FIGURE 1. Percentage of visits for influenza-like illness (ILI) reported by the U.S. Outpatient Influenza-Like Illness Surveillance Network (ILINet), by surveillance week --- United States, 2006--07, 2007--08, 2008--09, and 2009--10* influenza seasons
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail.
Update: Influenza Activity --- United States, August 30, 2009--March 27, 2010, and Composition of the 2010--11 Influenza Vaccine
The emergence and spread of 2009 pandemic influenza A (H1N1) virus resulted in substantial influenza activity in the United States throughout the summer and fall months of 2009, with activity peaking in late October. Activity declined beginning in November 2009 (1) but continued at lower levels through March 2010. The 2009 H1N1 virus remained the dominant circulating influenza virus throughout the season; <1% of characterized viruses were seasonal influenza A (H1), A (H3), and influenza B viruses. This report summarizes U.S. influenza activity* from August 30, 2009, the start of the 2009--10 influenza season, through March 27, 2010, and also reports on the 2010--11 Northern Hemisphere influenza vaccine strain selection.
Viral Surveillance
From August 30, 2009, through March 27, 2010, World Health Organization (WHO) and National Respiratory and Enteric Virus Surveillance System (NREVSS) collaborating laboratories in the United States tested 422,648 specimens. Of these, 89,585 (21.1%) were positive: 89,298 (99.7%) were positive for influenza A, and 287 (0.3%) were positive for influenza B. Among 66,978 influenza A viruses for which subtyping was performed, almost all (66,589 [99.4%]) were 2009 H1N1 viruses.
Of the 37,260 specimens reported during February 14--March 27, 2010, a total of 2,020 (5.4%) tested positive for influenza, of which 1,999 (98.9%) were positive for influenza A and 21 (1.0%) were positive for influenza B. Of the 1,510 influenza A viruses reported since mid-February for which subtyping was performed, almost all (1,506 [99.7%]) were 2009 H1N1 viruses. No seasonal influenza A (H1) viruses and only three influenza A (H3) viruses were reported. During February 14--March 27, states in the Southeast (Alabama, Florida, Georgia, Kentucky, Mississippi, North Carolina, South Carolina, and Tennessee) accounted for approximately 55% of the influenza positives reported but only 20% of the specimens tested.
Antigenic Characterization
States are requested to submit a subset of their influenza virus isolates to CDC for antigenic characterization. From August 30, 2009, through March 27, 2010, CDC antigenically characterized 1,647 influenza viruses submitted by WHO collaborating laboratories in the United States. These consisted of two seasonal influenza A (H1N1) viruses, 13 influenza A (H3N2), 23 influenza B, and 1,609 2009 influenza A (H1N1) viruses. Both seasonal influenza A (H1N1) viruses tested were related to the influenza A (H1N1) component of the 2009--10 Northern Hemisphere influenza vaccine (A/Brisbane/59/2007). The 13 influenza A (H3N2) viruses tested showed reduced titers with antisera produced against A/Brisbane/10/2007, the 2009--10 Northern Hemisphere influenza A (H3N2) vaccine component, and were antigenically related to A/Perth/16/2009, the WHO-recommended influenza A (H3N2) component of the 2010 Southern Hemisphere and 2010--11 Northern Hemisphere vaccine formulations.
Globally circulating influenza B viruses can be divided into two distinct lineages represented by the B/Yamagata/16/88 and B/Victoria/02/87 viruses. The influenza B component of the 2009--10 and 2010--11 Northern Hemisphere vaccines belongs to the B/Victoria lineage. The 23 influenza B viruses characterized to date this season belong to the B/Victoria lineage and are related to the influenza vaccine component for the 2009--10 and 2010--11 Northern Hemisphere influenza B vaccine strain (B/Brisbane/60/2008). Of the 1,609 2009 H1N1 viruses tested, nearly all (1,604 [99.7%]) were related to the A/California/07/2009 (H1N1) reference virus selected by WHO as the 2009 H1N1 virus vaccine component for the 2010--11 Northern Hemisphere vaccine. Five viruses (0.3%) tested showed reduced titers with antiserum produced against A/California/07/2009; these were collected in September (two), October (one), November (one), and December (one) 2009.
