COVID-19 Science Update released: April 14, 2020 Edition 4

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Here you can find all previous COVID-19 Science Updates.
PEER-REVIEWED
Effectiveness of surgical and cotton masks in blocking SARS–CoV-2: A controlled comparison in 4 patients.external icon Bae et al. Annals of Internal Medicine (Apr 6, 2020).
RETRACTED by Journal on June 1, 2020.
PEER-REVIEWED
Impact of school closures for COVID-19 on the US healthcare workforce and net mortality: A modelling study.external icon Bayham et al. Lancet Public Health (April 3, 2020).
Key findings:
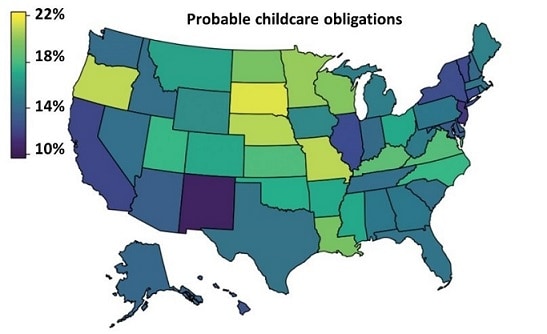
- 15% of US healthcare workers (HCWs) have unmet childcare needs due to school closures (Figure 1)
- Certain specialties (e.g., nurse practitioners, physician’s assistants, and diagnostic technologists/technicians) have greatest unmet childcare needs.
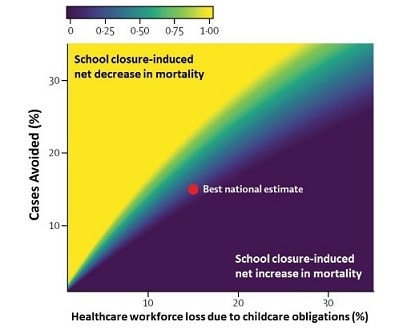
- Modelers predicted that school closures might increase net COVID-19 mortality if mortality rates increase from 2% to 2.3% and HCW with unmet childcare needs cannot go to work (Figure 2).
Methods: Mathematical model to explore how school closures and associated childcare burden could affect national COVID-19 net mortality. Assumptions: Model assumed childcare remains available for children 0–2 years. Limitations: Mortality estimates were based on limited empirical evidence; relatively small adjustments in parameters might lead to different conclusions.
Implications: Offering childcare resources for HCWs might help offset potential negative effects of school closures (e.g., mortality), particularly in states with greatest HCW childcare needs.
Figure 1.
Note: Adapted from Bayham et al. Figure shows estimated percentage of HCWs with unmet childcare needs by state (for children aged 3–12 years). Licensed under CC-BY-NC-ND.
Figure 2.
Note: Adapted from Bayham et al. The colors indicate the extent to which school closures are estimated to decrease (yellow) or increase (blue) net mortality, according to the level of workforce loss. The red point indicates the best national estimate of how school closures will affect the percentage of cases avoided (y-axis) and the percentage of HCW labor force lost due to unmet childcare needs. Licensed under CC-BY-NC-ND.
PEER-REVIEWED
SARS-CoV-2 is not detectable in the vaginal fluid of women with severe COVID-19 infection. external iconQiu et al. Clinical Infectious Diseases (April 2, 2020).
Key findings:
- Vaginal fluid of post-menopausal women does not appear to contain detectable SARS-CoV-2 RNA.
Methods: Vaginal fluid samples from 10 post-menopausal women aged 52 to 80 years with severe COVID-19 pneumonia were tested for SARS-CoV-2 RNA by RT-PCR. Vaginal swab samples were collected 17 to 40 days after COVID-19 diagnosis. Limitations: Samples only from post-menopausal women; long period between diagnosis and testing.
Implications: Sexual transmission of SARS-CoV-2 from post-menopausal women appears highly unlikely.
PREPRINTS (NOT PEER-REVIEWED)
Outbreak of COVID-19 in Germany resulting from a single travel-associated primary caseexternal icon. Böhmer et al. SSRN (Mar 31, 2020). Published as Investigation of a COVID-19 outbreak in Germany resulting from a single travel-associated primary case: a case seriesexternal icon. Lancet Infectious Diseases (May 15, 2020).
Key findings:
- A visitor from China with COVID-19 initiated a chain of transmission over four transmission generations in Germany; median incubation period was 4 days (range: 1–7 days).
- 8 of 16 diagnosed were infected during time that the source was pre-symptomatic or during the very earliest onset of symptoms.
