 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Cryptosporidiosis Surveillance --- United States, 2003--2005Please note: An erratum has been published for this article. To view the erratum, please click here. Jonathan S. Yoder, MSW, MPH
Corresponding author: Jonathan S. Yoder, MSW, MPH, Division of Parasitic Diseases, National Center for Zoonotic, Vector-Borne, and Enteric Diseases, CDC, 1600 Clifton Rd., N.E., MS F-22, Atlanta, GA 30333. Telephone: 770-488-3602; Fax: 770-488-7761; E-mail: jey9@cdc.gov. AbstractProblem/Condition: Cryptosporidiosis, a gastrointestinal illness, is caused by protozoa of the genus Cryptosporidium. Reporting Period: 2003--2005. System Description: State and two metropolitan health departments voluntarily reported cases of cryptosporidiosis through CDC's National Notifiable Diseases Surveillance System. Results: During 2003--2005, the total number of reported cases of cryptosporidiosis increased from 3,505 for 2003 to 3,911 for 2004 and to 8,269 for 2005. All reporting areas submitted reports, with more reports from northern states. Compared with other age groups, a greater number of case reports were received for children aged 1--9 years and adults aged 30--39 years. Peak onset of illness occurred annually during early summer through early fall. Interpretation: Transmission of cryptosporidiosis occurs throughout the United States, with increased diagnosis or reporting occurring in northern states. An increase in cases reported for 2005 was attributable primarily to the occurrence of a single large recreational water-associated outbreak. State incidence figures should be compared with caution because individual state surveillance systems have varying capabilities to detect cases, and reporting might vary. The seasonal peak in age-specific case reports coincides with the summer recreational water season and might reflect increased use of communal swimming venues (e.g., lakes, rivers, swimming pools, and water parks) by young children. Public Health Action: Cryptosporidiosis surveillance provides data to educate public health practitioners and health-care providers about the epidemiologic characteristics and the disease burden of cryptosporidiosis in the United States. These data are used to improve reporting of cases, plan prevention efforts, and establish research priorities. IntroductionCryptosporidiosis is a gastrointestinal illness caused by protozoa of the genus Cryptosporidium (1). In otherwise well persons, clinical illness is characterized by watery diarrhea, which can be accompanied by abdominal cramps, loss of appetite, low-grade fever, nausea, vomiting, and weight loss; however, asymptomatic infection occurs frequently (2). Cryptosporidium also can cause an opportunistic infection in human immunodeficiency virus (HIV)--infected patients, who might experience life-threatening infection with profuse, watery, cholera-like diarrhea. However, incidence of this parasitic infection among the HIV-infected population has decreased since the introduction of highly active antiretroviral therapy (HAART) for HIV infection (3). Nitazoxanide is the only broad-spectrum antiparasitic drug that has been approved in the United States for treatment of cryptosporidiosis. Nitazoxanide can be prescribed for children aged >1 year and adults, but it has not proven effective for immunocompromised persons (4,5). Cryptosporidium is a genus of protozoan parasites within the phylum Apicomplexa, and its taxonomy continues to evolve. Cryptosporidiosis is a zoonotic disease that causes clinical disease in both humans and animals; species names are based primarily on the animal species serving as host (6). The majority of human isolates were believed previously to belong to a single species, Cryptosporidium parvum, but recent advances in molecular methods have led to a more complete understanding of the taxonomy of Cryptosporidium. Revised taxonomic understanding has clarified that multiple species infect humans, including C. hominis (known previously as C. parvum genotype I), which primarily infects humans, and C. parvum (known previously as C. parvum genotype II), which infects both