 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Giardiasis Surveillance --- United States, 1992--1997Bruce W. Furness, M.D.1,2
1Epidemic Intelligence Service, Epidemiology Program Office, CDC
AbstractProblem/Condition: Giardia intestinalis, the organism that causes the gastrointestinal illness giardiasis, is the most commonly diagnosed intestinal parasite in public health laboratories in the United States. In 1992, the Council of State and Territorial Epidemiologists assigned giardiasis an event code that enabled states to begin voluntarily reporting surveillance data on giardiasis to CDC. Reporting Period: This report includes data that were reported from January 1992 through December 1997. Description of the System: The National Giardiasis Surveillance System includes data about reported cases of giardiasis from participating states. Because most states were already collecting data on occurrence of giardiasis, the assignment of an event code to giardiasis has allowed voluntary reporting of these data to CDC via the National Electronic Telecommunications System for Surveillance. Results: Since 1992, the number of states reporting cases of giardiasis to CDC has risen from 23 to 43. The annual number of giardiasis cases reported has ranged from 12,793 in 1992 to 27,778 in 1996. In 1997, cases per 100,000 state population ranged from 0.9 to 42.3, with 10 states reporting >20.0 cases per 100,000 population and a national average of 9.5 cases per 100,000 population. In 1997, New York State, including New York City, reported the highest number of cases (3,673, or 20.3 cases per 100,000 population), accounting for 14.5% of cases nationally; however, Vermont reported the highest incidence rate in 1997 (42.3 cases per 100,000 population). Both states have active surveillance systems in place for giardiasis. Cases have an approximately equal sex distribution. Nationally, rates were the highest among children aged 0--5 years, followed closely by persons aged 31--40 years. In these two age groups, most cases were reported during late summer and early fall --- an indication that transmission occurred during the summer. Interpretation: This report documents the first nationwide look at epidemiologic parameters and disease burden estimates for giardiasis in the United States. Transmission occurs in all major geographic areas of the country. The seasonal peak in age-specific case reports coincides with the summer recreational water season and might reflect the heavy use by young children of communal swimming venues (e.g., lakes, rivers, swimming pools, and water parks) --- a finding consistent with Giardia's low infectious dose, the high prevalence of diaper-aged children in swimming venues, the extended periods of cyst shedding that can occur, and Giardia's environmental resistance. Estimates based on state surveillance data indicate that as many as 2.5 million cases of giardiasis occur annually in the United States. Public Health Action: Giardiasis surveillance provides data to educate public health practitioners and health-care providers about the scope and magnitude of giardiasis in the United States. These data can be used to establish research priorities and to plan future prevention efforts. INTRODUCTIONGiardiasis is the gastrointestinal illness caused by the flagellated protozoan Giardia intestinalis, also known as G. lamblia or G. duodenalis. Giardia is the most commonly diagnosed intestinal parasite in public health laboratories in the United States (1--4). Giardia was the most