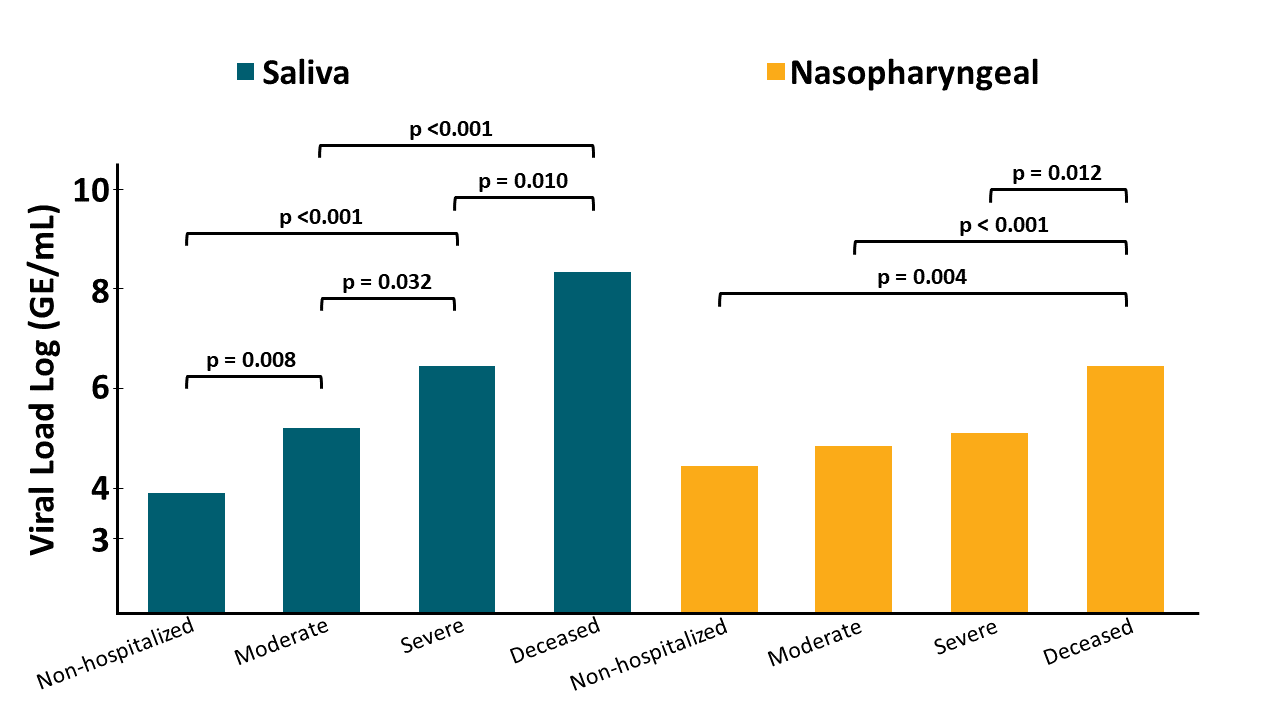COVID-19 Science Update released: January 22, 2021 Edition 73

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Section headings in the COVID-19 Science Update have been changed to align
with the CDC Science Agenda for COVID-19.
Here you can find all previous COVID-19 Science Updates.
Wide uptake of COVID-19 vaccines will be critical to ending spread of SARS-CoV-2. Here we present findings from 2 surveys on factors associated with intent to receive a COVID-19 vaccine.
PEER-REVIEWED
A. Attitudes toward a potential SARS-CoV-2 vaccineexternal icon. Fisher et al. Annals of Internal Medicine (December 15, 2020).
Key findings:
- Of 991 participants, 57.6% intended and 10.8% did not intend to be vaccinated; 31.6% were not sure.
- Intent to vaccinate was higher among men (64.0%) than women (51.6%) and among non-Hispanic Asian (77.5%) and non-Hispanic White (63.5%) persons than among non-Hispanic Black (39.3%) and Hispanic (44.5%) persons.