humans and ruminants. To a lesser extent, human infections also have been documented with C. felis, from cats; C. canis, from dogs; C. meleagridis, from birds; C. suis, from pigs; C. muris, from rodents; and a C. cervine genotype from various animals (6). Illnesses caused by infection from the different Cryptosporidium species are similar clinically (7). Molecular studies also have demonstrated that humans are susceptible to multiple subtypes of C. parvum and C. hominis (6). Infected cattle serve as an important reservoir of C. parvum and therefore are substantial contributors to sporadic human cryptosporidiosis in certain areas (8,9). A study conducted during 1996--2004 in the United Kingdom suggests that improvements in drinking water treatment and source water protection might be responsible for decreasing the number of cryptosporidiosis cases in humans caused by C. parvum from ruminants (10,11). This benefit was influenced by the culling of cattle herds during the British outbreak of foot-and-mouth disease, which appeared to have had a substantial impact on the transmission peak occurring during the spring calving season (10--12). Cryptosporidium infection is transmitted by the fecal-oral route and results from the ingestion of Cryptosporidium oocysts through the consumption of fecally contaminated food or water or through direct person-to-person or animal-to-person contact. The oocysts are infectious immediately upon being excreted in feces. The infectious dose is low; feeding studies have demonstrated that the ingestion of as few as 10--30 oocysts can cause infection in healthy persons (13,14). Infected persons have been reported to shed 108--109 oocysts in a single bowel movement and to excrete oocysts for up to 50 days after cessation of diarrhea (15,16). Persons at increased risk for infection include 1) persons who have contact with infected animals, 2) persons who have ingested contaminated recreational (e.g., lake, river, pool, or hot tub) or drinking water, 3) close contacts of infected persons (e.g., those in the same family or household or in day care settings), and 4) travelers to disease-endemic areas (1,8,9) Although cryptosporidiosis cases occur sporadically, outbreaks have been well documented since the first reported U.S. drinking water-associated outbreak in 1984 (17) and the first U.S. recreational water-associated outbreak in 1988 (18). During 1988--2004, Cryptosporidium was identified as the causal agent of 33% (63 of 189) of reported recreational water-associated outbreaks and of 5.5% (12 of 219) of reported drinking water-associated outbreaks of gastroenteritis (18--29). Cryptosporidium has emerged as the most frequently recognized cause of recreational water-associated outbreaks of gastroenteritis, particularly in treated (disinfected) venues. In addition, foodborne outbreaks of cryptosporidiosis linked to ill foodhandlers and unpasteurized apple cider have been reported (30,31). Outbreaks resulting from person-to-person transmission in day care centers and from animal-to-person transmission in an animal nursery also have been reported (32,33). In 1994, the Council of State and Territorial Epidemiologists agreed to reporting of cryptosporidiosis as a nationally notifiable disease; the first full year for reporting began in 1995. Surveillance data for 1995--2002 have been published previously (34,35). This report summarizes national cryptosporidiosis surveillance data for 2003--2005. MethodsA confirmed case is one that has a positive laboratory finding. Confirmed cases of cryptosporidiosis in humans are reported voluntarily to CDC. Confirmed cryptosporidiosis is defined as the detection (in symptomatic or asymptomatic persons) of Cryptosporidium