frequently identified etiologic agent of outbreaks associated with drinking water in the United States for the years 1976--1994 (5). Giardia is spread from person to person and from animals to humans through fecal-oral transmission, has an incubation period of 3--25 days (median, 7--10 days), and has a two-stage life cycle --- trophozoite and cyst. The life cycle begins with ingested cysts, which release trophozoites (10--20 µm x 5--15 µm) in the duodenum. These trophozoites attach to the surface of the intestinal epithelium by using a ventral sucking disk and then reproduce by binary fission. The trigger for encystment is unclear, but the process results in the inactive, environmentally resistant form of Giardia --- a cyst (11--14 µm x 7--10 µm) that is excreted in feces (6). Giardiasis occurs when cysts are ingested through person-to-person transmission or ingestion of fecally contaminated food or water. The infectious dose is low: humans can be infected with as few as 10 cysts (6,7). Persons at greatest risk of exposure to infection are children in day care, their close contacts, men who have sex with men, backpackers and campers (via ingestion of unfiltered, untreated drinking water), travelers to disease-endemic areas, and persons drinking water from shallow wells (8--11). Giardia is found worldwide and infects domestic and wild animals (e.g., cats, dogs, cattle, deer, and beavers) (6,7). Giardiasis usually occurs sporadically, although outbreaks do occur. Waterborne outbreaks, associated with ingestion of both drinking and recreational water (e.g., lakes, rivers, or swimming pools) (5), and foodborne outbreaks (12,13) are well documented as are person-to-person outbreaks among men who have sex with men (8) and among children and staff in day care centers (14). The relative contribution of waterborne, foodborne, and person-to-person transmission to sporadic giardiasis is unknown. Clinically, Giardia produces a broad spectrum of gastrointestinal symptoms, including one or more of the following symptoms: diarrhea, flatulence, bloating, weight loss, abdominal cramping, nausea, malabsorption, foul-smelling stools, steatorrhea, fatigue, anorexia, and chills. Although the hallmark of giardiasis is diarrhea, asymptomatic infections can occur (15,16), especially in children and in persons with prior infections (6). Giardia cysts can be excreted in the stool intermittently for weeks or months, resulting in a protracted period of communicability (14,17,18). Because infections can be asymptomatic or characterized by mild signs and symptoms, giardiasis is often regarded as a benign gastrointestinal illness, although chronic or debilitating giardiasis has been reported (15). From 1979 through 1988, an estimated 4,600 hospitalizations per year in the United States resulted from severe giardiasis and its complications, resulting in an average of 23,238 days per year in the hospital and a mean annual incidence of 2.0 hospitalizations per 100,000 persons (15). Volume depletion was reported for 33% of case-patients, and 19% of hospitalized children aged <5 years had a codiagnosis of failure to thrive (15). Many effective treatment alternatives are available for patients with symptomatic giardiasis (Table 1). Metronidazole is the treatment most often prescribed in the United States. Furazolidone is a less effective treatment option, but it is the only drug approved by the U.S. Food and Drug Administration (FDA) for treatment of giardiasis in the United States. Because furazolidone is available