- Factors independently associated with no intent to vaccinate included lower education, identifying as Black or Hispanic, not having received last year’s influenza vaccine, and rural setting.
- Reasons provided for not intending to vaccinate (n = 83) were lack of trust (32.5%), not feeling comfortable with vaccines (21.7%), and concerns about side effects or safety (16.9%).
Methods: Cross-sectional survey of AmeriSpeak panel members (representative sample covering ~97% of adults in the US household population), fielded from April 16 to 20, 2020. Limitations: Participants were surveyed before phase 3 vaccine effectiveness and safety trial data were available; low response rate (16.1%).
B. Predictors of intention to vaccinate against COVID-19: Results of a nationwide surveyexternal icon. Ruiz et al. Vaccine (January 9, 2021).
Key findings:
- Of 804 participants, 62.2% were likely and 14.8% unlikely to be vaccinated; 23.0% were unsure.
- Intent to vaccinate was higher among men (71.9%) than women (53.8%), White (67.2%) than among Black (59.8%), or Asian (56.5%) persons, and among non-Hispanic (63.7%) than Hispanic (47.3%) persons.
- Predictors of intent to vaccinate were vaccine knowledge, not believing vaccine conspiracies, perceiving COVID-19 as a threat, past year influenza vaccine, ≥5 pre-existing conditions, male sex, household income ≥$120,000, Democratic party identity, and preferring COVID-19 information sources other than social media.
Methods: Nationwide online survey of US English-speaking adults from an internet survey panel of 2.5 million residents on June 15 and 16, 2020. Limitations: Participants were surveyed before phase 3 vaccine effectiveness and safety trial data were available. Might have missed Spanish speakers and people without internet access.
Implications for both studies (Fisher et al. & Ruiz et al.): Increased vaccine hesitancy was more frequent among non-Hispanic Black or Hispanic persons in both studies. Credible information about vaccine safety and effectiveness might improve uptake. Later surveys (Nikolovski et alexternal icon.) suggest that COVID-19 vaccine acceptance among vulnerable populations might have increased with news reports on effective vaccines. Reducing vaccine hesitancy will require a multifaceted approach, including trustworthy information, effective communication, and trusted messengers, potentially including community leaders, clinicians, and social media influencers.