A probable case of cryptosporidiosis is a clinically compatible case that is linked epidemiologically to a confirmed case. Because the case definition used by CDC does not include probable cases (36), certain states or areas report only confirmed cases of cryptosporidiosis. However, certain jurisdictions also report probable cases. A discussion has been initiated with partners regarding adding probable cases to the case definition. This surveillance summary includes both confirmed and probable cases as reported by jurisdictions. Testing for Cryptosporidium is not always included in routine examination of stool for ova and parasites (1). Commercially available immunoassay kits are available and might be more sensitive and specific than routine microscopic examination. DFA is an extremely sensitive and specific detection method and is considered the "gold standard" by many laboratorians; other immunodiagnostic kits that do not require microscopy (e.g., EIA and rapid immunochromatographic cartridge assays) also are available (38). Rapid immunoassays do not take the place of routine ova and parasite examination but might be useful in diagnosing Cryptosporidium infections. Only molecular testing (e.g., polymerase chain reaction) can be used to speciate Cryptosporidium; however, no species data are reported to CDC. State, District of Columbia (DC), and New York City (NYC) health department jurisdictions voluntarily report cases of cryptosporidiosis to CDC through the National Notifiable Diseases Surveillance System (NNDSS). Reports include the patient's place of residence (i.e., state and county), age, sex, race, ethnicity (i.e., Hispanic or non-Hispanic) and date of illness onset and indicate whether the case is linked to a known outbreak. An outbreak-related case is a laboratory-confirmed or probable case that is linked epidemiologically to at least one laboratory-confirmed case. Before analysis, these surveillance data were verified for accuracy by the reporting source and were finalized as of February 16, 2007. Because this surveillance summary includes probable cases of cryptosporidiosis and the data were finalized by the reporting jurisdiction more recently, the number of cases might differ slightly from the number reported in CDC's annual Summary of Notifiable Diseases. Analysis of national cryptosporidiosis surveillance data for 2003--2005 was conducted using SAS® v.9.1 (SAS Institute Inc.; Cary, North Carolina) and the Food Safety Information Link (FSI Link). FSI Link is an intranet-based tool available to CDC staff that provides access to NNDSS data and is used to monitor trends in, and investigate outbreaks of, reportable foodborne and waterborne diseases. Population data from the U.S. Census Bureau were used to calculate incidence rates. ResultsDuring 2003--2005, the total number of reported cases of cryptosporidiosis increased 11.6%, from 3,505 for 2003 to 3,911 for 2004, and then increased 111.4% to 8,269 for 2005 (Table 1). Cases reported to be outbreak related ranged from 6.6% of all cases reported for 2003 to 41.7% of all cases reported for 2005 (Table 1). All jurisdictions reported cryptosporidiosis cases during the reporting period, and the number of jurisdictions reporting more than four cases per 100,000 population increased from five in 2003 to seven in 2005 (Table 1). For 2005, incidence of cryptosporidiosis per 100,000 population ranged from 0.1 cases (in multiple states) to 18.2 cases (in New York), with 12 jurisdictions reporting >2.5 cases per 100,000 population (Figure 1; Table 1). The higher incidence of cryptosporidiosis in New York was influenced by a single large outbreak among >3,000 persons. Increased reporting of cases occurred in northern states. These surveillance data display a bimodal age distribution, with the greatest number of reported cases occurring among children aged 1--9 years and among adults aged 30--39 years (Figure 2). Increased reporting among younger children in 2005 was influenced by reporting of a single large outbreak in New York that comprised many probable cases. When reports for which a patient's sex was unknown or missing are excluded, the percentage of cases reported to have occurred among males varied annually from 49.6% (4,069 of 8,197) for 2005 to 55.9% (1,919 of 3,430) for 2003 (Table 2). A tenfold increase in reported cryptosporidiosis cases by illness onset date occurred during June--October compared with January--March (Figure 3). Age-specific analysis indicated that the seasonality in onset of illness was exhibited across all age groups, particularly