in liquid form, it is often used to treat children (6). Quinacrine, an effective and inexpensive treatment option, is not available from any U.S. manufacturer, although several compounding pharmacies have made it available. Tinidazole is widely used throughout the world; however, it is not approved for use in this country. Albendazole has been reported to be as effective as metronidazole with fewer side effects among children aged 2--12 years (19). Paromomycin, a nonabsorbed aminoglycoside, is less effective than other agents but is used for treatment among pregnant women because of potential teratogenic effects of the other agents (20). A combination of metronidazole and quinacrine has been used to treat refractory cases (21). Despite the public health importance of giardiasis, reliable data on national incidence and prevalence in the United States are not available. Estimates have been extrapolated from published data collected by states with active giardiasis surveillance, laboratory surveys, and waterborne-disease outbreak reports. Giardiasis is the most frequently reported diarrheal disease in northern New England (10), and from 1983 through 1986, it was the most common reportable disease in Vermont (22). Giardia also was the most prevalent protozoan parasite in Arkansas during 1997 (23) as well as the most commonly reported enteric pathogen in Wisconsin during the years 1983--1986 (24). The prevalence of Giardia in stool specimens submitted for examination ranges from 2% to 5% in industrialized countries and from 20% to 30% in developing countries, and it can be as high as 35% among children attending day care centers in the United States in a nonoutbreak setting (4,6). Before the tightening of water treatment standards, an estimated 25% of endemic cases of giardiasis in the United States were waterborne (7,9). Furthermore, giardiasis was one of the two leading recognized etiologies in the 129 water-associated disease outbreaks that occurred during the years 1991--1994 in the United States (7). Although giardiasis reporting is required by 43 states, it is not a nationally notifiable disease (25,26). In 1992, the public health importance of the disease prompted the Council of State and Territorial Epidemiologists to assign an event code (code 11570) to giardiasis to allow states to voluntarily transmit their reported giardiasis data to CDC via the National Electronic Telecommunications System for Surveillance (NETSS) (27). State health departments collect data on giardiasis case-patients from both health-care providers and laboratories, and the data are subsequently transmitted with notifiable disease information to CDC via NETSS. The purpose of the National Giardiasis Surveillance System is to estimate the disease burden in the United States and to monitor the demographic parameters (sex, age, race, ethnicity), seasonality, and geographic variation of giardiasis. This report summarizes national surveillance data on giardiasis for the years 1992--1997. METHODSThe diagnosis of giardiasis is made through examination of stool specimens that are typically collected and preserved in 10% formalin, although fresh stool may also be examined. Light microscopy can be used to visualize the parasite via wet mount, staining (trichrome or iron hematoxylin), or the direct fluorescent antibody detection method (monoclonal antibodies). In addition, enzyme linked immunosorbent assay (ELISA)-based kits have been used to detect Giardia-specific antigen in stool (4,6). Approximately 85% of