PEER-REVIEWED
Bidirectional contact tracing could dramatically improve COVID-19 controlexternal icon. Bradshaw et al. Nature Communications (January 11, 2021).
Key findings:
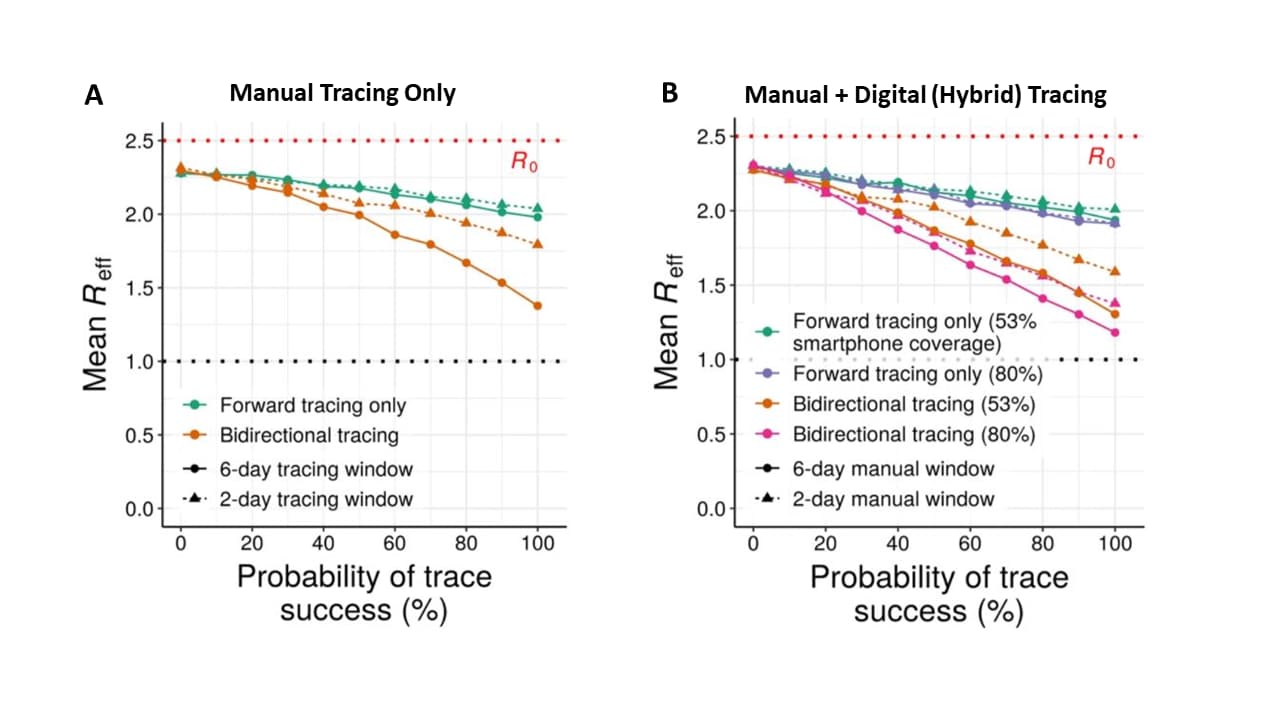
- Compared with forward-tracing alone, bidirectional contact tracing (including reverse tracing) reduced the effective reproduction number (Reff) by up to 24%, doubling the benefit relative to no tracing (Figure A).
- Bidirectional contact tracing with a 6-day pre-symptom onset window and high-uptake (80%) smartphone-based exposure notification dropped the Reff by up to 26% (Figure B).
Methods: A stochastic model was used to assess the efficacy of bidirectional contact tracing with digital notification using smartphones to broadcast chirps and allowing nearby persons with COVID-19 to “opt-in”, and manual tracing which asks individuals to remember their contacts. Limitations: Model only considers infected individuals without discerning between mild and severe symptoms and sensitivity of testing; model does not consider demographic, geospatial, or behavioral variation.
Implications: Bidirectional contact tracing, with manual and digital approaches, might be an important tool in improving overall COVID-19 control.
Figure:
Note: Adapted from Bradshaw et al. Comparing forward and bidirectional contact tracing at R0 = 2.5. Mean Reff achieved by manual tracing with 2 and 6-day manual tracing windows (A). Mean Reff achieved with hybrid manual plus digital tracing, assuming 90% data sharing and 53% or 80% smartphone coverage (B). Licensed under CC BY 4.0.
PEER-REVIEWED
Early transmissibility assessment of the N501Y mutant strains of SARS-CoV-2 in the United Kingdom, October to November 2020.external icon Leung et al. Eurosurveillance (January 7, 2021).
Key findings:
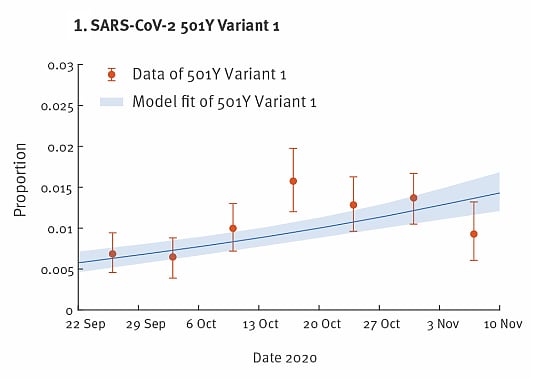
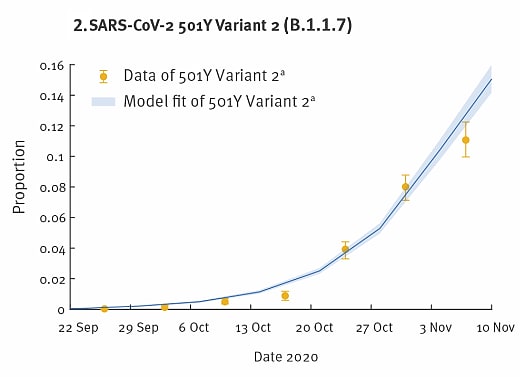
- SARS-CoV-2 variant 1 and variant 2 (B.1.1.7), both with the N501Y mutation, are 10% and 75% more transmissible than wild-type 501N, respectively.