Antiviral Resistance of Influenza Virus Isolates
CDC conducts surveillance for resistance of circulating influenza viruses to both classes of influenza antiviral medications: adamantanes (amantadine and rimantadine) and neuraminidase inhibitors (zanamivir and oseltamivir). A total of 64 oseltamivir-resistant 2009 H1N1 viruses have been identified in the United States since April 2009, with 55 identified from specimens collected after August 30, 2009. This number of oseltamivir-resistant 2009 H1N1 viruses might overestimate the prevalence of oseltamivir-resistant 2009 H1N1 viruses in the United States because most cases were selected for testing because of clinical suspicion for oseltamivir resistance. In 52 (81.3%) of the 64 identified cases of oseltamivir resistance, patients had documented exposure to oseltamivir through either treatment or chemoprophylaxis; eight patients have yet to have their exposure to oseltamivir determined, three patients had no known exposure, and oseltamivir exposure for one patient could not be determined. One seasonal influenza A (H1N1) virus was tested and found to be resistant to oseltamivir, whereas none of the 12 influenza A (H3N2) or 11 influenza B viruses tested was resistant to oseltamivir. All tested viruses were sensitive to the neuraminidase inhibitor zanamivir. The single seasonal influenza A (H1N1) virus tested was collected on September 8, 2009, and found to have sensitivity to the adamantanes. However, all 12 influenza A (H3N2) virus isolates and 1,491 (99.7%) of 1,495 2009 H1N1 virus isolates tested were found to have resistance to the adamantanes.
Outpatient Illness Surveillance
The weekly percentage of outpatient visits for influenza-like-illness (ILI)† reported by the U.S. Outpatient ILI Surveillance Network (ILINet) exceeded baseline levels§ (2.3%) for 18 weeks during the 2009--10 season and peaked at 7.7% in the week ending October 24, 2009. Since that time, ILI has declined and was at 1.6% in the week ending March 27, 2010 (Figure 1). On a regional level, the percentage of outpatient visits for ILI ranged from 0.4% to 3.3% during the week ending March 27, 2010. One of the 10 U.S. Department of Health and Human Services (HHS) regions (Region 9, comprised of Arizona, California, Hawaii, Nevada, American Samoa, Guam, Los Angeles County, the Commonwealth of Northern Mariana Islands, the Federal States of Micronesia, the Republic of Marshall Islands, and Palau) reported ILI at or above its region-specific baseline during that week.
Influenza-Associated Hospitalizations
Laboratory-confirmed influenza-associated hospitalizations are monitored using a population-based surveillance network that includes the 13 Emerging Infections Program (EIP) sites in 10 states and six new sites added during 2009.¶ This season's cumulative hospitalization rates from August 30, 2009, through the week ending March 27, 2010, remain highest in children aged 0--4 years and generally decline with increasing age. Cumulative rates of laboratory-confirmed, influenza-associated hospitalizations reported for children aged 0--4 years were 6.6 per 10,000 population in EIP sites and 10.5 per 10,000 population in the new sites (Figure 2). Rates for other age groups were as follows: 5--17 years, 2.5 in EIP and 3.6 in the new sites; 18--49 years, 2.4 in EIP and 1.7 in the new sites; 50--64 years, 3.2 in EIP and 2.0 in the new sites; and ≥65 years, 2.7 in EIP and 1.8 in the new sites.