infections can be diagnosed with a single stool specimen. Sensitivity increases with the number of stool specimens examined, so that three specimens collected every other day during a 5-day period will detect approximately 90% of infections (28). In the rare occasions when infections are suspected but multiple stool tests are negative, duodenal fluid can be sampled for Giardia trophozoites. Three procedures have commonly been used: the string test (Entero- test), endoscopy with upper intestinal aspiration, and endoscopy with upper intestinal biopsy (6). No serologic test is commercially available (4,6). The National Giardiasis Surveillance System collects data on persons who have either symptomatic or asymptomatic giardiasis, seek health care, have a positive diagnostic test result, and are reported to both the state and CDC (29). This report represents the first time that national surveillance information on giardiasis has been published and includes reports from the 43 participating states. RESULTSSince the inception of the National Giardiasis Surveillance System in 1992, the number of states voluntarily reporting cases and the number of states reporting >100 cases per year increased through 1994 and subsequently plateaued. In 1992, only 23 states reported giardiasis cases to CDC, with only 17 states reporting >100 cases. By 1997, 43 states reported giardiasis cases, with 40 of those states reporting >100 cases each. In 1992, a total of 12,793 cases of giardiasis were reported. The number of reported cases eventually doubled by 1994. Between 1994 and 1997, case reporting plateaued at 25,389--27,778 cases reported per year, with sex distribution being approximately equal (Figure 1). In 1997, New York State, including New York City, reported the highest number of cases (3,673), accounting for 14.5% of cases nationally. In 1997, cases per 100,000 population ranged from 0.9 to 42.3 in reporting states. Vermont reported the highest incidence rate (42.3 cases per 100,000 population) (Table 2). Ten states reported >20.0 cases per 100,000 population (Alaska, Colorado, Minnesota, Nebraska, New Hampshire, New York, North Dakota, Oregon, Vermont, and Wisconsin). Seven states reported no cases to CDC in 1997 (California, Connecticut, Kentucky, New Jersey, North Carolina, South Carolina, and Texas) (Table 2). A bimodal age distribution in giardiasis rates was observed. The highest rates of giardiasis occurred among children aged 0--5 years, followed closely by persons aged 31--40 years (Figure 2). In 1997, 34.4% (8737/25,389) of cases were reported with unknown race/ethnicity. Of those cases for whom information on race and ethnicity was reported, 78.8% (13,129/16,652) were reported as white, 10.9% (1809/16,652) as Hispanic, 6.0% (991/16,652) as black, 3.5% (575/16,652) as Asian, 0.7% (113/16,652) as Native American, and 0.2% (35/16,652) as of other race or ethnicity. A marked seasonality in reported giardiasis cases occurred, peaking during weeks 37--46, or late summer and early fall (Figure 3). Thus, most case reports arrived at CDC during September and October. Age-specific analysis of the data illustrates that this seasonality is primarily exhibited by persons in two age groups --- 0--5 and 31--40 years (Figure 4). DISCUSSIONSince NETSS giardiasis reporting began in 1992, both the number of states participating in voluntary reporting to CDC and the annual number of cases reported have increased. This national surveillance system provides data to assess epidemiologic characteristics and