08/09/2017: Lab Advisory: Cyclosporiasis Cases, Webinar: Zika MAC-ELISA, and WHO Editorial: Gaps in Lab Leadership

Dear Clinical and Public Health Laboratory Partners,
We would like share several updates: a Heath Alert Notice Advisory regarding Cyclosporiasis, a free webinar on quality control procedures for the CDC Zika MAC-ELISA, and WHO’s editorial regarding critical gaps in laboratory leadership to meet global health security goals.
Increase in Reported cases of Cyclospora cayetanensis Infection, United States, Summer 2017
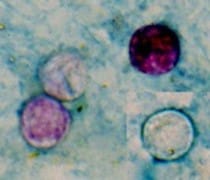
The Centers for Disease Control and Prevention (CDC), State and Local Health Departments, and the Food and Drug Administration (FDA) are investigating an increase in reported cases of cyclosporiasis. The purpose of CDC’s HAN Advisory is to notify public health departments and healthcare facilities, and to provide guidance to healthcare providers of the increase in reported cases. Please disseminate this information to healthcare providers in hospitals and emergency rooms, to primary care providers, and to microbiology laboratories.
Healthcare providers should consider a diagnosis of cyclosporiasis in patients with prolonged or remitting-relapsing diarrheal illness. Testing for Cyclospora is not routinely done in most U.S. laboratories, even when stool is tested for parasites. Healthcare providers must specifically order testing for Cyclospora, whether testing is requested by ova and parasite (O&P) examination, by molecular methods, or by a gastrointestinal pathogen panel test. Cyclosporiasis is a nationally notifiable disease; healthcare providers should report suspect and confirmed cases of infection to public health authorities.
Read more on CDC’s Cyclosporiasis website.
Free Webinar: Quality Control Procedures for the CDC Zika MAC-ELISA

Learn about the recommended additional Emergency Use Authorization (EUA) quality control check for Zika MAC-ELISA in this free webinar. The webinar will be available for 30 days.
For more information, please visit the CDC Types of Zika Virus Test webpage or visit APHL’s Zika Virus Response website.
WHO Editorial: Critical Gaps in Laboratory Leadership to Meet Global Health Security Goals

Public health laboratories play a critical role in the detection, prevention, and control of diseases. However, reliable laboratory testing continues to be limited in many low- and middle-income countries. The 2013–2016 Ebola virus disease outbreak in West Africa provided many examples of how functioning laboratories were needed for disease control and prevention efforts. This outbreak highlighted the need for laboratory directors to influence national laboratory policy, and to implement national laboratory strategic plans.
Read more on WHO’s Editorial webpage.
If you have any questions or feedback regarding LOCS, we’d like to hear from you. Please contact us at LOCS@cdc.gov.
Thank you,
The Laboratory Outreach Communication System (LOCS)