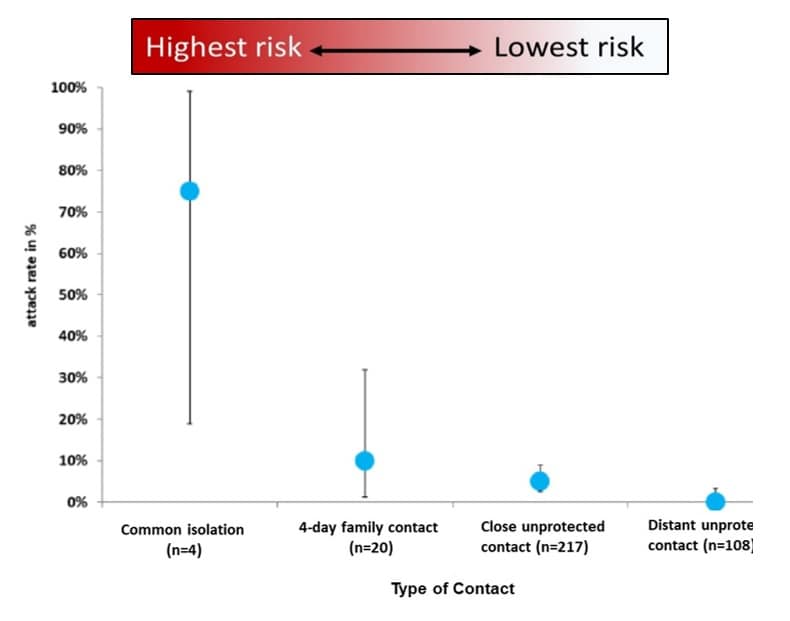
- Attack rates were highest in family clusters and diminished with decreasing contact (Figure).
Methods: Outbreak investigation with contact tracing and interviews in Bavaria, Germany. Cases were confirmed by RT-PCR; whole genome sequencing verified epidemiological links. Researchers calculated attack rates among monitored contacts. Limitations: Focused exclusively on human-to-human and not indirect (e.g., fomite) transmission.
Implications: Contact tracing can provide insights into human-to-human chains of SARS-CoV-2 transmission. Half of transmission events likely occurred before, or on the day of, symptom onset. Attack rates decreased with decreasing contact.
Figure:
Note: Adapted from Böhmer et al. Error bars represent 95% CIs. High-risk contacts included: face-to-face interaction with a COVID-19 patient (15 min, cumulative); direct contact with COVID-19 patient’s bodily fluid; interaction within 2-meters of a COVID-19 patient without PPE (healthcare worker). All other contacts were considered low risk. Used by permission from authors and SSRN.
PEER-REVIEWED
Baseline characteristics and outcomes of 1,591 patients infected with SARS-CoV-2 admitted to ICUs of the Lombardy region, Italyexternal icon. Grasselli et al. JAMA (April 6, 2020)
Key findings:
- Most ICU patients with COVID-19 were male (82%, no age effect), 68% had ≥ 1 comorbidity, including hypertension (49%) and cardiovascular disease (21%).
- Nearly all (99%) of COVID-19 patients in the ICU required respiratory support, including mechanical ventilation (88%).
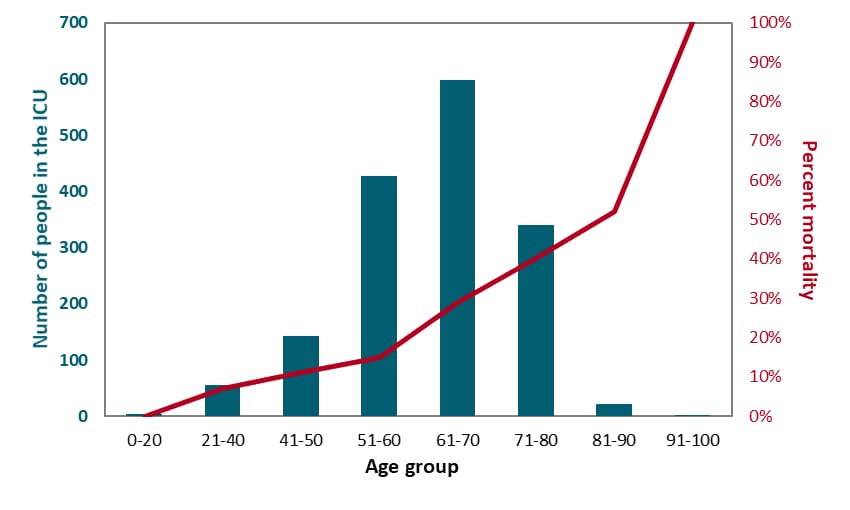
- Mortality was 26% and increased with age (Figure). Hypertension was associated with greater mortality (p <0.001).