- The proportion of total sequences accounted by 501Y lineages differed between October and late November 2020:
- Variant 1, with few mutations, had low circulation (2%) (Figure 1).
- Variant 2 (B.1.1.7), with at least 24 mutations, grew from 0.1% to 49.7% (Figure 2).
Methods: Sequences of wild-type 501N and 501Y mutations published in GISAIDexternal icon in the UK by December 19, 2020 were assessed. Transmissibility was compared between 501N and two 501Y lineages using a competition transmission model with a fitness interference framework. Limitations: Model does not account for age-related susceptibility, importation, or geographic separation; small sample.
Implications: The B.1.1.7 variant has a transmission advantage and is now the dominant lineage in the UK. Other studies suggest that although the B.1.1.7 variant has increased binding with ACE2 compared to wild-type (Ahmed et al.external icon), it should not escape natural immunity (Haynes et al.external icon). Widespread infection prevention efforts, including vaccination, are needed to curtail the spread of this new variant.
Figure 1
Figure 2
Note: Adapted from Leung et al. Proportion and model fit of SARS-CoV-2 501Y variants in the United Kingdom from September—November 2020. Figure 1 shows Variant 1 and Figure 2 shows Variant 2 (B.1.1.7). The co-circulation of both lineages is presented. Licensed under CC BY 4.0.
PREPRINTS (NOT PEER-REVIEWED)
Serologic surveillance and phylogenetic analysis of SARS CoV-2 infection in hospital health care workersexternal icon. Sikkens et al. medRxiv (January 12, 2021).
Key findings:
- Cumulative incidence of SARS CoV-2 infection was highest among healthcare workers (HCWs) working in COVID-19 patient care (13.2%) compared with HCWs in non-COVID-19 patient care (6.7%, hazard ratio [HR] 2.2, 95% CI 1.2-4.3) and HCWs not in patient care (3.6%, HR 3.9, 95% CI 1.8-8.6).
- Multiple potential HCW-to-HCW transmission clusters but no patient-to-HCW clusters were identified (Figure).
Methods: Prospective cohort study of 801 HCWs at 2 Dutch hospitals between March 23 and June 25, 2020 with SARS-CoV-2 serology testing and phylogenetic analyses of sequence data from viral samples. Limitations: Selection bias could cause underestimation of incidence; not all samples were available for phylogenetic analysis.
Implications: HCW-to-HCW transmission may play an important role in incidence of nosocomial SARS CoV-2 infection.
PEER-REVIEWED
Immunological characteristics govern the transition of COVID-19 to endemicityexternal icon. Lavine et al. Science (January 12, 2021).