In response to the emergence and widespread circulation of the 2009 H1N1 virus, the Council of State and Territorial Epidemiologists (CSTE) implemented reporting of influenza-associated hospitalizations and deaths to CDC. On August 30, CDC and CSTE instituted modified case definitions for aggregate reporting of influenza-associated hospitalizations and deaths. This cumulative jurisdiction-level reporting is referred to as the Aggregate Hospitalization and Death Reporting Activity (AHDRA).** During August 30, 2009--March 27, 2010, a median of 34 states each week reported a total of 41,689 hospitalizations associated with laboratory-confirmed influenza virus infections to CDC through AHDRA. Rates of hospitalization through AHDRA were highest among children aged 0--4 years (71.5 per 100,000) and ranged from 23.2 to 30.00 per 100,000 for older children and adults. Overall, weekly reported rates have declined consistently since January 2, 2010.
Pneumonia- and Influenza-Related Mortality
Pneumonia- and influenza-associated deaths are monitored by the 122 Cities Mortality Reporting System and AHDRA. For the week ending March 27, 2010, pneumonia or influenza was reported as an underlying or contributing cause of death for 7.9% of all deaths reported through the 122 Cities Mortality Reporting System, slightly above the week-specific epidemic threshold of 7.8%†† but below the threshold for the preceding 7 weeks (Figure 3). Pneumonia- and influenza-related mortality was above the epidemic threshold for 13 consecutive weeks from the week ending October 3, 2009, through the week ending December 12, 2009, was below threshold for 2 weeks, and above the threshold again during January 10--30, 2010 (epidemiologic weeks 2--4).
During August 30, 2009--March 27, 2010, a total of 2,096 deaths associated with laboratory-confirmed influenza virus infections were reported to CDC through AHDRA. The median number of states reporting laboratory-confirmed deaths each week to AHDRA was 36. Cumulative influenza-associated death rates since August 30, 2009, were highest among persons aged 50--64 years (1.56 per 100,000) and lowest in children (0.43 per 100,000 for children aged 0--4 years and 0.36 for children aged 5--18 years). Among persons aged 19--24 years, 25--49 years, and ≥65 years, cumulative influenza-associated mortality rates per 100,000 persons were 0.54, 0.87, and 0.95, respectively. Weekly reported death rates have declined steadily since November 2009 and are at their lowest level since the implementation of AHDRA in August 2009.
Influenza-Associated Pediatric Mortality
CDC has received 269 reports of pediatric deaths associated with laboratory-confirmed influenza infection that occurred since August 30, 2009, the start of the 2009--10 influenza season (Figure 4). A total of 219 (81%) cases were associated with infection with laboratory-confirmed 2009 H1N1 virus, and 49 (18%) were associated with an influenza A infection for which the subtype was undetermined. These deaths occurred during times when approximately 99% of subtyped influenza A viruses were 2009 H1N1 and were therefore likely to be associated with 2009 H1N1 because of the predominance of this virus. One death was associated with an influenza B virus infection.
Of the 269 reported pediatric deaths that occurred since August 30, 2009, a total of 48 (18%) were among children aged <2 years, 30 (11%) were among children aged 2--4 years, 100 (37%) were among children aged 5--11 years, and 91 (34%) were among children aged 12--17 years. A medical history was reported for 263 of the 269 decedents (98%). Of these 263 decedents, 182 (69%) had one or more medical conditions associated with an increased risk for influenza-related complications (2).
Since the week ending April 26, 2009, CDC has received 280 reports of pediatric deaths associated with laboratory-confirmed 2009 H1N1 virus. CDC also has received reports of 51 deaths with laboratory-confirmed influenza A for which subtype information was not available.
State-Specific Activity Levels
The largest number of states to date reporting widespread activity occurred during the week ending October 24, 2009, when 49 jurisdictions reported widespread activity.§§ During the week ending March 27, 2010, no states reported widespread influenza activity; three states reported regional influenza activity (Alabama, Georgia, and South Carolina); Puerto Rico and seven states (Arkansas, Hawaii, Louisiana, Maine, North Carolina, Tennessee, and Virginia) reported local influenza activity; the District of Columbia, Guam, and 30 states reported sporadic influenza activity; and 10 states reported no influenza activity. During the 2009--10 season, regional or widespread influenza activity has been reported during at least 1 week from all 50 states.
