Our Methods

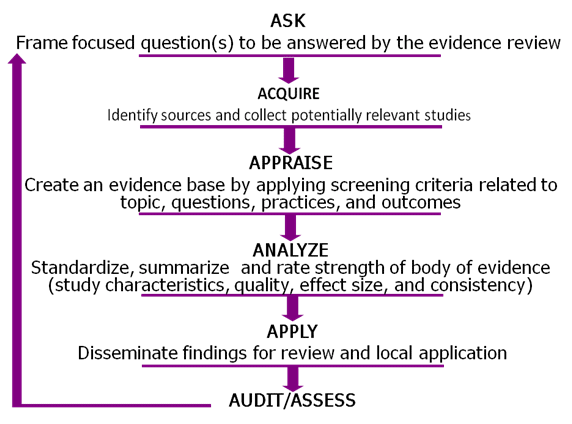
The LMBP systematic review methods are used to conduct reviews of practice effectiveness for pre- and post-analytic laboratory quality improvement practices. These methods were adapted from validated evidence-based review methods developed by the US Preventive Services Task Forceexternal icon (2008) and the Task Force on Community Preventive Servicesexternal icon (2002), among others. We use a six-step model, called “A-6.” These methods were initially pilot-tested across three quality improvement topics:
- Patient specimen identification
- Critical values test result reporting
- Blood culture contamination
Important elements of applying evidence-based methods to laboratory medicine include searching and locating evidence and appraising the study design and quality of the identified evidence. For more information on the LMBP methods, read our article published in Clinical Chemistry (2011) pdf icon[PDF – 508 KB].
The Systematic Review Team
Each review is conducted by a team of specialists in systematic review methods and subject matter experts. The aim is to:
- include input from people who know about and have experience with the practices reviewed
- reduce errors or bias when interpreting the information reviewed, and
- consider the viewpoints of key stakeholders.
The team typically includes experts in the topic area, a LMB Workgroup member, methods specialist, CDC staff including an economist, and one or more research fellows. This group develops the logic model for the review, conducts data collection and analysis, and presents findings.