Key findings:
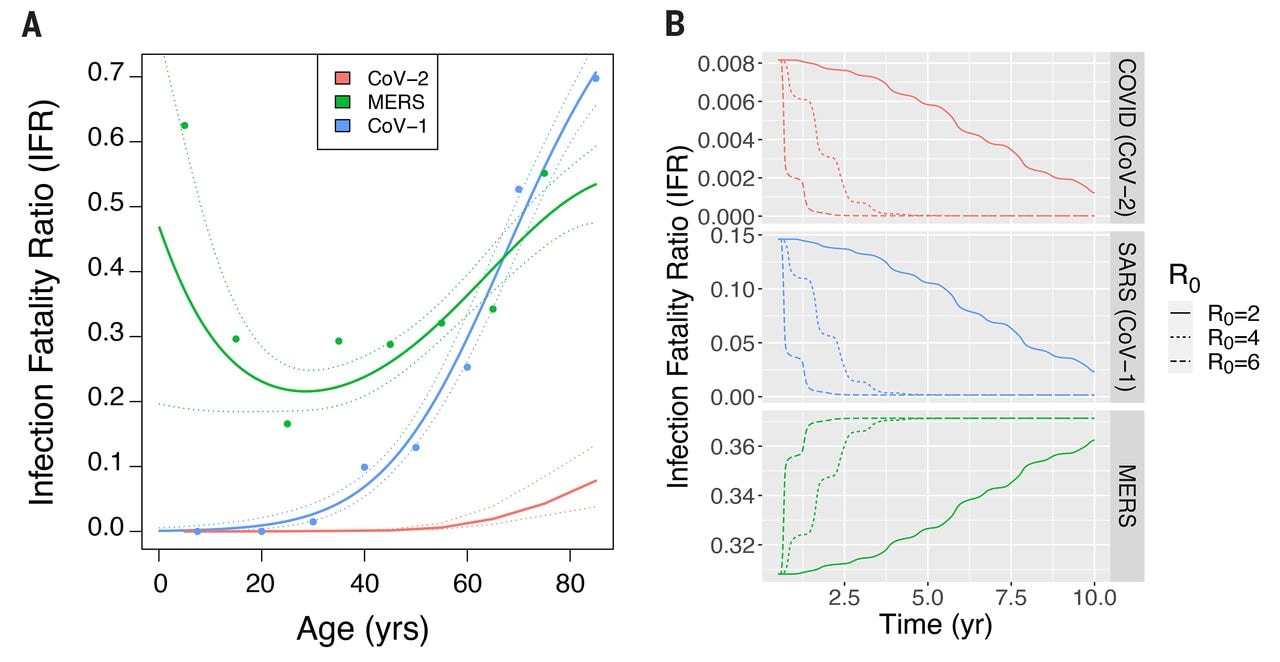
- As COVID-19 becomes endemic and the primary exposure is in childhood, SARS-CoV-2 may be no more virulent that the common cold.
- Ongoing need for mass vaccination will depend on the age-dependent infection fatality ratio (IFR) (Figure).
Methods: Modelling, incorporating susceptibility, pathology, and infectivity, to explore the changes in the transmission and severity of disease as COVID-19 becomes endemic. Limitations: Assumes immunity characteristics of all human coronavirus are similar and that the current pandemic is due to emergence into an immunologically naïve population.
Implications: Once endemic, mild disease in childhood might preclude the need for mass vaccination. These data should be interpreted with caution as there is still much to learn about the duration of immune memory and protective immunity to natural SARS-CoV-2 infection and in response to vaccines (See Sette et al.pdf iconexternal icon for review of current knowledge on this topic).
Figure:
Note: Adapted from Lavine et al. (A) Age dependence of the IFRs for the three emerging CoVs. Primary infections with MERS and CoV-1 are consistently symptomatic and the IFR and CFR are therefore assumed to be the same. CoV-1 and CoV-2 have J shaped profiles, with a monotonic increase in IFR with age. The age-specific IFR for MERS is U shaped, with high mortality in the young and old age groups. (B) The top panel illustrates how the overall IFR for CoV-2 drops dramatically, eventually falling below that of seasonal influenza (approximately 0.001) once the endemic steady-state is reached. From Lavine et al. Immunological characteristics govern the transition of COVID-19 to endemicity. Science. Reprinted with permission from AAAS.
