Japanese Encephalitis Vaccine
Inactivated Vero cell culture-derived Japanese encephalitis (JE) vaccine (manufactured as IXIARO) is the only JE vaccine licensed and available in the United States. This vaccine was approved in March 2009 for use in people aged 17 years and older and in May 2013 for use in children 2 months through 16 years of age. Other JE vaccines are manufactured and used in other countries but are not licensed for use in the United States.
IXIARO is given as a two-dose series, with the doses spaced 28 days apart. Adults aged 18–65 years can get the second dose as early as 7 days after the first dose. The last dose should be given at least 1 week before travel. A booster dose (third dose) should be given if a person has received the two-dose primary vaccination series one year or more previously and there is a continued risk for JE virus infection or potential for reexposure.
For adults and children aged 3 years or older, each dose of IXIARO is 0.5 mL. For children aged 2 months through 2 years, each dose is 0.25 mL.
More information about JE vaccine can be found in the Vaccine Information Statement for IXIARO Japanese Encephalitis Vaccine Cdc-pd [PDF – 2 pages] and in the IXIARO product information available at the FDA IXIARO web page.
JE Vaccine for Travelers
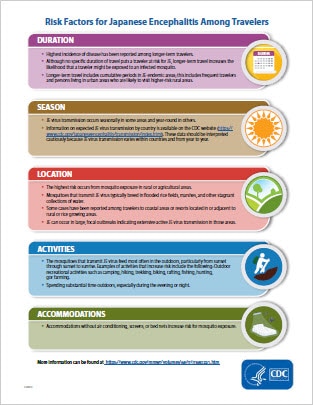
JE is a very low risk disease for most travelers to JE-endemic countries. However, some travelers will be at increased risk of infection based on factors including longer periods of travel, travel during the JE virus transmission season, spending time in rural areas, participating in a lot of outdoor activities, and staying in accommodations without air conditioning, screens, or bed nets.
All travelers to JE-endemic countries should take steps to avoid mosquito bites, and discuss the need for vaccination with their healthcare provider. The discussion should include the risks related to the specific travel itinerary, the likelihood of future travel to countries where JE occurs, the possible severe outcomes of JE disease, and information about the vaccine including cost and possible side effects.
JE Vaccine Recommendations
JE vaccine is recommended for persons moving to a JE-endemic country to live, longer-term (e.g., 1 month or longer) travelers, and frequent travelers to JE-endemic areas. JE vaccine also should be considered for shorter-term (e.g., less than 1 month) travelers with an increased risk of JE based on planned travel duration, season, location, activities, and accommodations. Vaccination also should be considered for travelers to endemic areas who are uncertain of specific duration of travel, destinations, or activities.
JE vaccine is not recommended for travelers with very low risk itineraries, such as shorter-term travel limited to urban areas or travel that occurs outside of a well-defined JE virus transmission season.
Healthcare providers may use CDC’s decision tree [PDF – 39 KB] to help determine when Japanese encephalitis vaccination for travelers is appropriate.
Precautions and Contraindications
- A serious allergic reaction after a previous dose of IXIARO, after any other JE vaccine, or after any component of IXIARO is a contraindication to further doses. The vaccine contains protamine sulfate, a compound known to cause allergic reactions in some people.
- Administration of IXIARO to pregnant women usually should be deferred. However, pregnant women who must travel to an area where risk for infection is high should be vaccinated when the theoretical risk of immunization is outweighed by the risk of infection.
Reactions to JE Vaccine
Reactions to IXIARO are generally mild and include pain and tenderness, mild headaches, myalgia (muscle aches), and low-grade fevers.
Healthcare providers are encouraged to report all adverse events that might be caused by vaccination to the CDC/FDA Vaccine Adverse Events Reporting System (VAERS) by one of the following methods:
- Submit a report online
- Print a VAERS Form [PDF – 2 pages] and fax or mail as instructed at https://vaers.hhs.gov/contact
- Call (800) 822-7967
For More Information
- MMWR Recommendations and Reports
Advisory Committee on Immunization Practices (ACIP) recommendations for use of JE vaccine - CDC’s Health Information for International Travel (Yellow Book)
Information about JE risk by country and recommendations for prevention of JE among travelers