Although most states have experienced steady declines in influenza activity since November 2009, sustained activity has been observed in some parts of the United States. In HHS Region 4 (consisting of Alabama, Florida, Georgia, Kentucky, Mississippi, North Carolina, South Carolina, and Tennessee), the proportion of ILINet provider visits for ILI was at or above the regional baseline level (2%) for 12 of the 17 preceding weeks. From early December 2009 through the week ending March 27, 2010, while influenza activity in most of the rest of the country declined to sporadic or none, several southeastern states continued to report local or regional activity. Beginning February 2010, the number of influenza-associated hospitalizations increased in Alabama, Georgia, Arkansas, Louisiana, Mississippi, and South Carolina, compared with the immediately preceding weeks. Georgia, in particular, had a moderate but steady increase in the number of hospitalizations reported to AHDRA from mid-February through March 27. During the week ending March 27, Georgia reported 16 hospitalizations, a decrease from a median of 38 per week during the preceding 5 weeks.
Composition of the 2010--11 Influenza Vaccine
WHO has recommended vaccine strains for the 2010--11 Northern Hemisphere trivalent influenza vaccine, and the Food and Drug Administration has made the same recommendations for influenza vaccine composition for the United States. Both agencies recommend that vaccines contain A/California/7/2009-like (2009 H1N1), A/Perth/16/2009-like (H3N2), and B/Brisbane/60/2008-like (B/Victoria lineage) viruses. A seasonal influenza A (H1N1) component is not included in the 2010--11 formulation, and the A (H3N2) component has been changed from A/Brisbane/59/2007 in the 2009--10 Northern Hemisphere vaccine formulation. This recommendation was based on surveillance data related to epidemiology and antigenic characteristics, serologic responses to 2009--10 trivalent seasonal and 2009 H1N1 monovalent vaccines, and the availability of candidate strains and reagents (3).
Reported by
WHO Collaborating Center for Surveillance, Epidemiology, and Control of Influenza. M Jhung, MD, L Brammer, MPH, S Epperson, MPH, L Blanton, MPH, R Dhara, MPH, T Wallis, MS, A Fiore, MD, L Gubareva, PhD, J Bresee, MD, L Kamimoto, MD, X Xu, MD, A Klimov, PhD, N Cox, PhD, L Finelli, DrPH, Influenza Div, National Center for Immunization and Respiratory Diseases; R Njai, PhD, EIS Officer, CDC.
Editorial Note
With the emergence of the 2009 H1N1 virus in April 2009, influenza activity increased and remained at higher than normal levels during the spring and summer until peaking in late October and early November 2009. Since that time, influenza activity has declined nationwide and currently is below epidemic thresholds or baseline levels across most influenza surveillance systems. In contrast to seasonal influenza, which usually peaks once during the first few months of the calendar year, the overall course of the 2009 H1N1 pandemic occurred in two distinct waves, a spring 2009 wave peaking in June and a second, larger, fall 2009 wave, peaking in late October. Similar two-wave patterns were observed in the 1918--19 H1N1 and the 1957--58 H2N2 pandemics in the United States (4). The magnitude and timing of ILI activity during the current season also were substantially different from those observed in previous years. ILI activity for the 2009--10 H1N1 influenza season peaked in late October at 7.7%, whereas activity typically peaks in February and in previous seasons has been much lower (3.5% in the 2008--09 season, 6% in the 2007--08 season, 3.6% in the 2006--07 season, and 3.3% in the 2005--06 season).