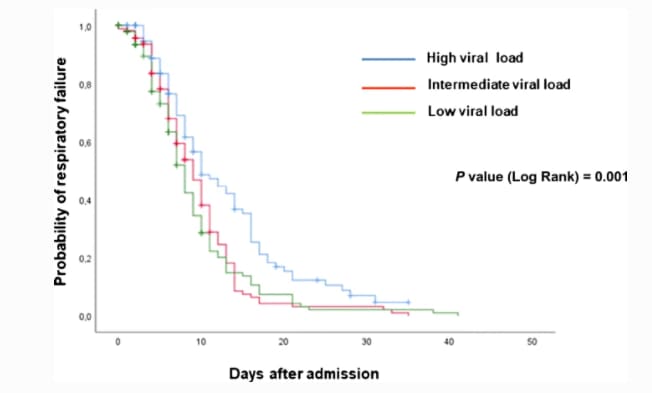
Impact of viral load at admission on the development of respiratory failure in hospitalized patients with SARS-CoV-2 infectionexternal icon. de la Calle et al. European Journal of Clinical Microbiology & Infectious Diseases (January 7, 2021).
Key findings:
- Among 455 hospitalized patients, 161 (35.4%) developed respiratory failure after a median of 9 days (IQR 6–11) (Figure).
- High viral load (Ct value <25) was associated with a higher risk of respiratory failure (OR 2.99, 95% CI 1.57-5.69) and in-hospital mortality (OR 2.04, 95%CI 1.44-4.00).
Methods: Retrospective cohort study involving medical record review was conducted among adults with RT-PCR confirmed SARS-CoV-2 infection at 1 hospital in Spain from March 1 to March 18, 2020. NP swabs were collected for RT-PCR testing with Ct value determination. Limitations: Single-center; RT-PCR does not distinguish between viable and non-viable virus.
Implications: SARS-CoV-2 viral load may predict disease severity among hospitalized COVID-19 patients.
Figure:
Note: Adapted from de la Calle et al. Probability of respiratory failure during hospitalization among patients with high, intermediate, and low viral load. Available via Nature Public Health Emergency Collection through Pubmed Central.
PREPRINTS (NOT PEER-REVIEWED)
Saliva viral load is a dynamic unifying correlate of COVID-19 severity and mortality.external icon Silva et al. medRxiv (January 10, 2021).
Key findings:
- High saliva viral load correlated with greater disease severity and was a better predictor of mortality compared to nasopharyngeal viral load (Figure).
- Saliva viral load was significantly higher in persons with COVID-19 risk-factors and decreased with rising levels of anti-RBD IgG.
Methods: Longitudinal cohort of 180 SARS-CoV-2-positive and 108 SARS-CoV-2-negative persons enrolled between March and June 2020 in the US were assessed for factors associated with disease severity and viral load kinetics. Limitations: Small sample size.
Implications: Saliva viral load is an important clinical measure of COVID-19 disease severity and mortality and should be monitored early. Serial saliva viral load monitoring could be useful, for example, in convalescent plasma treatment protocols.
Figure:
Note: Adapted from Silva et al. Mean salivary and nasopharyngeal viral load compared between disease severity and mortality categories of SARS-CoV-2 positive persons. Severity increases from left to right (non-hospitalized to deceased) within each sampling medium. Viral loads measured in saliva are indicated in teal; those measured in nasopharyngeal swabs are indicated in gold. Licensed under CC BY-ND 4.0.
PEER-REVIEWED
Umbilical cord mesenchymal stem cells for COVID‐19 acute respiratory distress syndrome: A double‐blind, phase 1/2a, randomized controlled trialexternal icon. Lanzoni et al. Stem Cells Translational Medicine (January 5, 2021).
Key findings:
- Umbilical cord mesenchymal stem cell (UC‐MSC) infusions in patients with COVID‐19 acute respiratory distress syndrome (ARDS) were safe.
