 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Cryptosporidiosis Surveillance --- United States 1999--2002Michele C. Hlavsa, MPH1,2 Corresponding author: Michael J. Beach, PhD, Epidemiologist, CDC/NCID/DPD, 1600 Clifton Rd., NE, MS F-22, Atlanta, GA 30341; Fax: 770-488-7761; E-mail: mjb3@cdc.gov. Abstract Problem/Condition: Cryptosporidiosis, a gastrointestinal illness, is caused by protozoa of the genus Cryptosporidium. Reporting Period: 1999--2002. System Description: State and two metropolitan health departments voluntarily reported cases of cryptosporidiosis through CDC's National Electronic Telecommunications System for Surveillance. Results: During 1999--2002, the total number of reported cases of cryptosporidiosis increased from 2,769 for 1999 to 3,787 for 2001 and then decreased to 3,016 for 2002. The number of states reporting cryptosporidiosis cases increased from 46 to 50, and the number of states reporting more than four cases per 100,000 population increased from two to five. A greater number of case reports were received for children aged 1--9 years and for adults aged 30--39 years compared with other age groups. Incidence of cryptosporidiosis was particularly high in the upper Midwest and Vermont. Peak onset of illness occurred annually during early summer through early fall. Interpretation: Transmission of cryptosporidiosis occurs throughout the United States, with increased diagnosis or reporting occurring in northern states. However, state incidence figures should be compared with caution because individual state surveillance systems have varying capabilities to detect cases. The seasonal peak in age-specific case reports coincides with the summer recreational water season and might reflect increased use of communal swimming venues (e.g., lakes, rivers, swimming pools, and water parks) by young children. Public Health Action: Cryptosporidiosis surveillance provides data to educate public health practitioners and health-care providers about the epidemiologic characteristics and the disease burden of cryptosporidiosis in the United States. These data are used to improve reporting of cases, plan prevention efforts, and establish research priorities. IntroductionCryptosporidiosis is a gastrointestinal illness caused by coccidian protozoa of the genus Cryptosporidium (1). The hallmark symptom of this disease is diarrhea, which can be accompanied by abdominal cramps, loss of appetite, low-grade fever, nausea, vomiting, and weight loss; however, asymptomatic infection occurs frequently (2). Cryptosporidium can also cause an opportunistic infection in human immunodeficiency virus (HIV)--infected patients, who might experience life-threatening infection with profuse, watery, cholera-like diarrhea. The incidence of this parasitic infection among the HIV-infected population, however, has decreased since the introduction of highly active antiretroviral therapy (HAART) for treating persons with HIV infection (3). Nitazoxanide (NTZ, Alinia™, Romark Laboratories, L.C.; Tampa Bay, Florida), the only drug approved in the United States for treating cryptosporidiosis, can be used solely to treat children aged 1--11 years (4). A zoonotic disease, cryptosporidiosis also affects domestic (e.g., dogs, cattle, and sheep) and wild animals. Cryptosporidium hominus (known