Methods: Retrospective case series describing the clinical course of COVID-19 patients in 72 ICUs across the Lombardy region of Italy (largest study to date). Limitations: Not adjusted for multiple comparisons; data on bacterial pneumonia not presented; data missing for some outcomes; short follow-up.
Implications: In this Italian population, men with COVID-19 were more likely to be admitted to the ICU than women; mortality rate increased with age. Most patients in the ICU required mechanical ventilation.
Figure:
Note: Adapted from Grasselli et al. Teal bars show the number, in each age category, of COVID-19 patients admitted to the ICU in the Lombardy region of Italy between February 20 and March 8, 2020 (total n = 1,591). The red line shows the percent mortality of each age group. Mortality was highest among the oldest patients. Notably, almost two-thirds of participants were still hospitalized at the end of this short study. Reproduced with permission from JAMA. doi:10.1001/jama.2020.5394. Copyright©2020 American Medical Association. All rights reserved.
Kidney disease is associated with in-hospital death of patients with COVID-19external icon. Cheng et al. Kidney International (March 19, 2020).
Key Findings:
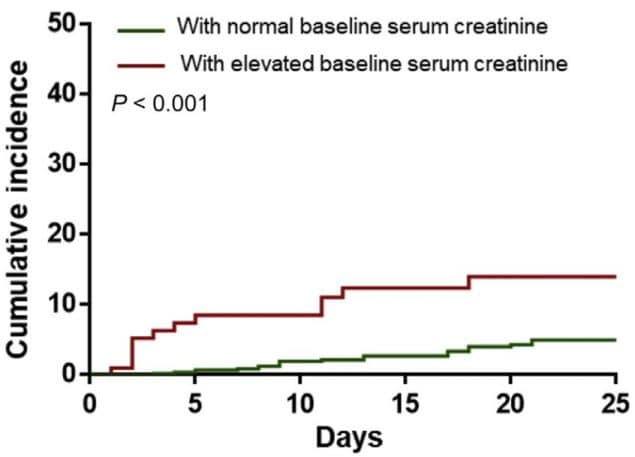
- During COVID-19 hospitalizations, 5.1% of adult patients developed acute kidney injury (AKI): 11.9% among those with kidney dysfunction at admission and 4% among those with normal kidney function at admission (Figure 1).
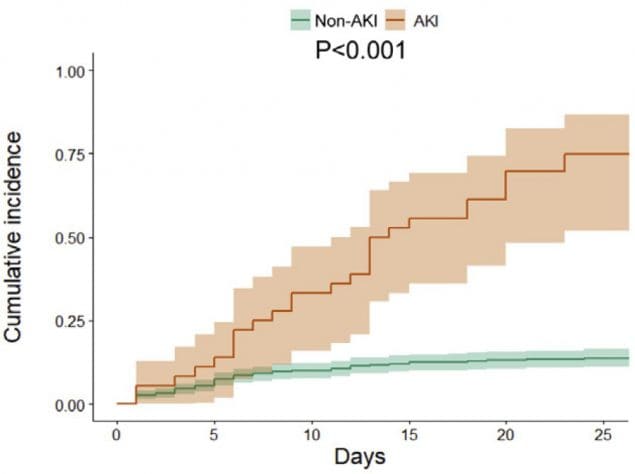
- AKI was associated with greater risk of in-hospital mortality (Figure 2).
- In-hospital mortality (16% overall) was higher in patients with kidney dysfunction on admission (34%).
Methods: Prospective cohort study of 701 hospitalized adult patients with COVID-19 in Wuhan that examined kidney function upon hospital admission, AKI during hospitalization, and in-hospital mortality. Limitations: No lab confirmation of SARS-CoV-2 infection; no information on renal function prior to hospitalization; insufficient data to assess a causal relationship between AKI and medications; other etiologies of kidney dysfunction not explored.
Implications: Risk of in-hospital death from COVID-19 was increased among patients with kidney dysfunction at admission or who developed AKI during hospitalization.
Figure 1.
Figure 2.