to estimate the disease burden of giardiasis. In the surveillance data, no sex-specific differences were observed. This finding is in contrast to what was expected on the basis of both the estimated national incidences of salmonellosis and shigellosis, which are higher for women than for men (30), and the estimated incidence of hospitalization for giardiasis, which was 22%--70% higher for women than for men; researchers have hypothesized that these estimates are higher for women because they have increased care-giving responsibility and exposure to infected children (15). Why these surveillance data do not show sex-specific differences remains unclear. Although giardiasis affects all age groups, a bimodal age distribution was observed. The incidence of giardiasis was highest for children aged 0--5 years, followed by adults aged 31--40 years. These data correlate with reports of giardiasis prevalence being higher than average among children who attend day care centers as well as the family members and day care workers who care for these children (11). This same age distribution has also been documented for state-based surveillance of giardiasis (10,22), hospitalizations for severe giardiasis (15), and cryptosporidiosis (31). Although all racial and ethnic groups were represented --- with rates per 100,000 population ranging from 3.1 (African American) to 6.9 (white) --- underdiagnosis and underreporting limit interpretation of these data. Only half of the case reports received through NETSS from 1994 through 1997 included information about both the race and ethnicity of patients. In comparison, 95%--99% of case reports during the same period included sex and age data (32). The seasonality of giardiasis has been documented (2,3,10,23,24). The greatest number of reports of giardiasis are received during the late summer and early autumn (Figure 3). Because case reports can take 1--2 months to reach CDC after onset of illness (2), this peak reflects increased transmission during the summer months. The seasonal peak in cases coincides with the summer recreational water season and might reflect the increased use by young children of community swimming (essentially communal bathing) venues --- a finding consistent with Giardia's low infectious dose, the high prevalence of diaper-aged children in swimming venues, and Giardia's role as one of the most common causes of recreational water-associated disease outbreaks in the United States (5). This seasonal variation has also been shown for cryptosporidiosis (31). Giardiasis is geographically widespread in the United States. States with >20.0 case reports per 100,000 population represent almost every major region of the country. Data collected and reported via NETSS, though, are not sufficient for interpreting geographic variation in the incidence of giardiasis. Data from state laboratories suggest that the incidence of giardiasis might be higher in the midwest and northwest regions (2,3). In contrast, the highest mean annual incidence of hospitalization for giardiasis occurs in southern states (15). In 1997, 25,389 cases of giardiasis were reported, or 9.5 case-patients per 100,000 population. This is an underestimation of the 1997 giardiasis disease burden in the United States because a) the system is not representative of all persons infected with Giardia, b) seven states did not submit any giardiasis reports to CDC in 1997, and c) diarrheal diseases are often underreported (15). Calculating the sensitivity of the National Giardiasis