previously as Cryptosporidium parvum genotype I) naturally infects only humans, whereas Cryptosporidium parvum (known previously as Cryptosporidium parvum genotype II) infects both humans and cattle (5). Infected cattle serve as an important reservoir of C. parvum and therefore are substantial contributors to sporadic human cryptosporidiosis (6,7). Use of the term Cryptosporidium denotes either species unless otherwise noted. Cryptosporidium infection is transmitted by the fecal-oral route and results from the ingestion of Cryptosporidium oocysts through the consumption of fecally contaminated food or water or through person-to-person or animal-to-person transmission. The oocysts are infectious immediately upon being excreted in feces. The infectious dose is low; ingestion of as few as 10--30 oocysts has been reported to cause infection in healthy persons (8,9). Certain infected persons have been reported to shed <109 oocysts in their stool per day and to excrete oocysts for <15 days after their symptoms have resolved (10,11). Persons at increased risk for infection include 1) persons who have contact with infected animals, 2) persons who have ingested contaminated recreational (e.g., lake, river, pool, or hot tub) or drinking water, 3) close contacts of infected persons (e.g., those in the same family or household or in child care settings), and 4) travelers to disease-endemic areas (1,6). Although cryptosporidiosis cases can occur sporadically, outbreaks are well documented. During 1991--2000, Cryptosporidium was identified as a causal agent of 37.7% (40 of 106) of reported recreational water-associated and 8.5% (11 of 130) of reported drinking water-associated outbreaks of gastroenteritis of known and suspected infectious etiology* (12--16). Additionally, foodborne outbreaks of cryptosporidiosis linked to ill foodhandlers and unpasteurized apple cider have been reported (17,18). Outbreaks resulting from person-to-person transmission in child care centers and from animal-to-person transmission in an animal nursery also have been reported (19,20). Reporting of cryptosporidiosis as a nationally notifiable disease began in 1995. This report summarizes national cryptosporidiosis surveillance data for 1999--2002. MethodsLaboratory-confirmed cases of cryptosporidiosis can be reported voluntarily to CDC. Laboratory-confirmed cryptosporidiosis is defined as the detection (in symptomatic or asymptomatic persons) of Cryptosporidium
Testing for cryptosporidiosis should be specifically requested because it is not always included in routine examination of stool for ova and parasites (1). DFA is the most sensitive and the most specific detection method; however, other immunodiagnostic kits that do not require microscopy (e.g., EIA and rapid immunochromatographic cartridge assays) are also available (22). Only genetic testing (e.g., PCR) can be used to speciate isolates of Cryptosporidium. State, District of Columbia (DC), New York City (NYC), commonwealth, and territorial health departments can voluntarily report laboratory-confirmed cases of cryptosporidiosis to CDC through the National Electronic Telecommunications System for Surveillance (NETSS). Reports include the patient's geographic information (i.e., state and county), age, sex, race, ethnicity (i.e., Hispanic or non-Hispanic) and date