
DPDx is an education resource designed for health professionals and laboratory scientists. For an overview including prevention and control visit www.cdc.gov/parasites/amebiasis.
Amebiasis
[Entamoeba histolytica]
Laboratory Diagnosis
Entamoeba histolytica must be differentiated from other intestinal protozoa including: E. coli, E. hartmanni, E. gingivalis, Endolimax nana, and Iodamoeba buetschlii (the nonpathogenic amebae); Dientamoeba fragilis (which is a flagellate not an ameba); and the possibly pathogenic Entamoeba polecki. Differentiation is possible, but not always easy, based on morphologic characteristics of the cysts and trophozoites. The nonpathogenic Entamoeba dispar, however, is morphologically identical to E. histolytica, and differentiation must be based on isoenzymatic or immunologic analysis. Molecular methods are also useful in distinguishing between E. histolytica and E. dispar and can also be used to identify E. polecki. Microscopic identification of cysts and trophozoites in the stool is the common method for diagnosing E. histolytica. This can be accomplished using:
- Fresh stool: wet mounts and permanently stained preparations (e.g., trichrome).
- Concentrates from fresh stool: wet mounts, with or without iodine stain, and permanently stained preparations (e.g., trichrome). Concentration procedures, however, are not useful for demonstrating trophozoites.
In addition, E. histolytica trophozoites can also be identified in aspirates or biopsy samples obtained during colonoscopy or surgery.
Immunodiagnosis
Antibody Detection
Enzyme immunoassay (EIA) kits for Entomoeba histolytica antibody detection as well as EIA kits for antigen detection are commercially available in the United States. Antibody detection is most useful in patients with extraintestinal disease (i.e., amebic liver abscess) when organisms are not generally found on stool examination. Antigen detection may be useful as an adjunct to microscopic diagnosis in detecting parasites and can distinguish between pathogenic and nonpathogenic infections.
The indirect hemagglutination (IHA) test has been replaced by commercially available EIA test kits for routine serodiagnosis of amebiasis. Antigen consists of a crude soluble extract of axenically cultured organisms. The EIA test detects antibody specific for E. histolytica in approximately 95% of patients with extraintestinal amebiasis, 70% of patients with active intestinal infection, and 10% of asymptomatic persons who are passing cysts of E. histolytica. If antibodies are not detectable in patients with an acute presentation of suspected amebic liver abscess, a second specimen should be drawn 7-10 days later. If the second specimen does not show seroconversion, other agents should be considered. Detectable E. histolytica-specific antibodies may persist for years after successful treatment, so the presence of antibodies does not necessarily indicate acute or current infection. Specificity is 95% or higher: false-positive reactions rarely occur. Although the immunodiffusion test is as specific, it is slightly less sensitive than the IHA and EIA and requires a minimum of 24 hours to obtain a result, in contrast to 2 hours required for the IHA or EIA tests. However, the simplicity of the procedure makes it ideal for the laboratory that has only an occasional specimen to test. The IHA and EIA tests are more suitable for laboratories that have frequent requests for amebiasis serology. Although detection of IgM antibodies specific for E. histolytica has been reported, sensitivity is only about 64% in patients with current invasive disease. Several commercial EIA kits for antibody detection are available in the United States.
Antigen Detection
Antigen detection may be useful as an adjunct to microscopic diagnosis in detecting parasites and to distinguish between pathogenic and nonpathogenic infections. Recent studies indicate improved sensitivity and specificity of fecal antigen assays with the use of monoclonal antibodies which can distinguish between E. histolytica and E. dispar infections. At least one commercial kit is available which detects only pathogenic E. histolytica infection in stool; several kits are available which detect E. histolytica antigens in stool but do not exclude E. dispar infections.
References:
- Haque R, Ali IKM, Akther S, Petri WA Jr. Comparison of PCR, isoenzyme analysis, and antigen detection for diagnosis of Entamoeba histolytica infection. J Clin Microbiol 1998;36:449-452.
- Healy, GR. Serology. In: Ravdin JI, ed. Amebiasis: human infection by Entamoeba histolytica. New York: John Wiley & Sons; 1988. p. 650-719.
Molecular Diagnosis
Conventional PCR
In reference diagnosis laboratories, molecular analysis by PCR-based assays is the method of choice for discriminating between the pathogenic species (E. histolytica) and the nonpathogenic species (E. dispar).
Agarose gel (2%) analysis of a PCR diagnostic test for differentiation between E. histolytica and E. dispar.
- Lanes 1 - 4: Amplification with the Psp3/Psp51 PCR primer pair specific for E. histolytica. Diagnostic band size - 876 bp.
- Lanes 6 - 9: Amplification with the NPsp3/NPsp51 PCR primer pair specific for E. dispar. Diagnostic band size - 876 bp.
- Lanes 1 and 6: E. histolytica 200:NIH, zymodeme II (positive control for E. histolytica).
- Lanes 2 and 7: E. dispar 351:IMMiT, zymodeme I (positive control for E. dispar).
- Lanes 3 and 8: Specimen from a patient with a liver abscess (positive with E. histolytica primers and negative with E. dispar primers). E. histolytica 333:IMMiT, zymodeme XIV.
- Lanes 4 and 9: Specimen from an asymptomatic patient (positive with E. dispar primers and negative with E. histolytica primers). E. dispar 389:IMMiT, zymodeme I.
- Lane 5: Molecular base pair standard, 100-bp ladder (from 600 to 1,000 bp).
Real-Time PCR
A TaqMan real-time PCR approach has been validated at CDC and is used for differential laboratory diagnosis of amebiasis.2 The assay targets the 18S rRNA gene with species-specific TaqMan probes in a duplex format, making it possible to detect both species in the same reaction vessel.
More on: TaqMan real-time PCR
**** I made the link to TaqMan real time PCR more 508 and internet friendly ****
References:
- Clark CG, Diamond LS. Differentiation of pathogenic Entamoeba histolytica from other intestinal protozoa by riboprinting. Arch Med Res 1992;23:15-16.
- Qvarnstrom Y, James C, Xayavong M, Holloway B, Moura I, Visvesvara GS, et al. Comparison of real-time PCR rationales for differential laboratory diagnosis of amebiasis. J Clin Microbiol 2005;43:5491-5497.
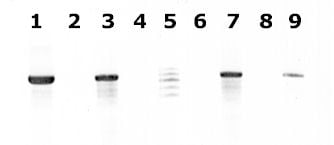
Figure contributed by Assist. Prof. Przemyslaw Myjak, Ph.D., Institute of Maritime and Tropical Medicine, Gdynia, Poland.
 ShareCompartir
ShareCompartir
 Laboratory Diagnosis of Amebiasis -- Entamoeba histolytica and Entamoeba dispar [PDF, 139 KB, 2 Pages]
Laboratory Diagnosis of Amebiasis -- Entamoeba histolytica and Entamoeba dispar [PDF, 139 KB, 2 Pages]