among children aged 1--4 and 5--9 years (Figure 4). The majority of cases for which data on race were available for 2003--2005 occurred among whites, followed by blacks, Asians/Pacific Islanders, and American Indians/Alaska Natives (Table 2). However, data on race were lacking for 28.3%--47.1% of total annual case reports. Of patients for whom data on ethnicity were reported, 10.5%--11.5% were reported to be Hispanic (Table 2). However, data on ethnicity were lacking for 41.0%--60.9% of total annual case reports for 2003--2005. DiscussionNational cryptosporidiosis surveillance data are used to assess the epidemiologic characteristics and disease burden of cryptosporidiosis in the United States. The total number of cases reported annually increased during 2003--2005. Whether this increase reflects changes in reporting patterns and diagnostic testing practices or a real change in infection and disease caused by Cryptosporidium is unclear but was clearly influenced by outbreak-related case reporting. Outbreak-related probable case reporting does not account for the entire increase in reporting (Figure 5). This increase also follows the introduction of nitazoxanide, the first licensed treatment for the disease (4). Nitazoxanide was licensed in 2002 for children aged 1--11 years and in 2004 for children aged >11 years and adults. This drug introduction might have affected clinical practice by increasing the likelihood of health-care providers requesting Cryptosporidium testing, leading to an increase in subsequent case reports. The geographic variation, age distribution, and late-summer through early-fall seasonality were consistent with previous cryptosporidiosis surveillance summaries (34,35). Cryptosporidiosis is widespread geographically in the United States. These data and data from previous national cryptosporidiosis surveillance summaries indicate that the diagnosis or reporting of cryptosporidiosis might be higher in northern states (34,35). However, differences in cryptosporidiosis surveillance systems and reporting among states can affect the capability to detect and report cases, making interpretation of this observation difficult. Although cryptosporidiosis affects persons in all age groups, the number of reported cases was highest among children aged 1--9 years and adults aged 30--39 years (Figure 2). These data are consistent with reports of cryptosporidiosis incidence being higher among younger children and of transmission to their caregivers (e.g., child care staff, family members, and other household contacts) (32,34,35,39,40). Because information regarding immune system status is not collected as part of NNDSS cryptosporidiosis reporting, the number of immunosuppressed persons aged 30--39 years included in these surveillance data is unknown. However, the introduction of HAART therapy has decreased the amount of severe cryptosporidiosis being diagnosed in the United States (3). A tenfold increase in transmission of cryptosporidiosis occurred during summer through early fall. This increase coincided with increased outdoor activities (e.g., swimming during the summer recreational water season) and might reflect use of community swimming (essentially communal bathing) venues by younger children (8,18--29). Cryptosporidium is the leading cause of reported recreational water-associated outbreaks of gastroenteritis (19). Transmission through recreational water is facilitated by the substantial number of Cryptosporidium oocysts that can be shed by a single person; the extended periods of time that oocysts can be shed (15,16); the low infectious dose (13,14); the resistance of Cryptosporidium oocysts to chlorine (41); and the prevalence of improper pool maintenance (i.e., insufficient disinfection, filtration, and recirculation of water), particularly of children's wading pools (18--29,42). This seasonal variation also has been noted in state, Canadian provincial, and previous U.S. national surveillance data for cryptosporidiosis and giardiasis (34,35,39,40,43,45). The marked seasonal variation, higher incidence among children, and link to recreational water for cryptosporidiosis reflects the