The magnitude and age distribution of influenza-associated hospitalizations during the 2009--10 H1N1 season were different compared with previous seasons. Hospitalization rates reported to EIP sites during the current season were much higher than previous seasons across most age groups. In 2008--09, when seasonal H1N1 was the predominant strain, hospitalizations occurred at approximately one sixth the 2009--10 rate for persons aged 18--49 years and one half the 2009--10 rate for those aged 0--4 years and ≥65 years. Similarly, in 2007--08, when H3N2 virus was predominant, hospitalizations occurred at approximately one quarter the 2009--10 rate for persons aged 5--17 years and one half the 2009--10 rate for those aged 0--4 years; however, the hospitalization rate for persons aged ≥65 years in 2007--08 was 2.6 times that of the rate reported for the current season. Moreover, although during typical influenza seasons the majority of influenza-associated hospitalizations occur among adults aged ≥65 years (5,6), hospitalizations among younger persons accounted for the majority of hospitalizations during the 2009 H1N1 pandemic (7). The relatively higher burden of disease borne by younger age groups might be due, in part, to previous exposure of older persons to viruses antigenically similar to 2009 H1N1 virus (8).
By March 27, 2010, national influenza activity had decreased to the lowest level measured during the 2009--10 season; however, isolated areas experienced sustained transmission during recent winter months. Notably, states in the southeastern United States (Alabama, Georgia, Mississippi, and South Carolina) continued to report elevated rates of influenza activity, influenza-associated hospitalizations, and increased prevalence of ILI compared with baseline. Although continued focal transmission of 2009 H1N1 influenza during the winter months is not unexpected, trends in the southeast region and particularly Georgia highlight the need to maintain public health surveillance and continue to offer 2009 H1N1 vaccine. The basis for sustained activity in the southeast is unclear but might be related, in part, to lower community attack rates in the spring and summer and lower vaccination rates, leading to an overall more susceptible population in the region (9).
Vaccination with 2009 H1N1 vaccine remains the key strategy for prevention of 2009 H1N1 influenza infection (2). Although national influenza activity has decreased substantially since peak activity in October 2009, the persistence of sustained transmission in some areas (resulting in additional cases, hospitalizations, and deaths) emphasizes the importance of a continued focus on vaccination of initial target groups as well as the rest of the susceptible population.
Changes in the geographic spread, type, subtype, and severity of the circulating influenza viruses will continue to be monitored and reported weekly in the online national influenza surveillance summary, FluView.¶¶ Additional information regarding prevention and treatment of the 2009 H1N1 influenza infection also is available online.***
Acknowledgments
This report is based, in part, on contributions by participating state and territorial health departments and state public health laboratories, World Health Organization collaborating laboratories, National Respiratory and Enteric Virus Surveillance System collaborating laboratories, the U.S. Outpatient ILI Surveillance Network, the Emerging Infections Program, the Aggregate Hospitalization and Death Reporting Activity, the Influenza Associated Pediatric Mortality Surveillance System, and the 122 Cities Mortality Reporting System.
References
- CDC. Update: influenza activity---United States, August 30, 2009--January 2, 2010. MMWR 2010;59:38--43.
- CDC. Use of influenza A (H1N1) 2009 monovalent vaccine: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2009. MMWR 2009;58(No. RR-10).
- World Health Organization. Recommended viruses for influenza vaccines for use in the 2010--2011 northern hemisphere influenza season. Geneva, Switzerland: World Health Organization; 2010. Available at http://www.who.int/csr/disease/influenza/recommendations2010_11north/en/index.html. Accessed April 12, 2010.
- Kilbourne ED. Influenza pandemics of the 20th century. Emerg Infect Dis 2006;12:9--14.
- Thompson WW, Shay DK, Weintraub E, et al. Influenza-associated hospitalizations in the United States. JAMA 2004;292:1333--40.
- Thompson WW, Shay DK, Weintraub E, et al. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA 2003;289:179--86.