of illness onset and indicate whether the case is related to a known outbreak. An outbreak-related case is a laboratory-confirmed case that is linked epidemiologically to another laboratory-confirmed case. The patient's HIV-status is not reported to CDC. Analysis of national cryptosporidiosis surveillance data for 1999--2002 was conducted by using SAS® v 8.2 (SAS Institute Inc.; Cary, North Carolina) and the Food Safety Information Link (FSI Link). FSI Link, an intranet-based tool available to CDC staff, provides access to NETSS data and is used to monitor trends in, and investigate outbreaks of, reportable foodborne and waterborne diseases. ResultsDuring the reporting period 1999--2002, the total number of reported cases of cryptosporidiosis increased 36.8% from 2,769 in 1999 to 3,787 in 2001 and then decreased 20.4% to 3,016 in 2002 (Table 1). Cases reported to be outbreak related made up 10.3%--13.7% of the total number of cases reported annually for 1999--2001 and 6.9% of the total reported for 2002. The number of states reporting cryptosporidiosis cases increased from 46 to 50 during the reporting period, and the number of states reporting more than four cases per 100,000 population increased from two in 1999 to five in 2002. For 2002, incidence of cryptosporidiosis ranged from 0.2 cases (multiple states) to 9.5 cases (Wisconsin) per 100,000 population, with Minnesota, North Dakota, South Dakota, Vermont, and Wisconsin each reporting more than four cases per 100,000 population (Figure 1) (Table 1). Wisconsin reported the greatest number of cases per 100,000 population for each of the 4 years of the reporting period. These surveillance data display a bimodal age distribution, with the greatest number of reported cases occurring among children aged 1--4 and 5--9 years and among adults aged 30--34 and 35--39 years (Figure 2). When reports for which patients' sex was unknown or missing are excluded, the percentage of cases reported to have occurred among males varied annually from 53.5% (2,002 of 3,745) for 2001 to 58.9% (1,623 of 2,755) for 1999 (Table 2). A five- to sixfold increase in reported cryptosporidiosis cases by illness onset occurred during June--October (i.e., weeks 25--42 [early summer through early fall]) compared with January--March (Figure 3). However, the date of illness onset was indicated for only 56.6% (1,567 of 2,769) of reported cases for 1999, for 51.7% (1,616 of 3,128) for 2000, for 60.6% (2,296 of 3,787) for 2001, and for 59.4% (1,791 of 3,016) for 2002. Age-specific analysis indicated that the seasonality in onset of illness was exhibited particularly among children aged 1--4 and 5--9 years (Figure 4) and across all age groups. The majority of cases for which data on race were available for 1999--2002 occurred among whites, followed by blacks, Asians and Pacific Islanders, and Native Americans (Table 2). However, data on race were lacking for 28.2%--36.5% of the total annual case reports. Of patients for whom data on ethnicity were reported, 9.8%--12.8% (185 of 1,889 for 2001 and 213 of 1,660 for 1999, respectively) were reported to be Hispanic (Table 2). However, data on ethnicity were lacking for 39.6%--50.1% of the total annual case reports for 1999--2002. DiscussionNational cryptosporidiosis surveillance data are used to assess the epidemiologic