substantial increase in cases reported for 2005 compared with previous years (Figure 5). Of the 8,269 cases reported, 3,513 were reported by New York state, including 2,995 outbreak-related cases. In July--August, 2005, New York state experienced a cryptosporidiosis outbreak that sickened >3,000 persons, including at least 425 persons with confirmed cases, primarily children (46). The outbreak was associated with use of a recreational water interactive fountain (i.e., a fountain or water/spray feature intended for or accessible to recreational use that typically does not have standing water as part of the design). C. hominis was detected in patient stools and the holding tank of the interactive fountain. Although the interactive fountain was filtered and chlorinated, the treatment was not sufficient to inactivate Cryptosporidium, which has high chlorine resistance. This outbreak likely occurred as a result of fecal contamination by an infected patron and demonstrates why Cryptosporidium has become the leading cause of outbreaks of gastrointestinal illness associated with disinfected recreational water venues (19). Outbreak reports reflecting the multiple routes of transmission for cryptosporidiosis also were reported during 2003--2005. In 2003, unpasteurized apple cider made from contaminated apples sickened 144 persons with cryptosporidiosis (47). The cider was treated with ozone despite the fact that no data exist regarding the efficacy of ozone in treating food or juice products whose turbidity and low temperature render ozonation less effective. Also in 2003, a cryptosporidiosis outbreak that was associated with a middle- and high-school educational farm program was reported (48). Students participated in animal science classes and had contact with calves; transmission was associated with inadequate hygiene practices following animal contact. The propensity for cryptosporidiosis outbreaks to expand into communitywide outbreaks was underscored by a Kansas outbreak in 2003; the investigation identified 96 laboratory-confirmed cases of Cryptosporidium infection and >600 probable cryptosporidiosis cases (19). Multiple modes of transmission were identified, including swimming pools used by different swim teams and day camps and multiple day care facilities in which water activities were prevalent (19). The need to educate swim staff and develop work policies for employees who are ill with diarrhea was demonstrated by a 2004 outbreak associated with a recreational water park in California in which 336 people were sickened (49). Many water park employees were ill with a median onset date that preceded that for ill members of the public. Park policy required employees to be in the water regularly, and no policy was in place for reassigning employees who were ill with diarrhea (49). Although previous surveillance reports have repeatedly demonstrated more reported cases among males (34,35,39), a majority of reported cases in 2005 occurred among females. Because data on race and ethnicity are incomplete, conclusions cannot be made about differences in the epidemiology of cryptosporidiosis among members of different racial and ethnic populations. The data reported likely underestimate the cryptosporidiosis burden in the United States. Infection with enteric pathogens is highly underreported because 1) not all infected persons are symptomatic, 2) those who are symptomatic do not always seek medical care (50,51), 3) health-care providers infrequently include laboratory diagnostics in their evaluation of nonbloody diarrheal diseases (50), 4) a majority of laboratories do not test for Cryptosporidium unless it is specifically requested (52), and 5) case-reports are not always completed for positive laboratory results or forwarded to public health officials (53). The licensing of the first treatment for cryptosporidiosis (4) might influence health-care providers to request Cryptosporidium testing more often when ova and parasite exams are ordered. Although the true burden of cryptosporidiosis in the United States is unknown, an estimated 300,000 cases occur annually (53). Its low infectious dose (13), protracted