- Jain S, Kamimoto L, Bramley AM, et al. Hospitalized patients with 2009 H1N1 influenza in the United States, April--June 2009. N Engl J Med 2009;361:1935--44.
- Hancock K, Veguilla V, Lu X, et al. Cross-reactive antibody responses to the 2009 pandemic H1N1 influenza virus. N Engl J Med 2009;361:1945--52.
- CDC. Interim Results: state-specific influenza A (H1N1) 2009 monovalent vaccination coverage----United States, October 2009--January 2010. MMWR 2010;59:363--8.
* The CDC influenza surveillance system collects five categories of information from eight data sources: 1) viral surveillance (World Health Organization collaborating U.S. laboratories, the National Respiratory and Enteric Virus Surveillance System, and novel influenza A virus case reporting); 2) outpatient illness surveillance (U.S. Outpatient ILI Surveillance Network); 3) mortality (122 Cities Mortality Reporting System and influenza-associated pediatric mortality reports); 4) hospitalizations (Emerging Infections Program); and 5) summary of geographic spread of influenza (state and territorial epidemiologist reports).
† Defined as a temperature of ≥100.0°F (≥37.8°C), oral or equivalent, and cough and/or sore throat, in the absence of a known cause other than influenza.
§ The national and regional baselines are the mean percentage of visits for ILI during noninfluenza weeks for the previous three seasons plus two standard deviations. A noninfluenza week is a week during which <10% of specimens tested positive for influenza. National and regional percentages of patient visits for ILI are weighted on the basis of state population. Use of the national baseline for regional data is not appropriate.
¶ EIP currently conducts surveillance for laboratory-confirmed, influenza-related hospitalizations in 61 counties and Baltimore, Maryland. The EIP catchment area includes 13 metropolitan areas located in 10 states. Beginning in September 2009, six new EIP sites covering 40 counties began reporting influenza-related hospitalization surveillance. Hospital laboratory, admission, and discharge databases, and infection-control logs are reviewed to identify persons with a positive influenza test (i.e., viral culture, direct fluorescent antibody assays, real-time reverse transcription--polymerase chain reaction, serology, or a commercial rapid antigen test) from testing conducted as part of their routine care.
** States report weekly to CDC either 1) laboratory-confirmed influenza hospitalizations and deaths, or 2) pneumonia and influenza syndrome-based cases of hospitalization and death resulting from all types or subtypes of influenza. Although only the laboratory-confirmed cases are included in this report, CDC continues to analyze data both from laboratory-confirmed and syndromic hospitalizations and deaths.
†† The seasonal baseline proportion of pneumonia and influenza deaths is projected using a robust regression procedure, in which a periodic regression model is applied to the observed percentage of deaths from pneumonia and influenza that were reported by the 122 Cities Mortality Reporting System during the preceding 5 years. The epidemic threshold is 1.645 standard deviations above the seasonal baseline.
§§ Levels of activity are 1) no activity; 2) sporadic: isolated laboratory-confirmed influenza cases or a laboratory-confirmed outbreak in one institution, with no increase in ILI activity; 3) local: increased ILI, or at least two institutional outbreaks (ILI or laboratory-confirmed influenza) in one region with recent laboratory evidence of influenza in that region, with virus activity no greater than sporadic in other regions; 4) regional: increased ILI activity or institutional outbreaks (ILI or laboratory-confirmed influenza) in at least two but less than half of the regions in the state, with recent laboratory evidence of influenza in those regions; and 5) widespread: increased ILI activity or institutional outbreaks (ILI or laboratory-confirmed influenza) in at least half the regions in the state, with recent laboratory evidence of influenza in the state.
¶¶ Available at http://www.cdc.gov/flu/weekly.
*** Available at http://www.cdc.gov/h1n1flu.
What is already known on this topic?
The 2009 pandemic influenza A (H1N1) virus emerged in April 2009 and caused substantial disease in the United States and worldwide.
What is added by this report?