characteristics and disease burden of cryptosporidiosis in the United States. The total number of cases reported annually increased during 1999--2001 and then decreased in 2002. Whether this decrease reflects changes in reporting patterns and behavior or a real change in infection and disease caused by Cryptosporidium is unclear. FoodNet data document a decrease in case reports of cryptosporidiosis and other enteric diseases for 1996--2003; this decrease has been attributed to increased government and food industry emphasis on improved food safety (23). Cryptosporidiosis is geographically widespread in the United States. These data and data from the previous national cryptosporidiosis surveillance summary (1995--1998) indicate that the diagnosis or transmission of cryptosporidiosis might be higher in northern states (24). However, differences in cryptosporidiosis surveillance systems among states can affect the capability to detect cases, making interpretation of this observation difficult. Although cryptosporidiosis affects persons in all age groups, the number of reported cases was highest among children aged 1--9 years and adults aged 30--39 years. These data are consistent with reports of cryptosporidiosis incidence being higher among younger children and of transmission to their caregivers (e.g., child care staff, family members, and other household contacts) (19,24--26). A marked seasonality in the onset of illness occurs in early summer through early fall, and a five- to sixfold increase in transmission of cryptosporidiosis occurs during the summer. This increase coincides with increased outdoor activities (e.g., swimming during the summer recreational water season) and might reflect heavy use of community swimming (essentially communal bathing) venues by younger children (6,12--16). Cryptosporidium is the leading cause of reported recreational water-associated outbreaks of gastroenteritis; transmission through recreational water is facilitated by the substantial number of Cryptosporidium oocysts that can be shed by a single person; the extended periods of time that oocysts can be shed; the low infectious dose; the resistance of Cryptosporidium oocysts to chlorine; and the prevalence of improper pool maintenance (i.e., insufficient disinfection, filtration, and recirculation of water), particularly of children's wading pools (8--16,27,28). This seasonal variation also has been noted in state, Canadian provincial, and previous U.S. national surveillance data for cryptosporidiosis, as well as for giardiasis (24--26,29). Although the reason is unknown, increased transmission appears to occur among males, a finding that has been reported repeatedly (24--26). Because data on race and ethnicity are incomplete, conclusions cannot be made about differences in the epidemiology of cryptosporidiosis among members of different racial populations and between Hispanics and non-Hispanics. The data reported likely underestimate the cryptosporidiosis burden in the United States. Diarrheal diseases are highly underreported because 1) not all infected persons are symptomatic; 2) those who are symptomatic do not always seek medical care; 3) health-care providers do not always include diagnostics in their workup of diarrheal diseases because they might treat patients without testing stool