Note: Adapted from Cheng et al. Figure 1 shows the cumulative incidence of acute kidney injury of patients with COVID-19 (i.e., the percentage of over time that experienced acute kidney injury) for those with normal baseline serum creatinine (green line) and those with an elevated baseline serum creatinine (red line) at hospital admission. Figure 2. Shows the cumulative incidence for in-hospital deaths of COVID-19 patients, among those that developed AKI during the hospitalization and those that did not develop AKI. Licensed under CC-BY-NC-ND 4.0.
Compassionate use of remdesivir for patients with severe COVID-19external icon. Grein et al. NEJM (April 10, 2020).
Key findings:
- Among COVID-19 patients who received at least one remdesivir dose (n = 53), 68% showed improvement in oxygenation during the 2–3 weeks after the first dose; improvement was primarily among those with milder disease.
- Among the 53 assessed patients, 23% developed liver dysfunction and 8% developed kidney dysfunction; 13% (n = 7) died after remdesivir completion.
- Four patients discontinued remdesivir early because of worsening kidney failure (n = 1), multiorgan system failure (n = 1), or liver dysfunction (n = 2).
Methods: Description of 61 hospitalized COVID-19 patients (n = 53 analyzed) who received at least one dose of a 10-day remdesivir course (200 mg IV for one day, 100 mg IV daily for nine days) available for compassionate use in 9 countries. Forty received the full remdesivir course. Limitations: Lack of a control group; not randomized; no explanation of how patients were chosen for remdesivir; variation in length of remdesivir therapy; remdesivir was started late in disease course (median 12 days); viral load not reported; exclusion of 8 patients, 7 of whom had no data after day 1; limited baseline data on clinical severity; unclear if adverse events due to COVID-19 or remdesivir; remdesivir manufacturer participated substantially in manuscript creation.
Implications: Remdesivir might be safe in patients with severe COVID-19. However, interpretations of safety are limited by substantial methodologic shortcomings. Data from ongoing randomized, placebo-control trialsexternal icon are needed.
PEER-REVIEWED
Severe acute respiratory syndrome coronavirus 2 RNA detected in blood donations. Chang et al. Emerging Infectious Diseases (April 3, 2020).
Key findings:
- SARS-CoV-2 RNA was detected in 0.05% of blood donors in Wuhan during the epidemic peak (Figure).
- All persons who had detectable SARS-CoV-2 RNA in their blood were asymptomatic at blood donation and had no detectable IgG and IgM antibodies.
- Telephone follow-up revealed 33 people who reported symptoms consistent with SARS-CoV-2 infection; donations from these individuals were removed from circulation.
Methods: Wuhan Blood Center, China, screened 7,425 blood donations collected between December 21, 2019 and March 4, 2020 for SARS-CoV-2 RNA. Retrospective screening by pooled RT-PCR occurred between December 21, 2019 and January 22, 2020 and prospective real-time screening began on January 24, 2020. All donors in January and February 2020 were followed-up by telephone; donations from donors reporting symptoms were tested by RT-PCR. Limitations: SARS-CoV-2 RNA detection does not necessarily indicate infection or transmissibility.
Implications: Screening potential blood donors for SARS-CoV-2 might help identify early SARS-COV-2 infections. No current data indicate transmissibility through blood products. But if blood-borne transmission is possible, screening donors, might prevent transmission via contaminated blood products.
Figure:
Note: Adapted from Le Chang et al. Timeline of donations and symptom onset of 2019 novel coronavirus disease from 4 blood donors, China. Gold indicates a negative result for SARS-CoV-2 RNA; Red indicates a positive result. Green indicates the donor was asymptomatic and was afebrile; black indicates fever; white asterisk (*) indicates the donor’s fever subsided after taking self-prescribed antipyretic medications. PLT – platelet donation; TS – throat swab; WB – whole blood donation. Open access journal; all content freely available.
- Alexander et al. Ensuring access to medications in the US during the COVID-19 pandemicexternal icon. JAMA. Recommendations to address potential disruption of medication availability during the pandemic.
- Mehta et al. Potential shortages of hydroxychloroquine for patients with lupus during the coronavirus disease 2019 pandemicexternal icon. JAMA Health Forum. Brings attention to how shortages of hydroxychloroquine (HCQ) might affect socioeconomically vulnerable patients with lupus treated with HCQ and in remission.
- Editorial. Pandemic school closures: Risks and opportunitiesexternal icon. Lancet Child & Adolescent Health. With at least 188 countries implementing country-wide school closures, the editors review the impact on children and their caregivers.
- NIH. NIH begins study to quantify undetected cases of coronavirus infectionexternal icon. Anyone can volunteer to be screened for participation and collect their own microsample of blood at home to send in.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.