Although recent declines in influenza activity have been observed, 2009 H1N1 viruses continue to circulate, particularly in the southeastern United States, and influenza-associated hospitalizations and deaths continue to be reported.
What are the implications for public health practice?
Epidemiologic data in this report support recommendations by CDC that the 2009 H1N1 vaccine continue to be offered to all persons aged ≥6 months, with children aged <10 years requiring 2 doses of the vaccine approximately 1 month apart.
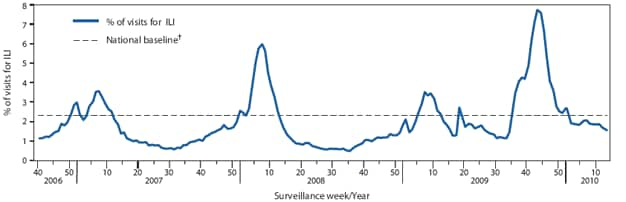
* Through March 27, 2010.
† The national baseline is the mean percentage of visits for ILI during noninfluenza weeks for the previous three seasons plus two standard deviations. A noninfluenza week is a week during which <10% of specimens tested positive for influenza. Use of the national baseline for regional data is not appropriate.
Alternate Text: The figure above shows the percentage of visits for influenza-like illness (ILI) reported by the U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet), by surveillance week in the United States for the 2006-07, 2007-08, 2008-09, and 2009-10 influenza seasons. In the week ending October 24, 2009, the weekly percentage of outpatient visits for ILI reported by the U.S. Outpatient ILINet reached 7.7%, the highest level to date this influenza season and a level higher than the three preceding seasons. After peaking, the ILI level decreased to 1.6% in the week ending March 27, 2010.
FIGURE 2. Number of laboratory-confirmed influenza-associated hospitalizations and cumulative hospitalization rates per 10,000 population, by age group and surveillance week --- Emerging Infections Program (EIP) and new sites,* 2006--07, 2007--08, 2008--09,† and 2009--10§ U.S. influenza seasons
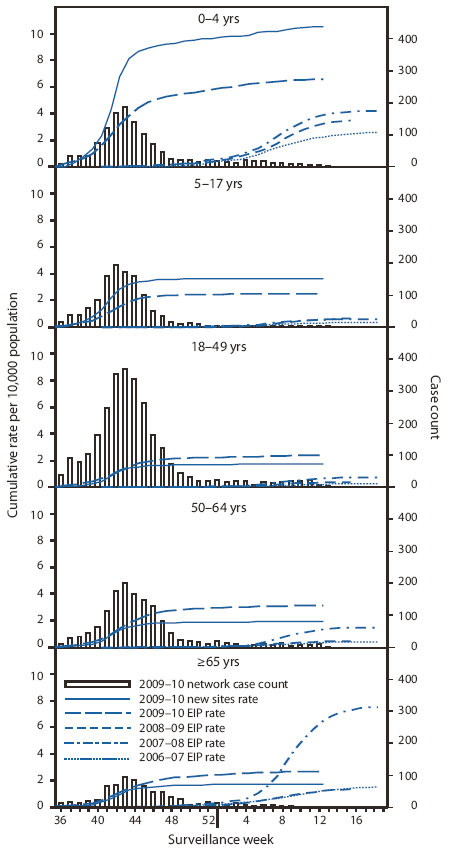
* In 2009, new sites in six additional states were added to the sites in the 10 states already participating in EIP. During August 30, 2009--March 27, 2010, total influenza-associated hospitalization rates were reported for EIP and the new sites for all types of influenza, including influenza A, influenza B, and 2009 pandemic influenza A (H1N1).
† Ending April 14, 2009, with reports of cases of 2009 H1N1.
§ Through March 27, 2010.
