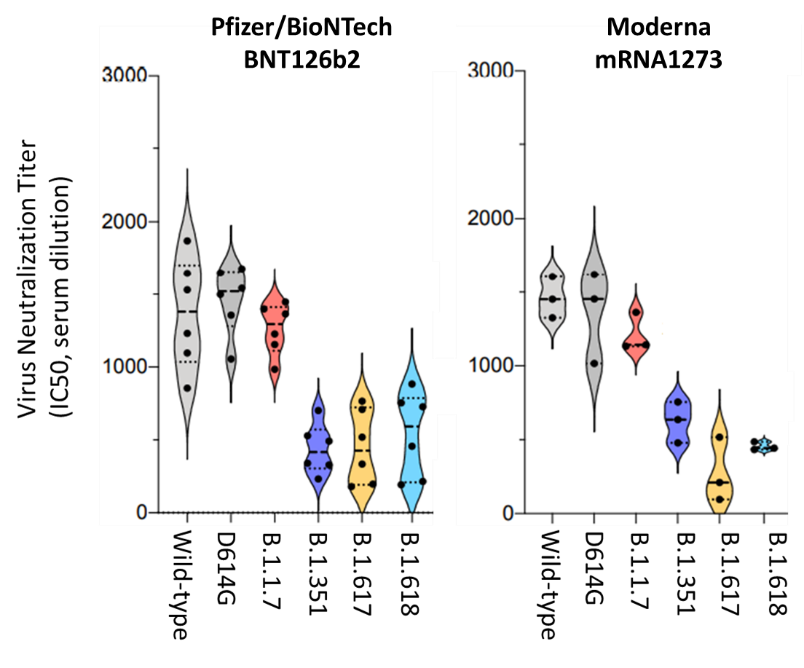
- Tada et al. The spike proteins of SARS-CoV-2 B.1.617 and B.1.618 variants identified in India provide partial resistance to vaccine-elicited and therapeutic monoclonal antibodies.external icon bioRxiv (Preprint; May 16, 2021). Published in iScience as Partial resistance of SARS-CoV-2 Delta variants to vaccine-elicited antibodies and convalescent seraexternal icon (November 19, 2021). Sera from mRNA-vaccinated individuals less efficiently neutralized SARS-CoV-2 B.1.617 and B.1.618 compared with wild-type SARS-CoV-2, D614G, or B.1.1.7; neutralizing titers to B.1.617 and B.1.618 were closer to those seen with variant B.1.351.
Note: adapted from Tada et al. Neutralizing antibody titers in sera from people vaccinated with mRNA vaccines against wild-type SARS-CoV-2 and the strains D614G, B.1.1.7, B.1.351, B.1.617, and B.1.618. Long-dashed lines show median titers, dotted lines show 25% and 75% range, and dots represent individual values; n = 6 for Pfizer/BioNTech and n = 3 for Moderna vaccines. IC50- half-maximal inhibitory concentration. Used by permission of authors.
Prevention, Mitigation, and Intervention Strategies
- Formeister et al. Preliminary analysis of association between COVID-19 vaccination and sudden hearing loss using US Centers for Disease Control and Prevention vaccine adverse events reporting system data.external icon JAMA Otolaryngology – Head and Neck Surgery (May 20, 2021). There was no association between vaccination with mRNA vaccines and incident sudden hearing loss based on data reported to the Vaccine Adverse Event Reporting System between December 14, 2020 and March 2, 2021.
- Shroff et al. Immune responses to COVID-19 mRNA vaccines in patients with solid tumors on active, immunosuppressive cancer therapyexternal icon. medRxiv (Preprint; May 14, 2021). Published in Nature Medicine as Immune responses to two and three doses of the BNT162b2 mRNA vaccine in adults with solid tumorsexternal icon (September 30, 2021). SARS-CoV-2 virus neutralization titers were lower in 52 cancer patients on active immunosuppressive therapy compared with 50 controls. Sera from 20% of cancer patients had no virus neutralization activity after the second dose of vaccine.