communicability (15,16), and chlorine resistance (41) make Cryptosporidium ideally suited for transmission through drinking and recreational water, food, and both person-to-person and animal-to-person contact. Although no formalized national prevention plan exists for cryptosporidiosis, multiple efforts are focused on reducing the transmission of the parasite through two major routes of transmission. For drinking water, the Environmental Protection Agency (EPA) has implemented multiple regulations designed to improve the safety of surface water (e.g., lakes and rivers) supplies, including multiple regulatory changes following an outbreak in 1993 in Milwaukee, Wisconsin, that sickened >400,000 persons using the city water supply (19,20,54). Since 1993, no Cryptosporidium outbreaks associated with use of community surface water supplies have occurred (20,22--26). The signing of EPA's Groundwater Rule in 2006 marked an increased effort to decrease the risk associated with use of groundwater (well) supplies (20). This regulatory change is likely to decrease the continued occurrence of Cryptosporidium outbreaks associated with groundwater use. For recreational water, Cryptosporidium remains the leading cause of diarrheal illness outbreaks, especially in chlorinated venues (19). Efforts to reduce the transmission of this chlorine-resistant pathogen in pools will require a concerted effort to move beyond existing pool practices to include new technology (ultraviolet light or ozone inactivation), operational improvements (use of filtration enhancements that improve Cryptosporidium removal), and public education to reduce the continued swimming of people ill with diarrhea. In the United States, no federal oversight of chlorinated swimming venues occurs. All pool codes are reviewed and approved by state or local public health officials. As a result, no uniform national standards exist governing design, construction, operation, and maintenance of swimming pools and other treated recreational water venues. To provide guidance and improve pool code uniformity across the nation, CDC has initiated an effort to develop a National Model Aquatic Health Code and Risk Reduction Plan (available at http://www.cdc.gov/healthyswimming/model_code.htm). This effort will involve state, local, and federal public health officials and the aquatics industry in creating a voluntary model regulatory structure for state and local health departments to use in developing pool codes. Continued vigilance regarding risk reduction efforts and improving diagnosis and reporting of cryptosporidiosis are needed to decrease the transmission of cryptosporidiosis in the United States. Recommendations include prevention measures (Box 1) and measures to improve surveillance for cryptosporidiosis and increase understanding of its epidemiology and the associated disease burden (Box 2). General information about cryptosporidiosis is available from CDC at http://www.cdc.gov/ncidod/dpd/parasites/cryptosporidiosis/factsht_cryptosporidiosis.htm. Acknowledgments Cryptosporidiosis data were reported to CDC by state, DC, and NYC surveillance coordinators. Ruth Ann Jajosky, DMD, and Roland Richard, MPH, National Center for Public Health Informatics, CDC, facilitated access to the data and assisted with the analysis. Technical assistance was provided by Michael Arrowood, PhD, Michele Hlavsa, MPH, Stephanie Johnston, MS, John Williamson, ScD, and Lihua Xiao, DVM, PhD, Division of Parasitic Diseases, National Center for Zoonotic, Vector-Borne, and Enteric Diseases, CDC. References
Table 1 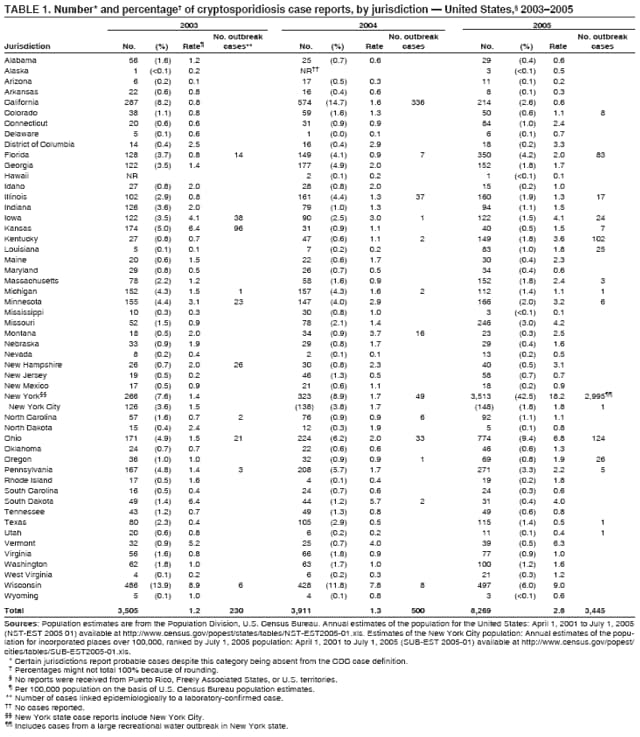 Return to top. Figure 1 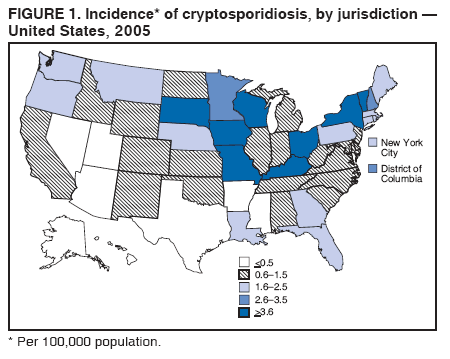 Return to top. Box 1  Return to top. Table 2 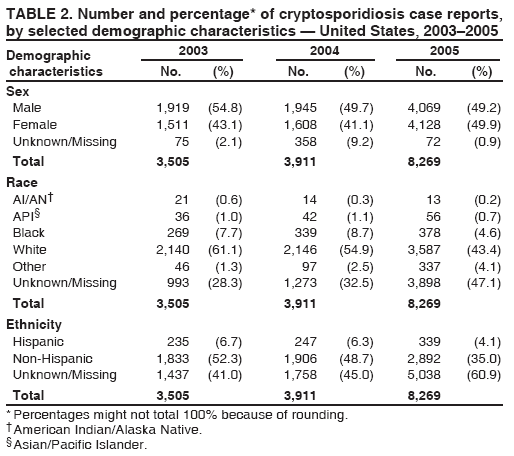 Return to top. Figure 2 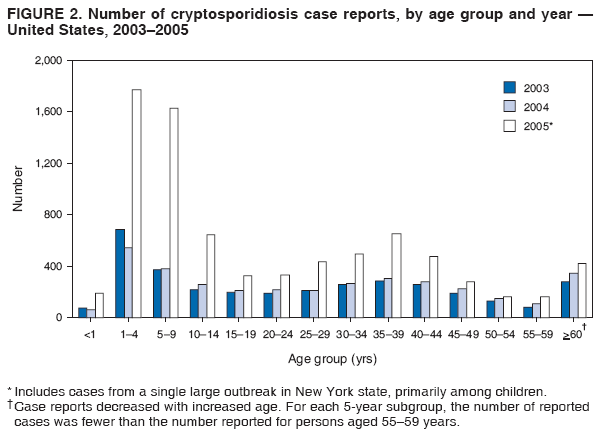 Return to top. Box 2  Return to top. Figure 3 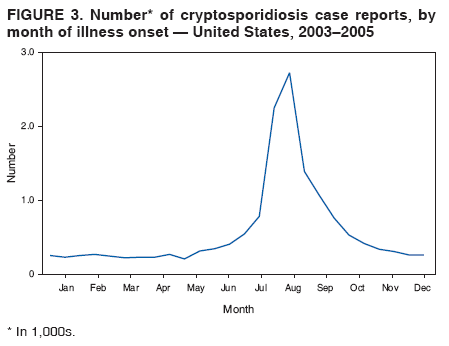 Return to top. Figure 4 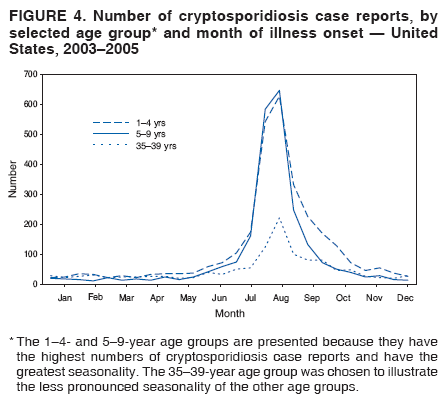 Return to top. Figure 5 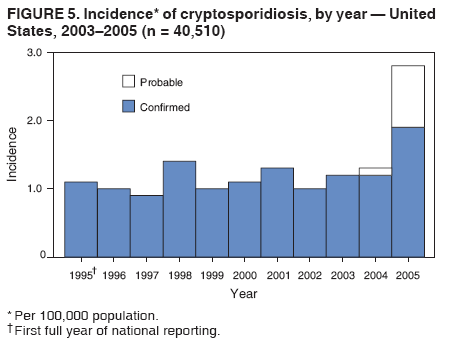 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Date last reviewed: 8/20/2007 |
|||||||||
|