- Compared with controls, treated subjects had decreased levels of inflammatory markers involved in cytokine storm, improved survival [hazard ratio (HR) 8.76 (95%CI: 1.07‐71.4)] , serious adverse event‐free survival [HR 6.22 (95% CI: 1.33‐28.96)], and time to recovery [HR 0.29 (95% CI: 0.09‐0.95)].
Methods: Double‐blind, phase 1/2a, randomized, controlled trial conducted to determine safety and explore efficacy of UC‐MSC infusions in subjects with COVID‐19 ARDS. Subjects randomized by ARDS severity to UC-MSC treatment (n = 12) and control (n = 12) groups. Primary endpoint was safety (adverse events [AE] within 6 hours post-infusion, cardiac arrest, or death within 24 hours post-infusion; and incidence of AEs). Secondary endpoints were survival 28 days post-infusion and time to recovery. Limitations: Small sample size.
Implications: UC-MSC infusions were safe and effective but warrant further investigation in a larger study. The decline in inflammatory cytokines seemed associated with the positive response to UC-MSC treatment.
Detection, Burden, and Impact
- Pater et al. Emergence and evolution of a prevalent new SARS-CoV-2 variant in the United States.external icon bioRxiv (January 13, 2021). A new variant of SARS-CoV-2 within the B.1.2 lineage called 20C-US/clade G emerged in the southern US in May 2020 and is likely the dominant variant in the US; it has high transmissibility without causing increased disease severity.
- Tu et al. Distinct patterns of emergence of SARS-CoV-2 spike variants including N105Y in clinical samples in Columbus, Ohioexternal icon. bioRxiv (January 19, 2021). Three SARS-CoV-2 clade 20C/G variants emerged in Ohio in December 2020; one has the N105Y mutation, similar to the B.1.1 lineage, and is likely highly transmissible.
- Pollock et al. Performance and implementation evaluation of the Abbott BinaxNOW rapid antigen test in a high-throughput drive-through community testing site in Massachusetts.external icon medRxiv (January 12, 2021). Performance characteristics presented support the use of the rapid antigen test, Abbott BinaxNOW, without RT-PCR confirmation.
- Bastos et al. The sensitivity and costs of testing for SARS-CoV-2 infection with saliva versus nasopharyngeal swabsexternal icon. Annals of Internal Medicine (January 12, 2021). Evidence suggests that saliva sampling for SARS-CoV-2 testing has similar sensitivity and costs less than nasopharyngeal swab collection.
Transmission of SARS-CoV-2
- Zimmerman et al. Incidence and secondary transmission of SARS-CoV-2 infections in schoolspdf iconexternal icon. Pediatrics (January 16, 2021). Within-school secondary transmission of SARS-CoV-2 infection was extremely rare during 9 weeks of in-person instruction in 11 North Carolina school districts.
Social, Behavioral, and Communication Science
- Agley et al. Misinformation about COVID-19: evidence for different latent profiles and a strong association with trust in science.external icon BMC Public Health (January 7, 2021). The authors found that many people believe both science-based facts and misinformation about COVID-19; mistrust in science was a predictor for believing misinformation. Using approaches specific to social media platforms can prevent misinformation (Merchant et al. Public health messaging in the era of social media.external icon JAMA).
- Nikolovski et al. Factors indicating intention to vaccinate among older US adults.external icon medRxiv (January 11, 2021). Of 7,402 respondents surveyed in a virtual clinical study of adults >65 years old, 91% were willing to vaccinate; willingness increased after the phase 3 Pfizer vaccine trial results were released.
- Zimba et al. SARS-CoV-2 testing service preferences of adults in the United States: discrete choice experiment.external icon JMIR Public Health and Surveillance (December 31, 2020). Online survey of 4,793 participants in a longitudinal cohort study found testing strategies with noninvasive methods and rapid turnaround time would improve uptake of SARS-CoV-2 testing.