for the pathogen; and 4) case-reports are not always completed for positive laboratory results or forwarded to public health officials (30). An estimated 1%--5% of cases of salmonellosis, another diarrheal illness, are reported to CDC through passive surveillance (31). If these estimates are used to extrapolate from the 3,016 cryptosporidiosis cases reported by the 50 states and DC for 2002, the cryptosporidiosis disease burden in the United States in 2002 could have been 60,320--301,600 cases (20.9--104.7 cases per 100,000 population). The true burden of cryptosporidiosis in the United States is likely to fall between these two estimates. Its low infectious dose, protracted communicability, and chlorine resistance make Cryptosporidium ideally suited for transmission through drinking and recreational water, food, and both person-to-person and animal-to-person contact. Prevention measures (Box 1) and measures to improve surveillance for cryptosporidiosis and increase understanding of its epidemiology and the associated disease burden (Box 2) have been recommended. Information about cryptosporidiosis is available from CDC at http://www.cdc.gov/ncidod/dpd/parasites/cryptosporidiosis/factsht_cryptosporidiosis.htm. Acknowledgments The authors thank the state, DC, and NYC surveillance coordinators for facilitating the reporting of cryptosporidiosis data to CDC; John Hatmaker and Man-huei Chang, Division of Public Health Surveillance and Informatics, Epidemiology Program Office, CDC, for facilitating access to the data and for their assistance during the course of its analysis; and Michael Arrowood and Stephanie Johnston, Division of Parasistic Diseases, National Center for Infectious Diseases, CDC, for technical assistance. References1. Chen X-M, Keithly JS, Paya CV, LaRusso NF. Cryptosporidiosis. N Engl J Med 2002;346:1723--31. 2. Hellard ME, Sinclair MI, Hogg GG, Fairley CK. Prevalence of enteric pathogens among community based asymptomatic individuals. Gastroenterol Hepatol 2000;15:290--3. 3. Kaplan JE, Hanson D, Dworkin MS, et al. Epidemiology of human immunodeficiency virus-associated opportunistic infections in the United States in the era of highly active antiretrorviral therapy. Clin Infect Dis 2000;30:S5--14. 4. Rossignol JA, Ayoub A, Ayers MS. Treatment of diarrhea caused by Cryptosporidium parvum: a prospective randomized, double-blind, placebo-controlled study of nitazoxanide. J Infect Dis 2001;184:103--6. 5. Xiao L, Fayer R, Ryan U, Upton SJ. Cryptosporidium taxonomy: recent advances and implications for public health. Clin Microbiol Rev 2004;17:72--97. 6. Roy SL, DeLong SM, Stenzel S, et al. Risk factors for sporadic cryptosporidiosis among immunocompetent persons in the United States from 1999 to 2001. J Clin Microbiol 2004;42:2944--51. 7. Goh S, Reacher M, Casemore DP, et al. Sporadic cryptosporidiosis, North Cumbria, England, 1996--2000. Emerg Infect Dis 2004;10: 1007--15. 8. DuPont HL, Chappell CL, Sterling CR, Okhuysen PC, Rose JB, Jakubowski W. The infectivity of Cryptosporidium parvum in healthy volunteers. N Engl J Med 1995;332:855--9. 9. Okhuysen PC, Chappell CL, Crabb JH, Sterling CR, DuPont HL. Virulence of three distinct Cryptosporidium parvum isolates for healthy adults. J Infect Dis 1999;180:1275--81. 