Surveillance System in estimating the disease burden in the United States is difficult. One way to estimate the burden is to look at the estimated proportion of persons with diarrheal illness who seek medical care and the proportion of cases reported. Only 8% of persons with a diarrheal illness seek medical care, and subsequently, only 1%--5% of foodborne disease cases are reported to CDC through passive surveillance systems, according to estimates from FoodNet, an active surveillance system for estimating the burden of foodborne disease transmission in the United States (33). Assuming this pattern is also true for Giardia, and using the National Giardiasis Surveillance System, the calculated incidence of gi-ardiasis in the United States during 1997 could be as low as 500,000 or as high as 2.5 million case-patients (185--926 cases per 100,000 population). Another way to approximate the giardiasis disease burden is to extrapolate from incidence data reported from states with active giardiasis surveillance systems. For example, Vermont reported 45.9 cases per 100,000 population per year from 1983 through 1986 (22), and Wisconsin reported 49.1 cases per 100,000 population in 1988 (24). These data are close to Vermont's 42.3 case-patients per 100,000 population reported for 1997 through the National Giardiasis Surveillance System. If this state surveillance information is extrapolated to the national level (i.e., 46--49 case-patients per 100,000 population) by using 1997 population estimates, approximately 124,000--132,000 case reports could be expected nationally. The true burden of giardiasis in the United States most likely ranges between these two estimates: from 46--49 cases to 185--926 cases per 100,000 population. Giardia is the most commonly detected intestinal protozoan parasite in the world (1--4), and it likely causes a range of 100,000 to 2.5 million infections each year in the United States, with an expected incidence equivalent to that reported for Salmonella and Shigella (34,35). Giardia's protracted communicability, low infectious dose, and environmental resistance make it easily transmitted by drinking and recreational water, by food, and from person to person. Because Giardia can be a cause of severe gastrointestinal illness and effective treatment options are available, health-care providers should include testing for giardiasis in the workup for diarrheal illness. The following recommendations would lead to an improved National Giardiasis Surveillance System:
Recommendations for Prevention and Control of GiardiasisPractice good hygiene.
Avoid water that might be contaminated.
Avoid food that might be contaminated.
Avoid fecal exposure during sex.AcknowledgmentsThe authors thank the state surveillance coordinators and the state epidemiologists for facilitating reporting of giardiasis data to CDC, the Epidemiology Program Office for facilitating access to the data, and David G. Addiss, Dennis D. Juranek, and Thomas R. Navin of the Division of Parasitic Diseases, National Center for Infectious Diseases, for thoughtful input. References
Table 1  Return to top. Figure 1 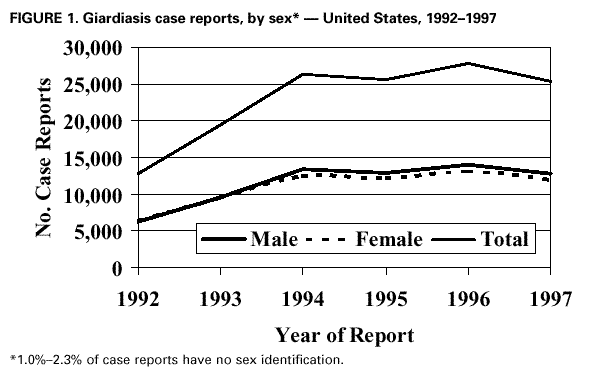 Return to top. Table 2 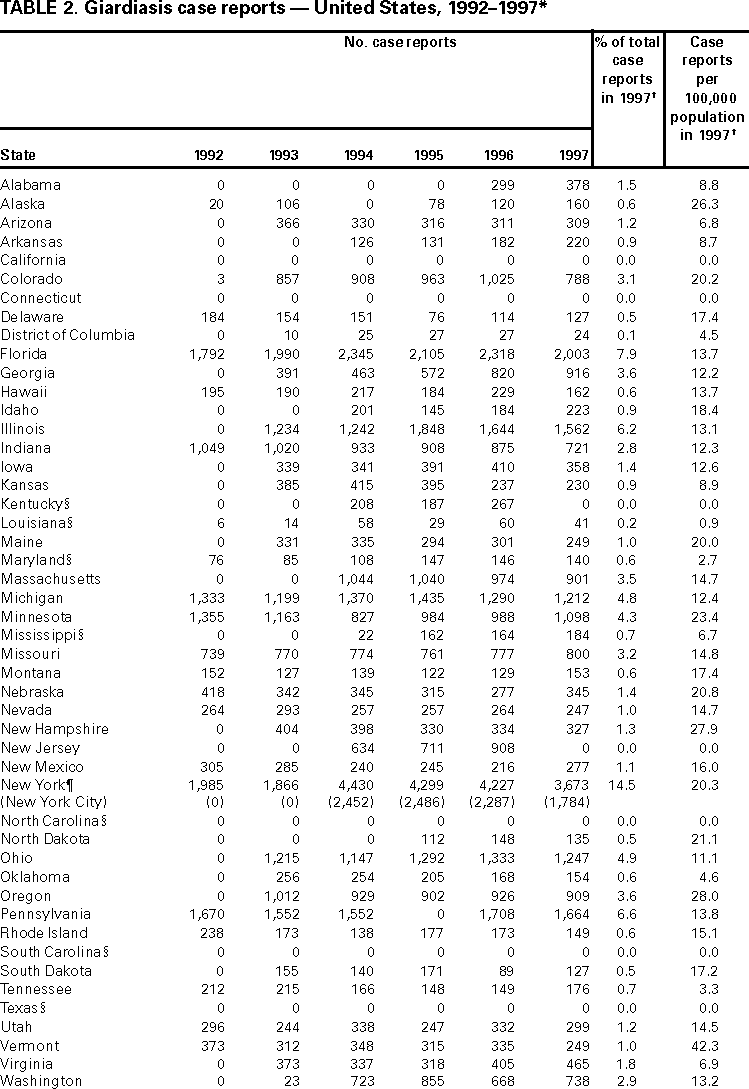  Return to top. Figure 2 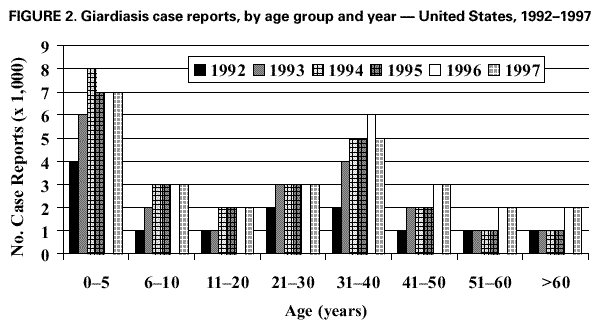 Return to top. Figure 3 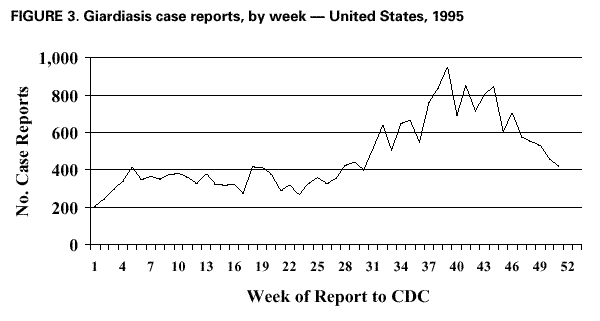 Return to top. Figure 4 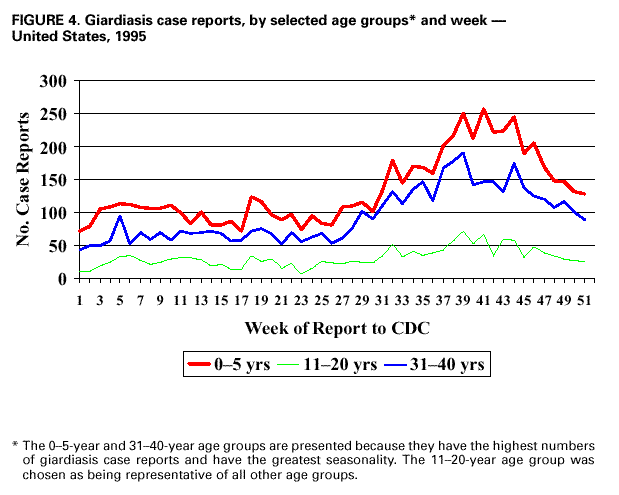 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 8/7/2000 |
|||||||||
This page last reviewed 5/2/01
|