Natural History of SARS-CoV-2 Infection
- Huang et al. 6-month consequences of COVID-19 in patients discharged from hospital: A cohort studyexternal icon. Lancet (January 8, 2021). Among 1,733 patients, 76% reported post-acute COVID symptoms at 6 months; the most common symptoms were fatigue, muscle weakness, and sleep difficulties. Outpatient clinics that provide follow-up care for sequelae of COVID-19 are being established and provide an opportunity for multidisciplinary research studies (Cortinovis et al. Long-term follow-up of recovered patients with COVID-19external icon. Lancet [January 8, 2021]).
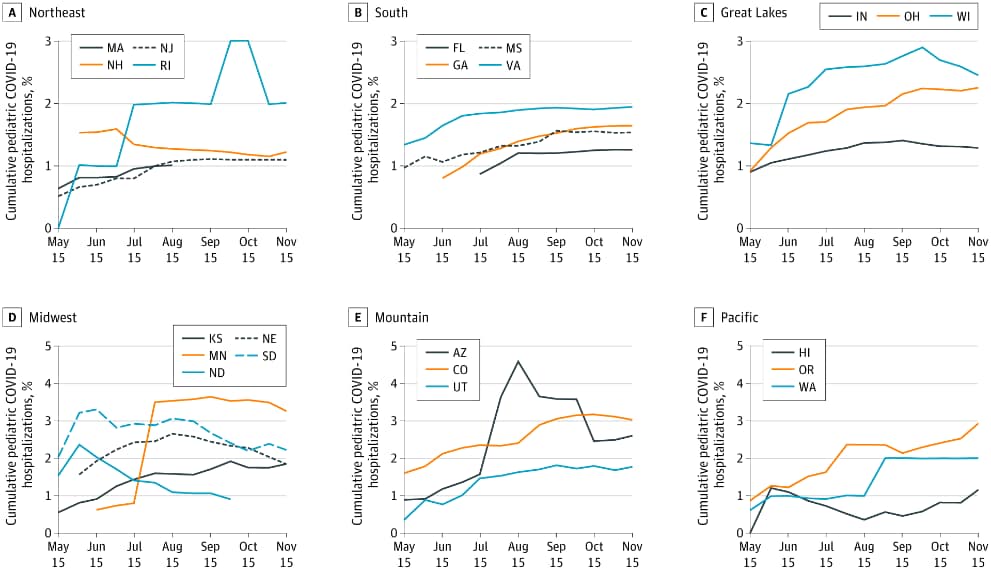
- Levin et al. Trends in pediatric hospitalizations for coronavirus disease 2019.external icon JAMA Pediatrics (January 11, 2021). From May 15 to November 15, 2020, the average cumulative pediatric hospitalization rate increased from 2.0 to 17.2 per 100,000 children in 22 states with variation across regions (Figure).
Note: Adapted from Levin et al. Trends in pediatric percentage of cumulative COVID-19 hospitalizations in 22 states, between May 15and November 15, 2020. Permission request in process.
Protection in Healthcare and Non-Healthcare Work Settings
- Howard et al. An evidence review of face masks against COVID-19.external icon PNAS (January 8, 2021). Widespread, population level use of masks is the most effective way to reduce SARS-CoV-2 community transmission; adherence and efficacy are two factors to consider in assessing impact.
Prevention, Mitigation, and Intervention Strategies
- Sadoff et al. Interim results of a phase 1-2s trial of Ad26.COV2.S COVID-19 vaccine.external icon NEJM (January 13, 2021). This multicenter, placebo-controlled, phase 1-2 trial of 805 participants found that Ad26.COV2.S vaccine is safe and immunogenic among adults.
Other Topics
- Langford et al. Antibiotic prescribing in patients with COVID-19: rapid review and meta-analysis.external icon Clinical Microbiology and Infection (January 5, 2021). A review of 154 studies found that among 35,263 patients, the antibiotic prescribing prevalence was 74.6% but the prevalence of bacterial infection was 8.6%; antimicrobial stewardship initiatives among patients with COVID-19 are urgently needed.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.