10. Chappell CL, Okhuysen PC, Sterling CR, DuPont HL. Cryptosporidium parvum: intensity of infection and oocyst excretion patterns in healthy volunteers. J Infect Dis 1996;173:232--6. 11. Jokipii L, Jokipii AMM. Timing of symptoms and oocyst excretion in human cryptosporidiosis. N Engl J Med 1986;315:1643--7. 17. Quiroz ES, Bern C, MacArthur JR, et al. An outbreak of cryptosporidiosis linked to a foodhandler. J Infect Dis 2000;181:695--700. 18. Millard PS, Gensheimer KF, Addiss DG, Sosin DM, Beckett GA, Houck-Jankoski A, Hudson A. An outbreak of cryptosporidiosis from fresh-pressed apple cider. JAMA 1994 272:1592--6. Erratum in: JAMA 1995;273:776. 19. Cordell RL, Addiss DG. Cryptosporidiosis in child care settings: a review of the literature and recommendations for prevention and control. Pediatr Infect Dis J 1994:13:310--7. 20. Ashbolt RH, Coleman DJ, Misrachi A, Conti JM, Kirk MD. An outbreak of cryptosporidiosis associated with an animal nursery at a regional fair. Commun Dis Intell 2003;27:244--9. 21. CDC. Cryptosporidiosis (Cryptosporidium parvum) (crypto) case definition. Atlanta, GA: US Department of Health and Human Services, CDC; 1998. Available at http://www.cdc.gov/epo/dphsi/casedef/cryptosporidiosis_current.htm. 22. Johnston SP, Ballard MM, Beach MJ, Causer L, Wilkins PP. Evaluation of three commercial assays for detection of Giardia and Cryptosporidium organisms in fecal specimens. J Clin Microbiol 2003; 41:623--6. 24. Deitz VJ, Roberts JM. National surveillance for infection with Cryptosporidium parvum, 1995--1998: what have we learned? Public Health Rep 2000;115:358--63. 25. Naumova EN, Chen JT, Griffiths JK, Matyas BT, Estes-Smargiassi SA, Morris RD. Use of passive surveillance data to study temporal and spatial variation in the incidence of giardiasis and cryptosporidiosis. Public Health Rep 2000;115:436--47. 26. Majowicz SE, Michel P, Aramini JJ, McEwen SA, Wilson JB. Descriptive analysis of endemic cryptosporidiosis cases reported in Ontario 1996--1997. Can J Public Health 2001;92:62--6. 27. Korich DG, Mead JR, Madore MS, Sinclair NA, Sterling CR. Effects of ozone, chlorine dioxide, chlorine, and monochloramine on Cryptosporidium parvum occyst viability. Appl Environ Microbiol 1990;56:1423--8. 28. CDC. Surveillance data from swimming pool inspections---selected states and counties, United States, May--September 2002. MMWR 2003;52:513--6. 30. Mead PS, Slutsker L, Dietz V, et al. Food-related illness and death in the United States. Emerg Infect Dis 1999;5:607--25. 31. Chalker RB, Blaser MJ. A review of human salmonellosis: III. Magnitude of Salmonella infection in the United States. Rev Infect Dis 1988;10:111--24. * The denominator includes outbreaks whose etiology was reported to be acute gastrointestinal illness because they were suspected to be caused by an unidentified infectious agent. Table 1 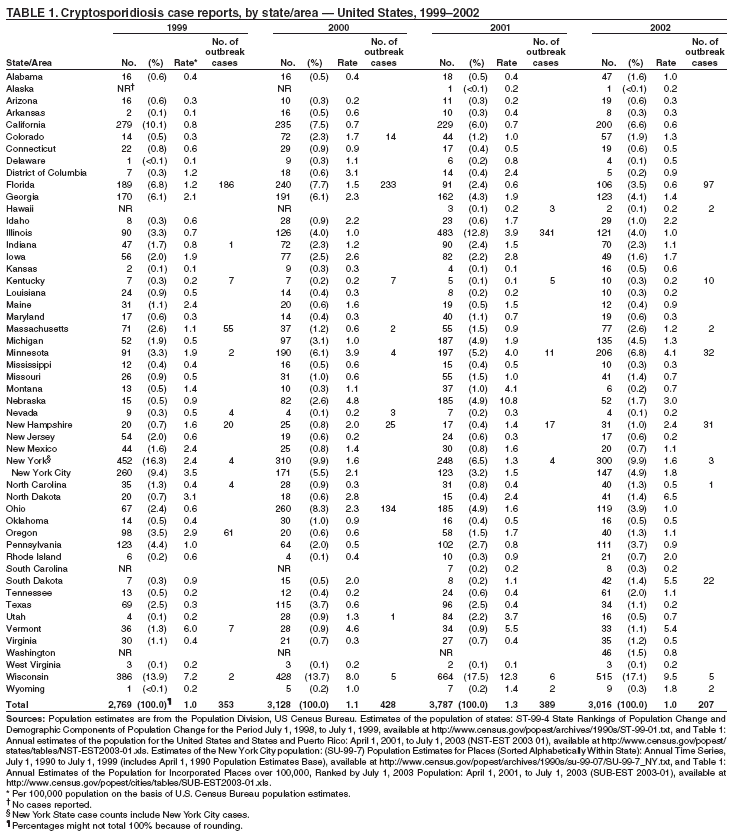 Return to top. Figure 1 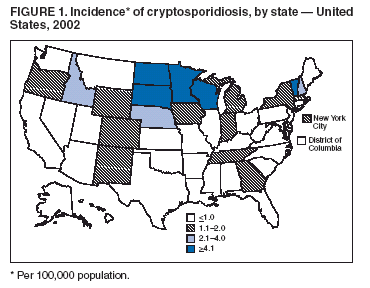 Return to top. Box 1  Return to top. Table 2 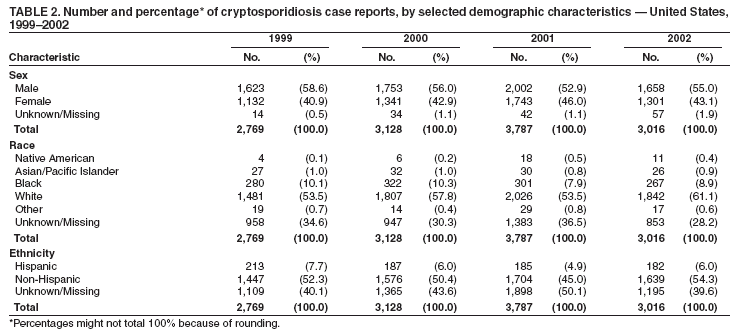 Return to top. Figure 2 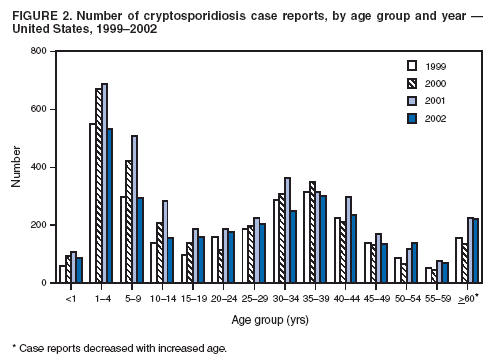 Return to top. Box 2  Return to top. Figure 3 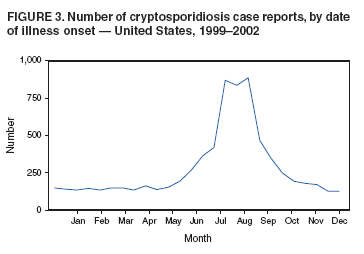 Return to top. Figure 4 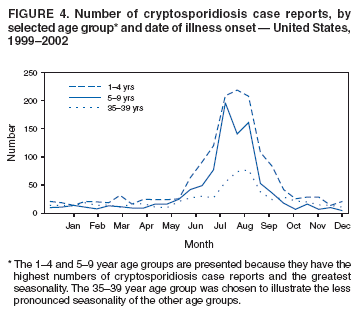 Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 1/19/2005 |
|||||||||
This page last reviewed 1/19/2005
|