Multistate Outbreak of Fungal Meningitis and Other Infections –Resources for Laboratories
On October 30, 2015, CDC updated its web resources for patients and clinicians. Patients affected by tainted steroid injections from the New England Compounding Center continue to receive treatment for their infections and clinicians should continue to monitor patient recovery. All relevant materials for patients and clinicians concerning the multistate outbreak of fungal meningitis and other infections are located on this page.
Testing and Results
Pathologic Evaluation
Pathologic Results
Microscopic Gallery
Information on this site is focused on important laboratory findings and guidance related to the outbreak of fungal meningitis and other infections linked to the use of injectable steroids from three recalled lots of preservative-free methylprednisolone acetate (MPA) distributed by the New England Compounding Center (NECC).
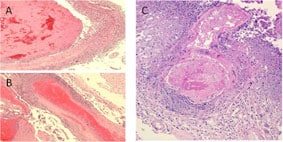
Major histopathologic findings in clinical cases of meningitis show evidence of necrotizing, suppurative vasculitis with thrombosis (A and B). These findings are seen in many cases involving a branch of the basilar artery (C), which are consistent with the clinical findings.
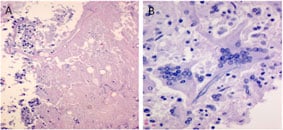
Another finding in the basilar artery is necrosis (A) and giant cell arteritis (B).
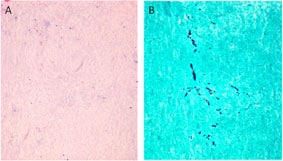
Arachnoiditis is a complication present in some of the patients. In tissue obtained adjacent to the affected site, dense collagen-rich soft tissue with no inflammatory infiltrate (A) is seen. Fungal hyphae are seen in this tissue by silver stain (GMS) with modified oxidation times (B).
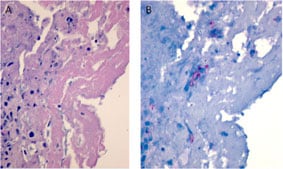
In patients with arthritis, fibrin and necrotic debris are seen in affected joints (A). Although rare, immunohistochemistry facilitates the focal detection of hyphae (B) in these areas (red staining).
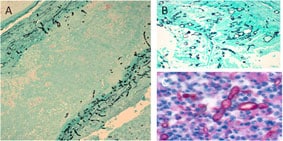
Fungal hyphae (black) can be visualized with silver stain within vessel walls (A) and in area of necrosis in basilar artery (B). Using a polyfungal immunohistochemistry reagent, fungal hyphae (red) is also seen in the purulent exudate in spinal meningitis (C).
CDC Morbidity and Mortality Weekly Report (MMWR)
- MMWR: Multistate Outbreak of Fungal Infection Associated with Injection of Methylprednisolone Acetate Solution from a Single Compounding Pharmacy — United States, 2012
- MMWR: Multistate Fungal Meningitis Outbreak — Interim Guidance for Treatment
CDC Resources
- Frequently Asked Questions for Patients
- CDC Digital Press Kit with Laboratory Photos and Footage
- CDC Fungal Diseases
- CDC Fungal Meningitis
- CDC Information About Additional Medical Products (non-MPA) From New England Compounding Center
- CDC Information About Voluntary Recall of All Ameridose Medical Products
Select Publications
- Gade L, Grgurich DE, Kerkering TM, Brandt ME, Litvintseva AP. Utility of Real-Time PCR for Detection of Exserohilum rostratum in Body and Tissue Fluids during the Multistate Outbreak of Fungal Meningitis and Other Infections. J Clin Microbiol 2015 Feb.external icon
- Gade L, Scheel CM, Pham CD, et al. Detection of fungal DNA in human body fluids and tissues during a multistate outbreak of fungal meningitis and other infections. Eukaryotic Cell 2013 Mar.external icon
- Kainer MA, Reagan DR, Nguyen DB, et al. Fungal infections associated with contaminated methylprednisolone in Tennessee. New Engl J Med 2012 Nov.external icon
- Litvintseva AP, Hurst S, Gade L, et al. Whole-genome analysis of Exserohilum rostratum from an outbreak of fungal meningitis and other infections. J Clin Microbiol 2014 Sep.external icon
- Litvintseva AP, Lindsley MD, Gade L, et al. Utility of (1–3)-β-d-Glucan Testing for Diagnostics and Monitoring Response to Treatment During the Multistate Outbreak of Fungal Meningitis and Other Infections. Clin Infect Dis 2013 Dec.external icon
- Lockhart SR, Pham CD, Gade L, et al. Preliminary Laboratory Report of Fungal Infections Associated with Contaminated Methylprednisolone Injections. J Clin Microbiol 2013 Augexternal icon.
- Lyons JL, Gireesh ED, Trivedi JB, et al. Fatal Exserohilum meningitis and central nervous system vasculitis after cervical epidural methylprednisolone injection. Ann Intern Med 2012 Oct.external icon
- Pettit AC, Kropski JA, Castilho JL, et al. The index case for the fungal meningitis outbreak in the United States. NEJM 2012 Oct.external icon