Note: Adapted from Shroff et al. Virus neutralization activity of sera from control (n = 50) and cancer (n = 52) cohorts following one or two doses of the Pfizer/BioNTech BNT126b2 vaccine. Solid lines connect data from the same individual after the first and second dose of vaccine. Numbers represent median neutralizing titers for each cohort. Licensed under CC-BY-NC-ND 4.0.
- Simon et al. Haemodialysis patients show a highly diminished antibody response after COVID-19 mRNA vaccination compared to healthy controls.external icon Nephrology Dialysis Transplantation (May 17, 2021). 21 days after the second dose of Pfizer/BioNTech BNT126b2 vaccine, neutralizing antibody (Nab) titers were 14.6-fold lower in 81 hemodialysis patients compared with 80 healthy controls. 22 hemodialysis patients did not have detectable NAb at 3 weeks post-vaccination, but at 10 weeks 5 of these patients (among 21 with samples) had detectable NAb.
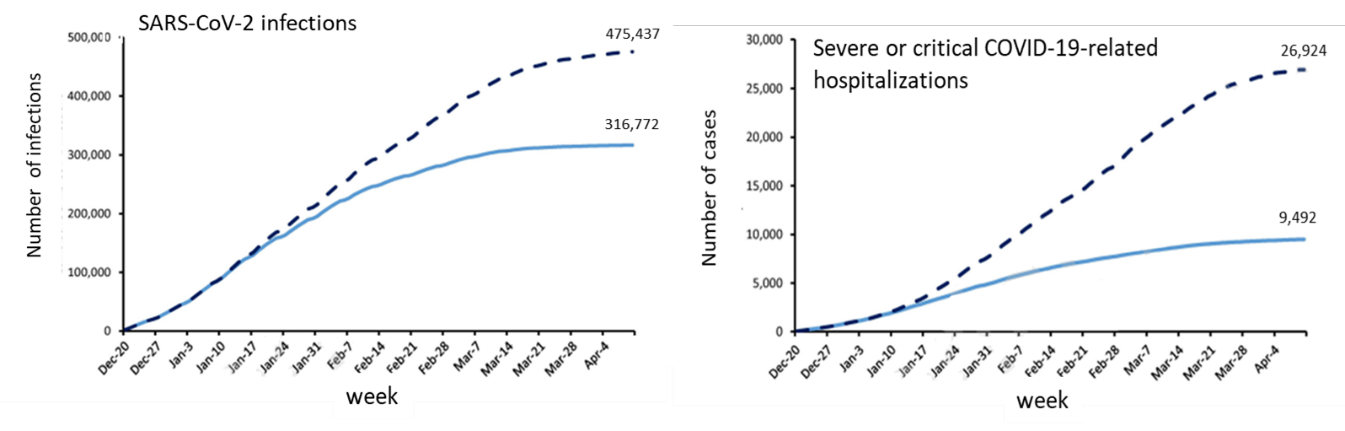
- Haas et al. Infections, hospitalizations, and deaths averted via direct effects of the Pfizer-BioNTech BNT162b2 mRNA COVID-19 vaccine in a nationwide vaccination campaign, Israel.external icon SSRN (Preprint; May 13, 2021). Published in The Lancet Infectious Diseases as Infections, hospitalisations, and deaths averted via a nationwide vaccination campaign using the Pfizer–BioNTech BNT162b2 mRNA COVID-19 vaccine in Israel: a retrospective surveillance studyexternal icon (September 22, 2021). National surveillance data from December 20, 2020 to April 10, 2021 showed that vaccination averted an estimated 158,665 (95% uncertainty range [UR] 115,899‒201,431) SARS-CoV-2 infections, 24,597 (95% UR 6,622‒42,571) hospitalizations, 17,432 (95% UR 3,065‒31,799) severe and critical hospitalizations, and 5,533 (95% UR -1,146‒12,213) deaths (note that for deaths, uncertainty range includes 0).
Note: adapted from Haas et al. Cumulative number of SARS-CoV-2 outcomes with vaccination (solid line) compared with estimated rates without vaccination (dashed line). Permission request in progress.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.