Alternate Text: The figure above shows the number of 2009-10 laboratory-confirmed influenza-associated hospitalizations and cumulative hospitalization rates per 10,000 population, by age group and surveillance week from Emerging Infections Program (EIP), and new sites for the 2006-07, 2007-08, 2008-09, and 2009-10 influenza seasons. As of March 27, 2010, cumulative rates of laboratory-confirmed influenza-associated hospitalizations reported for children aged 0-4 years were 6.6 per 10,000 population by EIP and 10.5 per 10,000 population by the new sites.
FIGURE 3. Percentage of all deaths attributed to pneumonia and influenza (P&I), by surveillance week and year --- 122 Cities Mortality Reporting Sytem, United States, 2005--2010
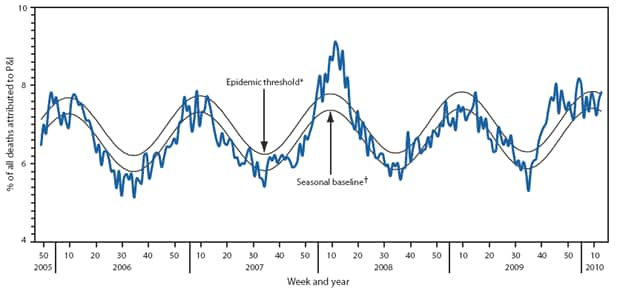
* The epidemic threshold is 1.645 standard deviations above the seasonal baseline.
† The seasonal baseline is projected using a robust regression procedure that applies a periodic regression model to the observed percentage of deaths from P&I during the preceding 5 years.
Alternate Text: The figure above shows the percentage of all deaths attributed to pneumonia and influenza (P&I), by surveillance week and year for 122 U.S. cities from 2006-2010 from the Mortality Reporting System. For the week ending March 27, pneumonia or influenza was reported as an underlying or contributing cause of death for 7.9% of all deaths reported through the 122 Cities Mortality Reporting System, above the week-specific epidemic threshold of 7.8%.
FIGURE 4. Number of influenza-associated pediatric deaths, by week of death --- United States, 2006--07, 2007--08, 2008--09, and 2009--10* influenza seasons
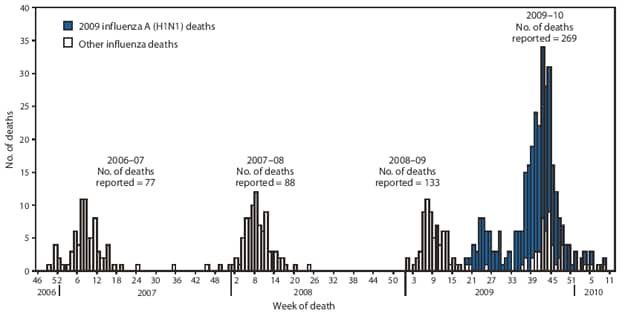
* Through March 27, 2010.
Alternate Text: The figure above shows the number of influenza-associated pediatric deaths, by week of death in the United States for the 2006-07, 2007-08, 2008-09, and 2009-10 influenza seasons. CDC has received 269 reports of pediatric deaths associated with laboratory-confirmed influenza infection that occurred and were reporting since August 30, 2009.
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of
Health and Human Services.
References to non-CDC sites on the Internet are
provided as a service to MMWR readers and do not constitute or imply
endorsement of these organizations or their programs by CDC or the U.S.
Department of Health and Human Services. CDC is not responsible for the content
of pages found at these sites. URL addresses listed in MMWR were current as of
the date of publication.
All MMWR HTML versions of articles are electronic conversions from typeset documents.
This conversion might result in character translation or format errors in the HTML version.
Users are referred to the electronic PDF version (http://www.cdc.gov/mmwr)
and/or the original MMWR paper copy for printable versions of official text, figures, and tables.
An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S.
Government Printing Office (GPO), Washington, DC 20402-9371;
telephone: (202) 512-1800. Contact GPO for current prices.
**Questions or messages regarding errors in formatting should be addressed to
mmwrq@cdc.gov.


